CHAPTER 5
Ventricular Septal Defects
Definition
Most VSDs occur in isolation; however, 40% occur as part of one or more other structural cardiac abnormalities.1 Isolated VSDs are the most commonly recognized cardiac defect, accounting for 30% of cardiac defects in live-born infants and 9.7% in fetuses.2 They may vary in size and may be single or multiple. VSDs have the highest recurrence rate and are the most teratogen-associated defect.3
Embryology
The partitioning of the primitive embryonic heart into the chambers of the atria and ventricles begins at approximately 28 days of gestation. Initially, the IVS forms as a median muscular ridge in the floor of the ventricle near the apex. The early primitive physiological septal defect that occurs as the septum closes is called the interventricular foramen. As a concave, thick, crescentshaped fold, the IVS grows (primarily because of dilation of the ventricles on either side). Later, active proliferation of the myoblasts occurs and the thick muscular septum grows. The free edge of the primitive septum joins with the fused endocardial cushions at approximately 49 days of gestation. The interventricular foramen closes at about 56 days of gestation as a fusion of tissue from (1) the right bulbar ridge, (2) the left bulbar ridge, and (3) the endocardial cushions.4,5 The IVS is thickest at the apex, and it narrows to become thinnest at the level of the atrioventricular valves.
• A cessation of closure of the subaortic portion of the IVS because of maldevelopment of the following4:
Classifications
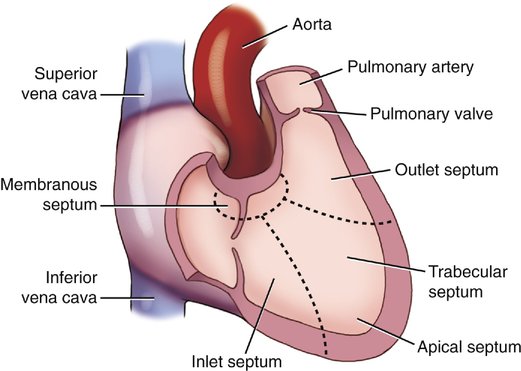
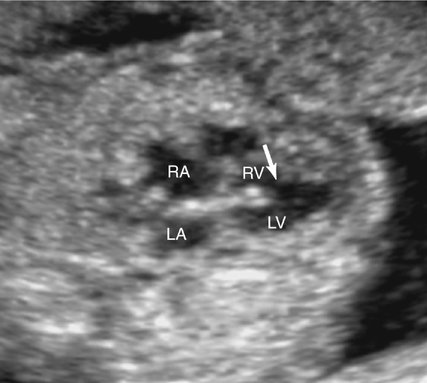
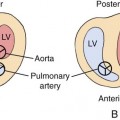
The IVS can be divided into two distinct regions: (1) the membranous septal region and (2) the muscular septal region. The muscular septal region can be subdivided into (1) the inlet, (2) the outlet, and (3) the trabecular regions (Fig. 5–1).6
According to these criteria, VSDs can be classified as membranous defects or as muscular defects.
Membranous Defects
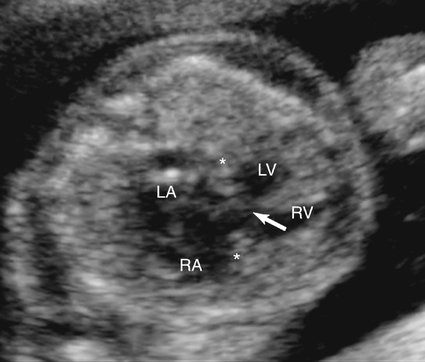
The membranous region of the septum is a small area close to the base of the heart that is bordered by the inlet and the outlet of the muscular septum and the commissure between the right and noncoronary cusps of the aortic valve. Because most defects in the membranous septum also involve a portion of the muscular septum, they are commonly described as perimembranous defects (Fig. 5–2).7,8 Perimembranous defects constitute 75% of all VSDs.8–11 Membranous VSDs are commonly associated with other structural cardiac abnormalities.12
Malalignment defects often involve the perimembranous septum. A malaligned perimembranous defect refers to a defect causing malalignment of the septum and great arteries (Fig. 5–3). When this occurs, both ventricles may empty into the overriding great artery. Conversely, the malalignment defect may obstruct the ventricular outflow, depending on the size of the defect and the extent of malalignment. An anterior shift of the septum may cause a right ventricular outflow obstruction, whereas a posterior shift could result in a left ventricular outflow obstruction.13 Malalignment defects can also occur in the muscular portion of the septum. In this setting the malalignment is between different portions of the septum (Fig. 5–4).
Muscular Defects
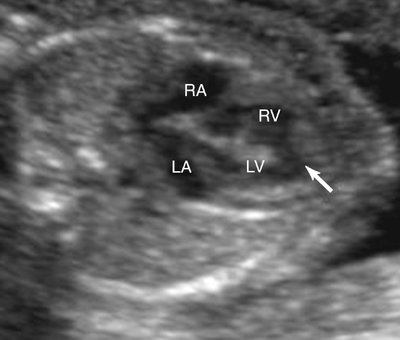
VSDs that are completely surrounded by muscular tissue make up 10% to 15% of VSDs (Fig. 5–5).11 These defects vary in size and are frequently multiple.13–15 Defects of the muscular septum are characterized by their location.16
Inlet Defects.
Inlet defects are found in the predominantly smooth-walled muscular septum that extends from the muscular attachments of the tricuspid valve leaflets to the distal attachments of the tricuspid valve apparatus.17 They are posterior and inferior to the membranous defects and account for 5% to 8% of VSDs.18 VSDs that occur as part of an atrioventricular septal defect (AVSD) are inlet defects (Fig. 5–6).13
Outlet Defects.
Outlet defects are anterior to the septal band of the right ventricle and occur in the most superior portion of the IVS, adjacent to the pulmonary and aortic valves. If the defect is found above the crista supraventricularis, it is described as a supracristal muscular VSD. Outlet defects are also referred to as subaortic, subpulmonary, or doubly committed subarterial defects.11,13 Five percent to 7% of VSDs are the outlet type, except in the Asian population, where the incidence is much higher, approximating 30%.8,19
Trabecular Defects.
Trabecular defects are posterior to the trabecula septomarginalis (septal band of the crista) and the mid portion of the septum. Marginal or anterior defects are usually small, multiple, and tortuous, occurring near the septal free wall margins; they may be distributed all along the margin.14 Trabecular defects are also termed midmuscular or central defects.
Apical Defects.
Apical defects are often included with trabecular defects. They occur close to the apex of the heart, distal to the insertion of the right moderator band. They are difficult to visualize with echocardiography even when large (Fig. 5–7).14
The term “Swiss cheese defect” has been used to describe the VSD that presents with multiple interventricular communications between the ventricles. These are typically apical in location. Nine percent of patients with VSDs have two or more defects.17
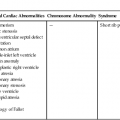
The frequency of occurrence of these types of muscular defects in the neonate is shown in Table 5–1.17 Central trabecular defects are identified most frequently (44%), and apical defects are seen least often (5%). On the basis of these published data, anterior and central defects tend to be identified in the immediate postpartum period, with a mean age of diagnosis of 1.3 and 2.2 weeks, respectively. Conversely, inlet and apical defects tend to be identified later (14 and 18 months, respectively).
TABLE 5–1
Frequency of Muscular Ventricular Septal Defects in Neonates
| Position | Frequency of All Muscular Ventricular Septal Defects (%) |
| Apical | 25 |
| Anterior | 26 |
| Midmuscular | 44 |
| Posterior | 5 |
From Ramaciotti C, Vetter JV, Bornemeier RA, et al: Prevalence, relation to spontaneous closure and association of muscular ventricular septal defects with other cardiac defects. Am J Cardiol 1995; 75:61–65.
Occurrence Rate
VSDs occur in 1% to 2% of live-born infants.20–23 VSD is the most common congenital heart defect in children, accounting for 20% to 57% of cases.21,24–29 The VSD has an incidence that is 20 times more common than the next most frequently encountered congenital heart defects.21 VSDs are slightly more common in female patients than in male patients (56% versus 44%).30
The risk of congenital heart disease in a fetus with a VSD-affected sibling is approximately 3%. If two previous siblings are affected, this risk increases to 10%. If the mother of the fetus is the affected relative, the risk of congenital heart disease is 6% to 10%. If the affected relative is the father, the risk is approximately 2%.22
The occurrence rate of VSDs has been observed to decrease with prenatal and postnatal age. This is largely accounted for by spontaneous or elective termination of complicated pregnancies and spontaneous closure of VSDs with advancing gestational age.23–26
Spontaneous Closure of a Ventricular Septal Defect
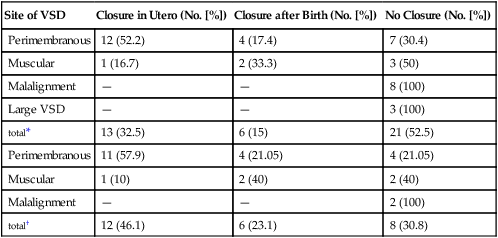
In 74% of pregnancies in which an isolated fetal VSD was confirmed by two independent observers, the VSD resolved spontaneously before birth.21,27 Of those that do not close prenatally, 75% to 90% will close within the first year of life.25,26,28 The size and location of the defect influence the rate of spontaneous closure.17 In general, large or malalignment defects tend to remain patent, whereas smaller perimembranous defects have a greater tendency to close (Table 5–2).*
TABLE 5–2
Closure of VSD by Location of Defect
| Site of VSD | Closure in Utero (No. [%]) | Closure after Birth (No. [%]) | No Closure (No. [%]) |
| Perimembranous | 12 (52.2) | 4 (17.4) | 7 (30.4) |
| Muscular | 1 (16.7) | 2 (33.3) | 3 (50) |
| Malalignment | — | — | 8 (100) |
| Large VSD | — | — | 3 (100) |
| TOTAL* | 13 (32.5) | 6 (15) | 21 (52.5) |
| Perimembranous | 11 (57.9) | 4 (21.05) | 4 (21.05) |
| Muscular | 1 (10) | 2 (40) | 2 (40) |
| Malalignment | — | — | 2 (100) |
| TOTAL† | 12 (46.1) | 6 (23.1) | 8 (30.8) |
*Included all continuing pregnancies (40 cases).
†Included only neonates who reached 1 year of age (26 cases).
From Paladini D, Palmieri S, Lamberti A, et al: Characterization and natural history of ventricular septal defects in the fetus. Ultrasound Obstet Gynecol 2000; 16:118–122.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree