CHAPTER 6
Atrioventricular Septal Defects
Definition
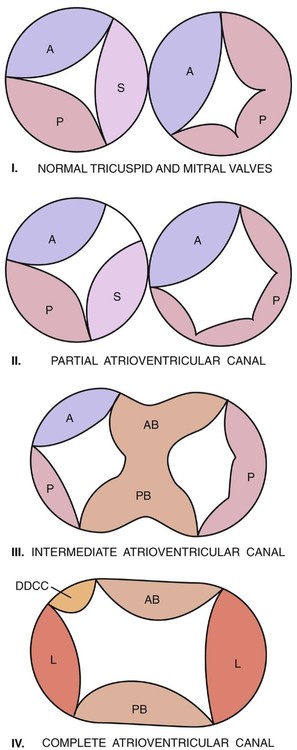
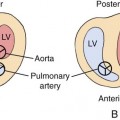
Atrioventricular septal defect (AVSD) refers to a spectrum of cardiac malformations that include abnormalities of the interatrial septum, the interventricular septum, and the atrioventricular (AV) (mitral and tricuspid) valves. An AVSD results from the endocardial cushions of the heart failing to fuse properly. AVSDs are also referred to as AV canal defects or endocardial cushion defects. AVSDs are usually categorized as partial, intermediate, or complete (Fig. 6–1). All forms tend to be large defects, and they share many similarities.1–12
Complete Atrioventricular Septal Defect
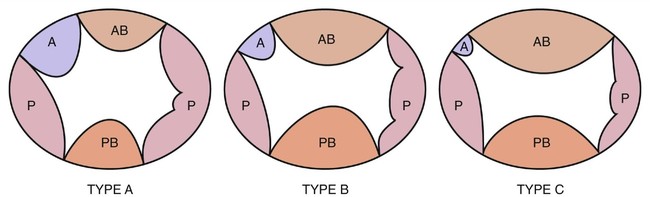
Rastelli et al13,14 and Calabrò and Limongelli15 have subclassified the complete form of AVSD on the basis of the chordal insertions and the degree of bridging of the anterosuperior bridging leaflet of the common AV valve (Fig. 6–2).
In type A, the anterosuperior bridging leaflet is attached almost entirely to the left ventricle, and its commissure with the anterior tricuspid leaflet lies along the right anterosuperior rim of the ventricular septum. Beneath this commissure, multiple direct chordal insertions into the septum usually exist. Interventricular communication beneath the anterior bridging leaflet is not as significant as that seen in type B or C defects. A type A AVSD usually occurs as an isolated defect or in association with Down syndrome (trisomy 21). Most type A AVSDs are not associated with other cardiac malformations; however, they have been associated with left-sided obstructions.10,11,15–18
Type B AVSD is the rarest form of a complete defect; it consists of a large anterior septal bridging leaflet overhanging the ventricular septum to a greater degree than occurs in type A. The medial papillary muscle is usually attached apically on the septal band or on the moderator band. Because there are no chordal attachments between the anterosuperior bridging leaflet and the underlying ventricular septum, free interventricular communication exists.15
Type C AVSD consists of an anterosuperior bridging leaflet that is larger and overhangs the ventricular septum to a greater degree than in either type A or type B. In type C AVSD, the medial papillary muscle attaches to the anterior tricuspid papillary muscle and the anterior tricuspid leaflet is usually very small. Free interventricular communication is also present in type C defects. Type C AVSDs are frequently associated with other congenital cardiac abnormalities such as tetralogy of Fallot, double-outlet right ventricle, complete transposition of the great arteries, and heterotaxic syndromes (asplenia and polysplenia).1,2,15,18–22 Complete AVSD occurring with tetralogy of Fallot is usually seen in patients with Down syndrome, whereas complete AVSD with double-outlet right ventricle is more commonly seen in the asplenia syndrome.9,11,17
AVSD can be further described as either balanced or unbalanced. In most cases of AVSD, the AV junction is connected equally to the right and left ventricles so that each ventricle receives a similar amount of blood and the ventricles are symmetrical in size. This is known as a balanced AVSD. An unbalanced AVSD occurs when the AV junction is predominantly committed to either the right or left ventricle, leading to hypoplasia of the opposing ventricle receiving the smaller amount of blood.18,23
In all types of complete AVSDs, the right atrium and pulmonary arteries are usually dilated, with the right ventricular outflow tract bulging anteriorly. The right ventricle may be hypertrophied. The left atrium may also be dilated. Because of the loss of normal AV valve continuity, the left ventricular outflow tract will usually appear as a long, narrow chamber. Dextroposition of the aorta is commonly associated with complete AVSD.24
Partial Atrioventricular Septal Defect
The partial form of AVSD differs from the complete form in that the mitral and tricuspid orifices are distinct and separate. It is usually defined by four criteria, which may occur alone or in any combination: (1) primum atrial septal defect, (2) ventricular septal defect, (3) cleft anterior mitral leaflet, and (4) widened anteroseptal tricuspid commissure (cleft septal tricuspid leaflet). The most commonly occurring form of partial AVSD consists of a primum atrial septal defect and a cleft anterior mitral leaflet. A partial AVSD is also known as an ostium primum atrial septal defect.15,25
This differs from an isolated mitral cleft (not associated with AVSD), in which the cleft is directed toward the aortic valve annulus. Associated anomalies commonly include secundum atrial septal defects and a persistent left superior vena cava that drains into the coronary sinus. Less commonly associated cardiac defects include pulmonary stenosis, discrete subaortic stenosis, tricuspid stenosis or atresia, coarctation of the aorta, a membranous ventricular septal defect, and hypoplastic left ventricle.15,21,22,24–26
Intermediate Atrioventricular Septal Defect
The intermediate type of AVSD occurs least frequently. It is similar to the complete form; however, the anterior and posterior septal bridging leaflets are fused above the ventricular septum, dividing the common AV valve into separate mitral and tricuspid components.1
Embryology
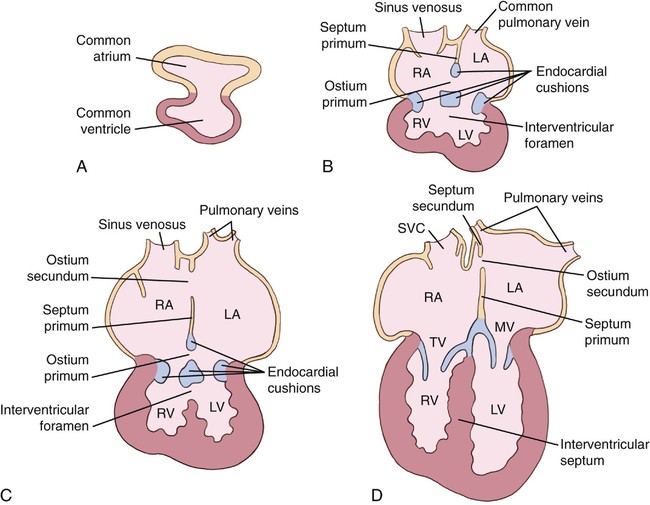
Normal embryogenesis of the AV canal consists of two processes: (1) separation of the common AV orifice into mitral and tricuspid valves and (2) closure of the atrial septum (ostium primum) and of the ventricular septum (secondary interventricular foramen).1 Development of the AV canal occurs by proliferation of the four endocardial cushions (superior, inferior, right lateral, and left lateral) and the dextrodorsal conus cushion. The superior and inferior cushions develop initially, followed by the right and left lateral cushions.
This is followed by a portion of the dextrodorsal conus cushion interposing between the superior and right lateral endocardial cushions. As the superior and inferior cushions grow toward each other, the common AV canal becomes two separate and distinct orifices: mitral and tricuspid (Fig. 6–3).
Next, the anterior mitral leaflet forms from the superior cushion and the left half of the inferior cushion. The posterior mitral leaflet arises from the left lateral endocardial cushion, and the anterior tricuspid leaflet arises from the right lateral endocardial cushion and the dextrodorsal conus cushion. Finally, the posterior tricuspid leaflet forms from the right lateral endocardial cushion, and the septal tricuspid leaflet forms from the inferior endocardial cushion.1–5
Occurrence Rate
AVSDs account for approximately 2.9% of all congenital heart defects and have an occurrence rate of 0.19 per 1000 live births.9,15,27–29 The incidence has been reported to be as high as 17% in fetuses with congenital heart disease.4,27,28 AVSDs are associated with a variety of syndromes and chromosomal abnormalities (Table 6–1). AVSD accounts for 40% of congenital heart malformations in individuals with Down syndrome.8,10 Complete AVSDs are also prevalent in patients with heterotaxic syndromes such as asplenia (right atrial isomerism) and polysplenia (left atrial isomerism).11,12,17,20,26 They occur slightly more often in females.8 The incidence of congenital heart defects in the offspring of mothers with AVSD is 14%, much higher than that of most congenital heart defects.24,29
TABLE 6–1
Conditions Associated with Atrioventricular Septal Defect
| Maternal | Fetal | ||
| Condition | Associated Cardiac Abnormalities | Chromosome Abnormalities | Syndromes |
| Diabetes |