Cardiovascular magnetic resonance (CMR) perfusion imaging is a robust noninvasive technique to evaluate ischemia in patients with coronary artery disease (CAD). Although qualitative and semiquantitative methods have shown that CMR has high accuracy in diagnosing flow-obstructing lesions in CAD, quantitative ischemic burden is an important variable used in clinical practice for treatment decisions. Quantitative CMR perfusion techniques have evolved significantly, with accuracy comparable with both PET and microsphere evaluation. Routine clinical use of these quantitative techniques has been facilitated by the introduction of automated methods that accelerate the work flow and rapidly generate pixel-based myocardial blood flow maps.
Key points
- •
Cardiovascular magnetic resonance (CMR) perfusion can assess ischemia with high accuracy and has been assessed against different modalities in well-designed randomized clinical trials.
- •
Noninvasive quantification of ischemia has a potential clinical impact in the management of patients with coronary artery disease beyond qualitative evaluation.
- •
Quantitative CMR perfusion techniques have significantly developed over the years and have shown robust accuracy compared with PET studies or invasive measurements of coronary flow.
- •
Automated quantitative CMR perfusion allows rapid and accurate creation of pixel-based myocardial blood flow (MBF) maps with in-line processing and improvement in clinical workflow.
- •
The use of automated CMR MBF maps in clinical routine may allow more accurate diagnosis of coronary artery disease as well as evaluation of the different phenotypic expression of atherosclerosis in both epicardial arteries and microvascular vessels.
Introduction
Coronary heart disease is the most common cause of morbidity and mortality globally. It is caused by the atherosclerotic narrowing of the coronary arteries and is amenable to treatment with medical therapy and revascularization. However, the suitability of a lesion for intervention depends on its functional significance. Coronary stenoses of hemodynamic significance are amenable to percutaneous intervention but intervening on those that are not flow limiting may confer a worse prognosis. Furthermore, the amount of ischemia is important. Subgroup analysis of the COURAGE (Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation) trial has shown that patients with greater than 10% left ventricle (LV) ischemia benefit from revascularization rather than medical therapy alone. The gold standard assessment for suspected coronary artery disease (CAD) is coronary angiography, but this is invasive and therefore associated with risks, and exposes patients to ionizing radiation. Therefore, high-quality assessment of ischemia and the functional significance of CAD is required to appropriately manage patients.
There are various noninvasive techniques to assess ischemia, including stress echocardiography, single-photon emission computed tomography (SPECT), PET, computed tomography with fractional flow reserve (FFR), and cardiovascular magnetic resonance (CMR). There are advantages to each and they have high sensitivities and specificities for the detection of coronary heart disease. CMR does not use ionizing radiation and is the gold standard for cardiac structure, function, and tissue characterization, giving extra useful information to clinicians.
In clinical practice, perfusion CMR is a qualitative technique. Typically, 3 LV short-axis images (base, mid, and apex) are acquired per heartbeat during the first pass of a gadolinium (Gd)-based contrast agent under conditions of vasodilator stress and rest. Experienced observers compare the images and look for areas of relative hypoperfusion at stress, which correspond with functionally significant coronary stenosis. Clinical decisions are then made based on this qualitative assessment. However, truly balanced ischemia caused by disease in all 3 of the coronary artery arteries could cause a global flow reduction that could be missed. Coronary microvascular dysfunction, a cause of chest pain and a common feature in cardiomyopathy, is not well characterized, and visual quantification may have reduced accuracy and reproducibility. Therefore, it is desirable to fully quantify myocardial perfusion and bring this into clinical practice.
Advanced imaging techniques can now quantify myocardial perfusion, the myocardial blood flow (MBF; mL/g/min) at stress and at rest. The myocardial perfusion reserve (MPR; referred to as the coronary flow reserve, CFR, in PET studies) is the ratio of the stress MBF to rest MBF. Relative flow reserve can also be measured by dividing the stress MBF of different myocardial segments. Most of the evidence for quantitative perfusion to date is from the PET literature but quantifying perfusion is also possible with CMR and recent advances have brought this to practice.
This article discusses the benefits of the noninvasive quantification of perfusion, discusses the methods of quantifying perfusion with CMR, and suggests how through automating the process it is possible to introduce quantitative CMR into clinical practice.
The importance of quantitative perfusion in clinical practice
CMR quantitative perfusion has historically been time consuming and difficult, which has kept it out of the realms of clinical practice and meant that most of the evidence for quantitative perfusion is in PET studies. Early evidence that MBF could be measured noninvasively came from the initial studies from Gould and colleagues showing the value of quantification to detect significant coronary stenosis. From those initial studies, quantification evolved to show its capability to characterize different levels of CAD severity in a more accurate way than qualitative analysis, especially identifying single-vessel versus 3-vessel disease and microcirculatory involvement. Patients with 3-vessel CAD had more extensive perfusion abnormalities on fully quantitative assessment than patients with single-vessel disease. Similarly, in a small CMR study of 41 patients with suspected CAD, fully quantitative perfusion was able to reliably discriminate between single-vessel and triple-vessel disease, which was not possible with qualitative perfusion. In one PET study, which enrolled 104 patients at moderate risk of CAD, absolute quantification had a significantly higher positive predictive value, negative predictive value, and accuracy for the detection of obstructive disease compared with qualitative perfusion. Furthermore, the interobserver variability of perfusion assessment was lower for the quantitative method. All these findings have important clinical implications where the extent of ischemia influences treatment choices.
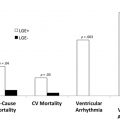
Quantitative perfusion also gives additional information compared with qualitative analysis. Absolute stress perfusion and CFR are prognostic. In one study, 256 patients, including 150 with known CAD, underwent ammonia PET evaluation and were followed up for 5.5 years. Those with impaired CFR (<2) had higher rates of death and major adverse cardiovascular events than those with normal CFR (>2). CFR was additive to risk determined by qualitative clinical read. Those with perfusion defects and abnormal CFR had worse clinical outcomes than those with normal CFR.
In patients with impaired LV function, myocardial perfusion is also important. Neglia and colleagues enrolled 67 patients with LV impairment with dipyridamole stress PET and followed them up for 45 months. Patients with severely depressed stress (≤1.36 mL/g/min) and rest perfusion (≤0.65 mL/g/min) had a relative risk of death or the progression of heart failure of 3.5 and 3.3 respectively compared with those with normal perfusion. On multivariate regression analysis, only stress MBF, resting heart rate, and end-diastolic dimensions were independently prognostic: 5-year event-free survival was 35.8% in patients with stress MBF less than or equal to 1.36 mL/g/min compared with 79% in those with MBF greater than 1.36 mL/g/min.
Perfusion may also be impaired in nonischemic cardiomyopathies. Patients with hypertrophic cardiomyopathy (HCM) often present with chest pain and have ischemic features on electrocardiogram thought to be related to microvascular dysfunction. Ischemic damage (both acute/subacute and chronic) is seen at autopsy and ischemia detected by SPECT is associated with ventricular tachycardia. Patient with HCM have impaired stress perfusion compared with healthy controls (even in nonhypertrophied segments) and this is associated with increasing wall thickness and fibrosis. In a prospective cohort study of 51 patients and 12 controls with atypical chest pain, the degree of perfusion impairment was an independent predictor of death and adverse cardiovascular events. Other cardiomyopathies with hypertrophy, such as Fabry disease and amyloidosis, also have impaired perfusion. In Fabry, this has been used to evaluate treatment efficiency. In amyloidosis, microvascular dysfunction has been shown using PET even in the absence of epicardial CAD and with lower stress and rest absolute perfusion values compared with patients with hypertensive LV hypertrophy. Using CMR, semiquantitative perfusion has been shown to differentiate amyloidosis from normal patients and to identify patients with normal and lower LV function.
In summary, fast, efficient quantitative perfusion for clinical practice and research would have advantages for disease identification and characterization, adding prognostic information and increasing reliability and adding the ability to characterize microvascular disease in CAD and cardiomyopathies, potentially aiding therapeutic drug development and treatment monitoring.
Qualitative and semiquantitative perfusion cardiovascular magnetic resonance
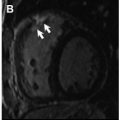
The baseline technique, qualitative stress perfusion CMR, is sensitive and specific for CAD detection and a normal CMR scan confers a good prognosis. Using the American Heart Association 17 (or 16) segment model, ischemia extent can be evaluated and used to target revascularisation with either 10% or 1.5 ischemic segments defining patients with a worse prognosis. To improve on this, semiquantitative assessment has been used. This method uses time–signal intensity curves in each myocardial segment to estimate the myocardial perfusion ( Fig. 1 ). There are various different methods that may be used for estimating perfusion, including contrast enhancement ratio (CER), myocardial to LV upslope index ratio, and upslope integral ratio. The CER is calculated from (SI peak – SI baseline )/SI baseline where SI peak is the maximum signal intensity (SI) in the region of interest and SI baseline is the mean baseline SI. The myocardial to LV upslope method is calculated by dividing the initial upslope of the myocardial time-SI curve by the initial upslope of the LV blood pool myocardial time-SI curve. The upslope integral ratio is the area under the curve (AUC) for the myocardial time-SI curve once the baseline has been adjusted for. The diagnostic accuracy of semiquantitative perfusion has been compared, using PET, with absolute MBF using animal models and microspheres and invasive coronary angiography. Compared with absolute MBF as measured using animal models and microspheres, at low flows there is a linear relationship between semiquantitative perfusion and MBF. However, as the absolute flow increases (hyperemic flow), the semiquantitative methods all significantly underestimate flow. The CER and the LV to myocardial upslope method begin to underestimate absolute MBF from 1 mL/g/min. Of the 3, the most linearly associated method is the upslope integral ratio, but even with this method the linearity decreased with flows greater than 3 mL/g/min.

Schwitter and colleagues found that semiquantitative perfusion had a sensitivity, specificity, and AUC of 91%, 94%, and 93% respectively for the detection of CAD with PET as the gold standard but lower compared with quantitative angiography (diameter stenosis >50%): 87%, 85%, and 91% respectively. Not all studies are so positive; for example, Mordini and colleagues compared each of the semiquantitative methods with quantitative coronary angiography, finding CER (57%, 91%, and 78%), the LV to myocardial upslope method (87%, 68%, and 82%), and the upslope integral ratio (83%, 68%, and 75%).
Overall the nonlinear relationship between semiquantitative perfusion and absolute MBF with the underestimation of hyperemic flow make semiquantitative assessment of perfusion only modestly incremental for accuracy compared with qualitative approaches for routine clinical practice, a benefit that is at best marginal given the associated time penalty of the analysis.
Quantitative cardiovascular magnetic resonance perfusion
Standardized full quantification is desirable for more accurate measurements of CMR perfusion but has been hard. The steps involved can be described as follows:
- 1.
Precise measurement of the arterial input function (AIF)
- 2.
Precise measurement of myocardial enhancement
- 3.
Sufficient temporal-spatial resolution to detect disease
- 4.
The ability to convert the above signals into contrast concentrations [Gd]
- 5.
A model of blood myocardium contrast behavior
- 6.
The computing power to solve the model to derive MBF
- 7.
The ability to do the above with sufficient accuracy, low time penalty, and in a generalizable way to be useful for clinical care
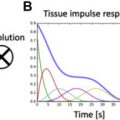
To perform these steps requires further capabilities. To convert MR signal to Gd concentration requires deep magnetic resonance (MR) imaging sequence knowledge (eg, gradient performance, understanding of prepulse limitations, coil performance, contrast nonlinearity, and signal clipping), the ability to image fast (every heartbeat, pixels across the myocardium, number of slices), the ability to motion correct images (at the varying contrast concentrations present), and the ability to segment the blood pool and therefore the myocardium. For clinical utility, this needs to be automated, but in a way that permits quality control overview by the reporting physician (ie, the display of quality control outputs) and display in a standardized format for clinicians. During first pass, Gd is very concentrated in the blood pool, resulting in T1, T2, and T2* effects not present when diluted during passage into the myocardium. A single measurement (readout) technique cannot be optimized for both. Two approaches are used: a dual-bolus approach, involving stress and rest perfusion done twice, initially with a low dose (eg, 10 × lower) of Gd for blood AIF, repeated at normal dose for myocardium; or a dual-sequence approach, involving full coverage optimized for myocardium, 1 slice repeated optimized to measure blood with its high Gd concentrations (this can be low resolution). There are a variety of different models of blood myocardial contrast exchange and a variety of different ways to solve these. Increasing model sophistication requires increasing computational power but supplies more potential accuracy. This domain is not yet standardized and a variety of approaches are available (a more comprehensive review on the models and approaches used for the quantification methods can be found in Katia Menacho and colleagues’ article, “ T2* Mapping Techniques: Iron Overload Assessment and Other Potential Clinical Applications ,” in this issue).
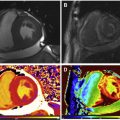
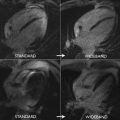
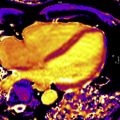
To assess the performance of such models requires both animal and human experimentation with increasingly robust gold standards ranging from microsphere experiments (animal models), comparator noninvasive testing (PET), and invasive testing based on coronary angiography, which needs to either quantitate luminal narrowing (three-dimensional quantitative coronary angiography, or via intracoronary wires with intravascular ultrasonography or optical coherence tomography) or measure intracoronary hemodynamics (FFR or instantaneous wave-free ratio). A summary of quantitative CMR studies using different approaches is listed in Table 1 . The first studies in 1993 and followed up in 1998 compared MR imaging measurements with microspheres in a dog model. These studies used a dual-bolus technique and compared with microsphere data with good correlation within a range of flow up to 5.0 mL/min/g both at rest and under pharmacologic stress. The first human studies compared quantitative perfusion with functional assessment of stenosis using invasive FFR as the gold standard and showed good sensitivity of 92.9% but low specificity of 56.7% using an MPR cutoff of 2.04. Using a high-resolution sequence at 3.0 T, there was an improvement in the accuracy of quantitative MPR versus FFR using a cutoff of 1.58 with a sensitivity of 0.80 and an improved specificity of 0.89. An example of a normal quantitative stress CMR perfusion examination is shown in Fig. 2 . Other investigators have also shown that quantitative perfusion may outperform qualitative and semiquantitative approaches with different techniques, either comparing the results with quantitative coronary angiography or invasive FFR as the gold standard. Compared with PET studies, CMR showed similar accuracy for the detection of significant coronary lesions but the absolute myocardial flow values were only weakly correlated, with mean CMR MBF values slightly different than the ones obtained with PET both for stenotic and nonstenotic territories with the methods used. Given the higher spatial resolution provided by CMR, analysis of differences in perfusion between the endocardial and epicardial layers can now be assessed more accurately and quantified, as shown in Fig. 3 in a patient with a severe left anterior descending artery proximal lesion. This assessment of transmural perfusion gradients quantitatively may become one of the unique applications made possible by CMR because it depends on high-resolution maps for correct analysis.
| Study | Field Strength (T) | AIF | Calculation Model | Automation | Software Used | Validation |
|---|---|---|---|---|---|---|
| Costa et al, 2007 | 1.5 | Single bolus | Fermi deconvolution | No | In-house development | FFR |
| Lockie et al, 2011 | 3.0 | Single bolus | Fermi deconvolution | No | In-house development | FFR |
| Hsu et al, 2012 | 1.5 | Single bolus, dual sequence | Model-constrained deconvolution | Semiautomated; pixel-wise quantification | In-house development | Microspheres (dogs); Visual invasive coronary angiography (human studies) |
| Huber et al, 2012 | 1.5 | Single bolus | Model-independent deconvolution | No | In-house development | QCA + FFR |
| Morton et al, 2012 | 1.5 | Dual bolus | Fermi deconvolution | No | ViewForum Software | PET |
| Miller et al, 2014 | 1.5 | Single bolus | Fermi, truncated singular valued, Tikhonov regularization | No | In-house development | PET |
| Mordini et al, 2014 | 1.5 | Dual bolus | Fermi deconvolution | No | In-house development | QCA |
| Motwani et al, 2014 | 3.0 | Single bolus | Fermi deconvolution | No | In-house development | QCA |
| Papanastasiou et al, 2016 | 3.0 | Single bolus | Fermi and 1-barrier, 2-region distributed parameter | No | In-house development | FFR |
| Chung et al, 2016 | 3.0 | Single bolus | Flexible tissue homogeneity and adiabatic tissue homogeneity | No | In-house development | Visual invasive coronary angiography |
| Kellman et al, 2017 | 1.5 | Single bolus, dual sequence | Fermi + blood tissue exchange | Yes | Gadgetron framework | Phantom, PET |
| Qayyum et al, 2017 | 1.5 | Single bolus | Tikhonov regularization | No | In-house development | PET |
| Hsu et al, 2018 | 1.5 | Single bolus, dual sequence | Model-constrained deconvolution | — | In-house development | QCA and CTA |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree