Bone Tumors and Related Diseases
FIBROUS, HISTIOCYTIC, AND FIBROHISTIOCYTIC ORIGIN
MISCELLANEOUS OR UNKNOWN ORIGIN
Bone tumors are one of the most serious diagnostic differentials in patients with musculoskeletal complaints. Bone tumors are categorized as either primary or secondary. Primary bone tumors arise from bone and related soft tissues directly in their site of involvement and may be either benign or malignant. Secondary bone tumors arise “secondary” to a primary lesion and also may be benign (e.g., secondary aneurysmal bone cyst arising in an area of past trauma) or malignant (e.g., bone metastasis from a lung carcinoma).4
Benign tumors usually are designated as such by the suffix -oma (e.g., enchondroma) and generally are not regarded as cancers. Malignant primary tumors of bone and other connective tissues are designated by the use of the term, or suffix, sarcoma after the tissue type involved (e.g., osteosarcoma).
Metastatic bone disease refers to a malignant tumor that secondarily seeds to bone, usually from a primary malignancy of the epithelial tissue (designated as carcinomas) of the lung, breast, prostate, kidney, liver, and so on. For instance, if the patient has a bronchogenic carcinoma that metastasizes to the thoracic spine, the lung lesion is the primary lesion and the thoracic spine lesion is bone metastasis of the lung lesion.
In general terms, both benign tumors of bone and bone metastasis are many times more common than primary malignancies of bone. Primary malignancy of bone is relatively uncommon. Malignancy in general is thought to affect approximately 1 in 3 people over their lifetime. Approximately 1.4 million Americans are diagnosed with a malignancy each year, with less than 2800 of these cases representing primary malignancies of the bone and joints and another 16,500 cases identified as myeloma.180a By comparison, there are approximately 263,000 new cases of malignancy annually of the digestive system, 215,000 cases of primarly malignancy of the breast, and 186,000 cases of primary malignancy of the respiratory system diagnosed in the United States.180a The incidence of primary malignant bone lesions (excluding multiple myeloma) is estimated at 8 per million persons.61 The most common sarcomas include osteosarcoma (35.1%), chondrosarcoma (25.8%), Ewing sarcoma (16.0%), chordoma (8.4%), and fibrosarcoma (5.7%). Because it arises from the plasma cell of bone marrow, multiple myeloma is not considered a primary malignancy of bone. Instead, osteosarcoma is the most common primary malignancy of bone. However, multiple myeloma is by far the most common primary malignancy of bone if considered in the group of primary bone malignancies135.
The evaluation of a bone tumor requires careful assessment of the patient’s history and application of clinical studies in developing a concise list of differential possibilities. Diagnostic imaging plays a major role in developing and narrowing this list. With advances in technology, biologic tissue assessments have added a valuable tool to the arsenal of the investigator. Therefore, the diagnosis of tumors is accomplished along three dimensions of assessment: clinical (e.g., gender, age, symptoms), imaging (e.g., location, appearance), and pathologic (e.g., microscopic cell type and molecular assessment).
Imaging Modalities
Imaging studies should define the lesion, determine its location, grade its aggressiveness, decide if the lesion is limited to one bone (monostotic) or if multiple bones are involved (polyostotic), assess soft-tissue involvement, and identify the lesion’s matrix. Plain film radiography remains the chief imaging modality for the initial assessment of bone tumors. Sometimes the radiographic presentation reveals a lesion that is nonaggressive with classic characteristics, thus requiring no further examination and leading to an immediate diagnosis. However, further assessment often is necessary when lesions are poorly defined or accompanied by significant clinical signs and symptoms.
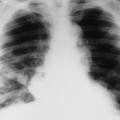
Computed tomography (CT) provides direct thin axial slices of anatomy, allowing for a more detailed assessment of complex anatomy (e.g., spine) than can be accomplished with plain film. CT demonstrates calcification well and therefore is capable of demonstrating calcification within the lesion’s matrix and the cortical response of the lesion’s host bone. Plain film radiographs and CT scans of the chest help to assess the possibility of pulmonary metastasis, the presence of which may alter the treatment plan.
Although radionuclide bone scanning lack specificity, it is sensitive to the presence of early disease and is widely used to assess the possibility of multiple lesions, a finding that substantially narrows down the diagnostic list of possibilities. There is one notable exception to the sensitivity of bone scans. Bone scans have elevated rates of false negative findings in the presence of multiple myeloma and purely lytic lesions. In this and other cases it may be best to apply radiographic surveys or multiregional magnetic resonance imaging (MRI) studies.
MRI has the ability to demonstrate abnormality of the bone marrow and delineate extraosseous involvement. Replacement of normal marrow by pathologic processes (e.g., metastasis, multiple myeloma, osteomyelitis) is readily demonstrated and provides an early sign of disease. However, MRI remains inferior to both plain film and CT for detailing calcification, ossification, cortical destruction, and periosteal reaction. MRI is especially valuable for assessing the neurologic impact of the lesion. Plain film radiographs efficiently provide information about the rate of growth and aggressiveness of a lesion, but do not establish a histologic diagnosis with the same accuracy as a biopsy. On MRI studies, most bone tumors are dark on T1-weighted images and bright on T2-weighted images. Fibrous tissue, cortical bone, desmoids, and scar tissue are dark on both T1- and T2-weighted images. Hemangiomas, lipomas, and liposarcomas are bright on both T1- and T2-weighted images because of the blood components of these lesions.
The information offered through imaging is coupled with clinical data, laboratory studies, and possibly biopsy to identify the specific lesion present.
Laboratory Tests
Laboratory tests are less helpful than imaging studies for diagnosing bone tumors. Benign bone tumors demonstrate normal laboratory values, and malignant tumors often demonstrate normal laboratory values. However, a few characteristic laboratory findings may be seen in malignancy. For example, increased serum calcium levels and increased hydroxyproline in the urine are associated with massive bone osteolysis, as seen in generalized lytic metastasis or multiple myeloma. Some osteosarcoma, osteoblastic metastasis, and other bone-proliferating malignancies often are accompanied by increased serum alkaline phosphatase levels. Multiple myeloma is associated with monoclonal immunoglobulins “M-spike” on serum electrophoresis, Bence Jones proteins in the urine, hyperglobulinemia (reversed albumin-to-globulin [A : G] serum ratio), elevated creatine, and decreased hematocrit levels.
Bone Biopsy
Bone biopsy is the removal of suspect tissue from the body for examination by a pathologist. In most circumstances a biopsy provides the most accurate diagnosis possible, typically more accurate than can be obtained by imaging. However, biopsy is not especially helpful for determining the lesion’s rate of growth and aggressiveness. The latter qualities are best assessed by conventional and specialized imaging modalities; therefore, a biopsy and imaging studies are complementary, leading to an end diagnosis.
The accuracy of the biopsy results depends on which region of the lesion is collected, the skill of the person performing the biopsy, and the clinical circumstances in which the biopsy takes place. It is generally most accurate if the biopsy is taken from the most aggressive and viable portion of the lesion. This often requires an open biopsy approach. However, an open biopsy is associated with higher complication rates than a closed (percutaneous) biopsy approach.171 Open biopsy procedures may contaminate malignant cells in normal tissue, interfering with otherwise successful limb-sparing procedures. Closed biopsy procedures generally are safer, but may not produce enough tissue for accurate diagnosis. Biopsy of highly vascular lesions is cautioned generally, given the risk of massive hemorrhage.
Tumor Staging
Tumor staging is integral to patient management. Once a tumor is found, the extent of disease is defined along three parameters. The first parameter defines the size of the tumor (T) and whether it has invaded surrounding tissues. The second parameter examines the extent of lymph node involvement (N). Lastly, one must know whether the tumor has metastasized to other regions of the body (M). These three parameters define the “TNM” system of staging lesions. Although some variation in the stages exists depending on the source and type of tumor being staged, a general presentation of the TNM method is presented in Box 13-1.
Signs and Symptoms
Clinically important lesions are usually detected secondary to the patient’s complaint of pain or attention to a palpable mass. Aggressive bone lesions generate signs and symptoms directly, secondary to bone and tissue destruction. Classically the bone pain experienced is more dramatic at night and unrelated to physical activity. The patient may exhibit fever, impaired mobility, and cachexia with aggressive lesions. In contrast, most benign lesions are clinically asymptomatic, but commonly are recognized after pathologic fracture. This concept has been termed traumatic determinism; that is, the presence of the benign lesion is only determined as a result of trauma and subsequent fracture. Less frequently, asymptomatic bone lesions are detected serendipitously on radiographs obtained for unrelated reasons.
Tumor Discriminators
Many tumors and tumor-like conditions produce similar imaging findings. The following list of radiologic and clinical parameters assists in narrowing the usually broad number of pathologic possibilities for a given radiographic appearance. In addition, the following parameters assist in evaluating the aggressiveness and clinical importance of a lesion.
Patient Age
The age of the patient is an important clue helping to differentiate between lesions that look the same but are unique to certain age groups. Conversely, the patient’s age may suggest a diagnostic possibility that would not otherwise be considered from the radiographic appearance because of an atypical presentation. Age is more helpful when the age range associated with the tumor is narrow. Given only the patient’s age, an examiner can determine which tumor is present with a high degree of accuracy.69 (Tables 13-1 and 13-2 present the ages at which common benign and malignant tumors develop, respectively.) Generally with malignant tumors, Ewing sarcoma and osteosarcoma are present in children, and metastatic bone disease and multiple myeloma are found in patients older than 40 years of age. Benign tumors are most common in patients 10 to 30 years of age, slightly younger for simple bone cysts, and slightly older for lipomas and hemangiomas.
TABLE 13-1
TYPICAL AGES FOR THE DEVELOPMENT OF SELECTED BENIGN TUMORS
| Age (in Years) | Tumor |
| 5 to 10 | Simple bone cyst |
| 10 to 20 | Chondroblastoma, nonossifying fibroma, osteoid osteoma |
| 10 to 30 | Aneurysmal bone cyst, chondromyxoid fibroma, osteoblastoma, osteochondroma |
| 15 to 35 | Enchondroma, osteoma |
| 20 to 40 | Giant cell tumor |
| 30 to 50 | Lipoma |
| 40 to 50 | Hemangioma |
TABLE 13-2
TYPICAL AGES FOR THE DEVELOPMENT OF SELECTED MALIGNANT TUMORS
| Age (in Years) | Tumor |
| Less than 1 | Metastatic neuroblastoma |
| 1 to 30 | Ewing sarcoma, osteosarcoma |
| 20 to 40 | Giant cell tumor, parosteal osteosarcoma |
| 30 to 50 | Fibrosarcoma, malignant fibrous histiocytoma, primary lymphoma of bone |
| More than 40 | Chordoma, chondrosarcoma, metastatic bone disease, multiple myeloma |
Location
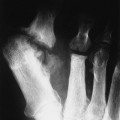
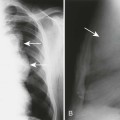
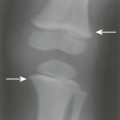
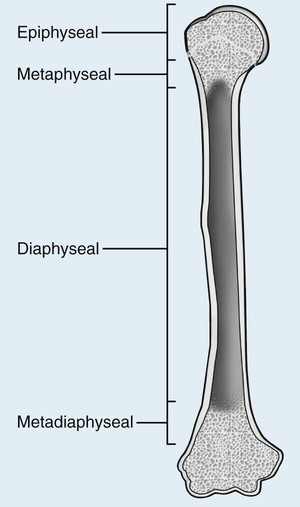
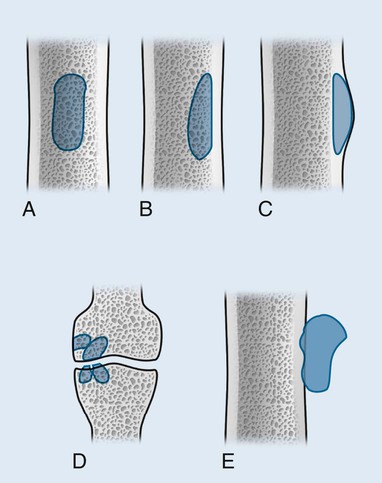
Individual tumors are often predisposed to occur in certain bones (Table 13-3). Even more suggestive is the longitudinal (diaphysis, metadiaphysis, metaphysis, and epiphysis) (Fig. 13-1) and transverse (central or eccentric medullary, cortical, periosteal, and parosteal) (Fig. 13-2) location within bone (Table 13-4 and Fig. 13-3).
TABLE 13-3
SUMMARY OF COMMON BENIGN AND MALIGNANT LESIONS
| Tumor | Frequency | Age (Median) at Diagnosis | Typical Location | Radiologic Features | Comments |
| Benign | |||||
| Aneurysmal bone cyst | Uncommon | 10 to 30 | Metaphysis of long bones; sometimes posterior arch of vertebrae | Widely expansile defect of the cortex with cortical buttressing at the region of expansion from the host bone and very thin cortices at outer edge of the lesion. The lesions are osteolytic and exhibit “a soap bubble” appearance | The lesion represents cavities filled with extravasated blood. Lesions may occur secondary to trauma, or concurrent to other tumors (e.g., giant cell tumor). Pain and swelling at the site of involvement |
| Bone island | Very common | Any age | Any location but skull | Homogenously radiodense focus with spiculated “brush” border | The lesion represents a hamartoma of bone. They are painless, usually small, and of no clinical significance except for their differentiation from blastic metastasis. Multiple bone islands are termed osteopoikilosis. Osteomas are similar lesions found in the skull |
| Chondroblastoma | Rare | 10 to 20 | Proximal femur, about the knee, and proximal humerus | Small (<5 cm) osteolytic subarticular lesion with a well-defined margin of sclerosis | A rare lesion composed of chondroblasts and immature cartilage. They appear in the secondary centers of enchondral ossification of skeletally immature patients |
| Chondromyxoid fibroma | Rare | 10 to 30 | Proximal tibia and fibula | Radiolucent, oval, eccentric lesion | Tumor involving the cartilage-forming connective tissue of marrow space |
| Enchondroma | Common | 15 to 35 | Small tubular bones of the hands and feet; however, possible in any enchondrally formed bone | Small round or oval cystic defects, typically with stippled matrix calcification and endosteal scalloping | Discrete islands of cartilage surrounded by layered bone. Enchondromas occur within a bone; periosteal chondromas occur next to the cortex and under the periosteum. Long bone lesions are more likely to be painful. Multiple enchondromas is termed Ollier disease, and if soft-tissue hemangiomas are also presenting, it is termed Maffucci syndrome. Solitary enchondromas have a malignant potential of <1%; this rate rises to 30% for Ollier disease and higher for Maffucci syndrome |
| Fibrous cortical defect | Very common | 4 to 8 | Distal femur and proximal and distal tibia | Well-marginated, eccentric cortex-based osteolytic defect smaller than 3 cm | Nonaggressive fibrous lesion of bone, usually healing spontaneously |
| Fibrous dysplasia | Common | <20 | Femur, tibia, craniofacial bones, ribs, and pelvis | Wide variety in appearance depending on the bone involved. Long bones demonstrate a midly expansile medullary lesion that may be mildly sclerotic (“smoky” or “ground glass” appearance). “Shepherd’s crook” deformity is a lateral bowing and coxa vara deformity occurring in the proximal femur. Individual lesions often exhibit a thick “rind” of marginal sclerosis | Nonneoplastic disturbance of bone maintenance resulting in fibrous tissue replacing normal bone. Albright syndrome is the triad of polyostotic fibrous dysplasia, precocious puberty, and cutaneous hyperpigmentations (café-au-lait spots) |
| Giant cell tumor | Common (18% of benign bone tumors) | 20 to 40 | Distal radius, distal femur, proximal tibia | Subarticular, osteolytic, mildly expansile, eccentric, no marginal sclerosis, and sometimes with internal septation | Tumor of the supportive tissues of bone marrow; composed of large mononuclear stromal cells and interspersed osteoclastic-like multinucleated giant cells. Approximately 5% to 10% may be malignant, marked by pain, swelling, and aggressive radiographic appearance |
| Hemangioma | Common | 40 to 50 | Craniofacial bones and vertebral bodies | Osteolytic defect with characteristic coarsened, honeycomb trabeculae; or vertical striations (“corduroy cloth”) | Represent vascular lesion of bone; typically asymptomatic; rarely expanding vertebral lesion may relate to spinal stenosis |
| Nonossifying fibroma | Very common | 10 to 20 | Metaphysis of tibia and femur | Well-marginated, eccentric cortex-based osteolytic defect >3 cm. Malignant lesions show cortical breakthrough and soft-tissue mass | Nonaggressive fibrous lesion of bone, usually healing spontaneously |
| Osteoblastoma | Uncommon | 10 to 30 | Posterior elements of the vertebrae; less commonly in proximal femur and tibia | Geographic, osteolytic, eccentric, mildly expansile lesion | Same as osteoid osteoma; highly vascular connective tissue nidus with interlaced osteoid tissue. Lesions may be painful |
| Osteochondroma | Common | 10 to 30 | Metaphyses of long bones, mostly lower extremities. Hereditary multiple exostoses of the proximal femora and pelvis is common | Sessile or pedunculated (“coat hanger” exostosis or “cauliflower cap”) cartilage capped bony outgrowth that is continuous with underlying bone | Abnormal outgrowth of lateral portion of the growth plate; affects epiphyseal growth. Multiple osteochondromas are referred to as hereditary multiple exostoses (HME), and are associated with a higher rate of malignancy (up to 25%) compared with solitary lesions (1% to 2%) |
| Osteoid osteoma | 11% of benign bone tumors | 10 to 20 | Cortex of proximal femur or tibia; less commonly the posterior elements of the vertebrae | Small radiolucent nidus surrounded by radiodense reactive sclerosis | Highly vascular connective tissue nidus with interlaced osteoid tissue. Classically, pain at night that is alleviated by aspirin. Spine lesions present on the concave side of a painful scoliosis. Low recurrence following excision |
| Simple bone cyst | Common | 5 to 10 | Proximal humerus and proximal femur | Oblong, central, expansile, radiolucent subepiphyseal (less frequently diaphyseal) osteolytic lesion. At times the partial internal septations may fracture and inferiorly migrate (“fallen fragment” sign) | Fluid-filled lesion of uncertain etiology; clinically silent unless fractured |
| Malignant | |||||
| Bone metastasis | Very common | >40 | Spine, ribs, skull, and pelvis; rarely distal to the knees and elbows | Individual lesions appear osteolytic, less commonly osteoblastic, and characteristically with no or small soft-tissue mass or periosteal reaction | The most common mechanism is hematogenous seeding from primary tumors; the breast gives rise to 70% of female lesions, and prostate to 60% of male lesions. Advanced osteolytic destruction may exhibit increased serum calcium. Osteoblastic lesions may reveal increased levels of alkaline phosphatase |
| Multiple myeloma | Common | >40 (60) | Vertebrae, rib, innominate, femur | Osteopenia, “punch-out” lesions, “raindrop” skull | Malignant plasma cell proliferation; with abnormal laboratory findings of Bence Jones proteins, anemia, thrombocytopenia, Rouleaux formation, Mott cell |
| Osteosarcoma | 35.1* | Bimodal: first mode 1 to 30 (19); second mode in elderly | Metaphyses of long bones (distal femur, proximal tibia, and proximal humerus) | Osteolytic (25%), osteoblastic (50%), or mixed (25%) lesion with poorly defined margins, aggressive periosteal reaction (“onion skin” or Codman triangle), and soft-tissue mass | Aggressive lesion of mesenchymal bone-producing cells. Lesions are painful, rapidly expanding, with malignant potential usually requiring amputation |
| Chondrosarcoma | 25.8* | >40 (53) | Femur, innominate, humerus | Destructive lesions appearing as a central lesion in bone with stippled calcifications, and cortical scalloping, or as a peripheral lesion extending from the bone’s surface appearing as an exploded osteochondroma | Malignancy of mesenchymal cartilage-producing cells exhibiting varying degrees of malignancy. Lesions are painful and expanding with malignant potential. They may be primary malignant lesions of bone, or secondary to benign cartilage lesions (e.g., enchondroma, osteochondroma) |
| Ewing sarcoma | 16.0* | 1 to 30 (15) | Femur, innominate, vertebrae, humerus | Permeative osteolytic eccentric pattern of bone destruction, often presenting as a scalloped deformity of the diaphyseal cortex (“saucerization”); in flat bones it appears as a mildly expansile, soap-bubble, osteolytic defect | Small round cell malignancy of bone. The patient exhibits a tender, warm, swollen limb |
| Chordoma | 8.4* | 30 to 70 (59) C2 vertebral body | Skull, sacrum, and | Appear as midline lesions, characterized by the presence of bone destruction and a large soft-tissue mass; calcification is noted in up to 40% of lesions on plain films | Develops from remnant portions of the notochord; patients complain of pain and enlarging soft-tissue mass, and regional visceral and neurologic involvement, such as headaches and diplopia for sphenooccipital lesions, and bowel and bladder dysfunction for sacrococcygeal lesions. Radiation is applied to those lesions that cannot be resected |
| Fibrosarcoma and malignant fibrous histiocytoma | 5.7* | 30 to 50 (59) | Distal femur and proximal tibia | Lesions appear as osteolytic destruction in the metaphysis, often extending to the diaphysis marked by cortical destruction and soft-tissue mass | Malignancy of deep fibrous tissue; patient complaints of pain and tenderness in the region of involvement (usually around the knee) |
*This percentage reflects the portion of primary malignant tumors of bone, excluding multiple myeloma.
From Dorfman HD, Czerniak B: Bone tumors, St. Louis, 1998, Mosby; Dorfman HD, Czerniak B: Bone cancer, Cancer 75:2003, 1995.
TABLE 13-4
LOCATION OF SELECTED TUMORS WITHIN A BONE
| Location | Comments |
| Longitudinal | |
| Diaphysis | Round cell lesions (Ewing sarcoma, primary lymphoma of bone, multiple myeloma), malignant fibrous histiocytoma, adamantinoma |
| Diametaphysis | Osteoblastoma, chondromyxoid fibroma, nonossifying fibroma, Ewing sarcoma |
| Metaphysis | Aneurysmal bone cyst, chondrosarcoma, enchondroma, fibrosarcoma, giant cell tumor, osteosarcoma |
| Epiphysis | Chondroblastoma, giant cell tumor |
| Axial | |
| Central | Central chondrosarcoma, Ewing sarcoma, fibrous dysplasia, primary lymphoma of bone, solitary bone cyst |
| Eccentric | Aneurysmal bone cyst, fibrosarcoma, giant cell tumor, nonossifying fibroma |
| Cortical | Fibrous cortical defect |
| Parosteal | Parosteal chondrosarcoma, periosteal chondroma |

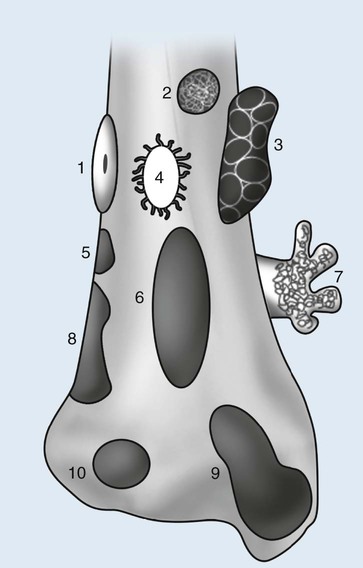
Soft-Tissue Involvement
A soft-tissue mass, created when the tumor mass breaks through the host bone’s cortex, suggests an aggressive tumor. Soft-tissue masses related to tumors distort but do not disrupt intramuscular soft-tissue planes; soft-tissue masses secondary to infections may disrupt intramuscular soft-tissue planes. Tumors tend to respect soft-tissue boundaries; infections may not.
Host Bone Reaction
A number of pathologic processes are capable of accentuating or reviving normal mechanisms of bone growth resulting in periosteal or endosteal reactions. The appearance of the periosteal and endosteal reactions relates to the aggressiveness, intensity, and duration of the inciting process.
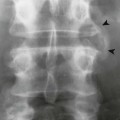
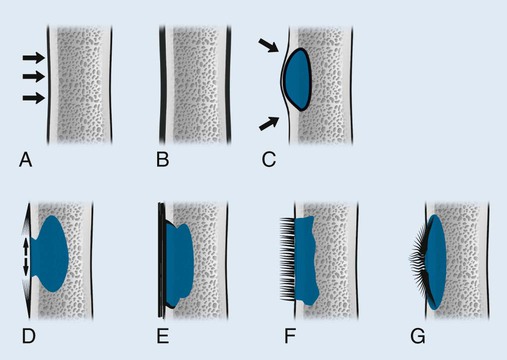
Periosteal and endosteal reactions must mineralize to be visible on radiographs; several patterns are identified (Fig. 13-4). Mineralization takes between 1 and 3 weeks. If the inciting process is indolent (e.g., vascular stasis), a thick, wavy periosteal reaction develops. Layered or lamellar periosteal reactions indicate a mildly aggressive underlying pathology (e.g., acute osteomyelitis). Aggressive pathology (e.g., osteosarcoma, Ewing tumor) may disrupt the periosteum, producing a radiating (sunburst) or parallel (hair-on-end) spiculated pattern (Fig. 13-5). The disrupted periosteum may form an acute angle with the cortex of the bone (Codman triangle) (see Figure 13-4). Endosteal reactions are more limited, appearing thickened or scalloped in response to pathology within the medullary canal. Table 13-5 shows radiologic tumor grades based on the reaction of the host bone.
TABLE 13-5
RADIOGRAPHIC CHARACTERISTICS OF TUMOR GRADES
| Aggressiveness | Characteristics |
| Low grade—nonaggressive | Geographic destruction surrounded by sclerotic rim of bone |
| Medium grade—moderately aggressive | Geographic destruction, short transition zone, possible sclerotic rim, possible bone expansion, possible thick periosteal reaction |
| High grade—highly aggressive | Permeative or moth-eaten destruction, wide transition zone, no surrounding sclerosis, possible bone expansion, aggressive periosteal reaction |
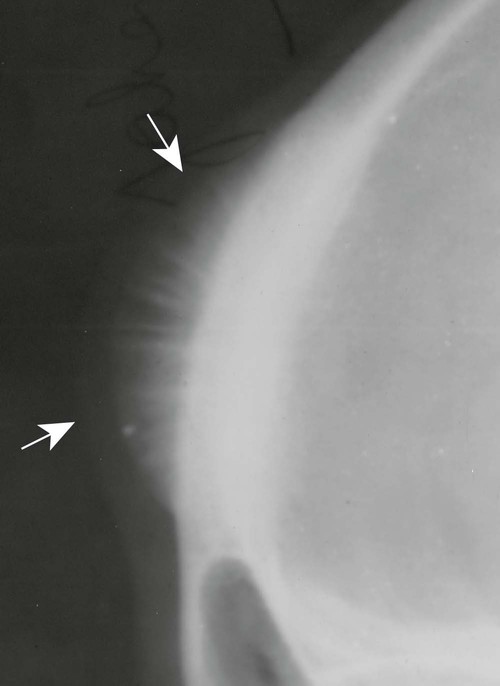
The tumor grade ultimately is derived from microscopic appearance of the tumor, as performed by a pathologist in association with the imaging findings and clinical data. Tumor staging (see Box 13-1) and grading relate inversely to the patient’s chance of survival.
Pattern of Bone Destruction
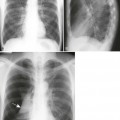
A number of explanations exist as to why tumors produce bone destruction. Tumors may stimulate osteoclastic activity by direct pressure, local hyperemia, or invasion. Bone destruction may be permeative, moth-eaten, or geographic (Fig. 13-6). Geographic lesions generally are less aggressive than the more subtle moth-eaten–pattern lesions, which generally are less aggressive than the nearly imperceptible permeative pattern.
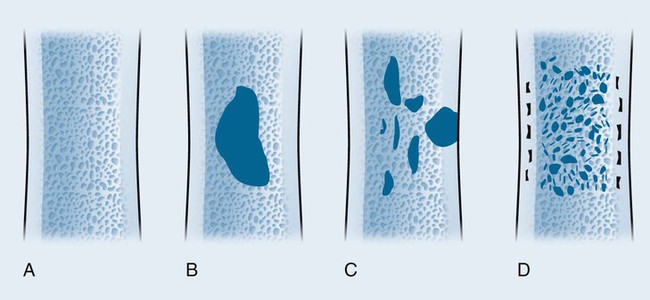
An observer’s ability to recognize bone destruction depends primarily on the size and location of the lesion. Destructive lesions in cortical bone are more easily recognized than those in cancellous bone because greater contrast exists between osteolytic lesions and compact bone than osteolytic lesions and cancellous bone. Although technical factors should be considered, in general, lesions in cancellous bone that are less than 1 cm in diameter are difficult to recognize. Moreover, 30% to 50% of cancellous bone may be destroyed before an osteolytic lesion is recognized on the most optimally exposed and processed radiographs.36,41,71,101 Osteolytic lesions in the diaphysis are more easily recognized because of the higher proportion of compact bone.
Size of the Lesion
Each tumor has a unique growth rate influenced in part by the nature of the lesion and the response of the host bone. Markedly expansile lesions result when endosteal bone reabsorption of the inner cortex occurs in concert with periosteal appositional, intramembranous new bone growth of the outer cortex (Fig. 13-7). Although it is generally true that larger lesions are more aggressive than smaller ones, one should be careful not to judge a tumor’s clinical importance by size alone.
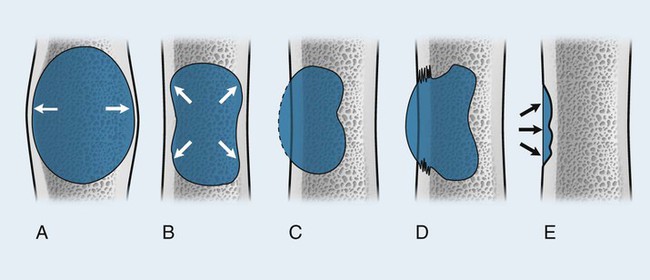
Rate of Growth
The rate of growth is an important characteristic for assessing the aggressiveness of a lesion. Benign lesions grow slowly and have thick margins. Malignant lesions grow quickly and have less-defined margins. Although highly important, the assessment of a lesion’s growth rate is difficult because of the usual lack of available serial studies.
Margination and Zone of Transition
The appearance of the zone of transition between a lesion and the host bone is probably the single best indicator of a lesion’s aggressiveness. An abrupt transition from normal to abnormal is a feature of benignancy. A wide transition is a feature of malignancy. The zone of transition is a direct reflection of the lesion’s aggressiveness and the response of the host bone to the lesion infiltration (Fig. 13-8). Margination describes the presence and thickness of the rim around the lesion. Malignant tumors typically are nonmarginated, a feature of their wide zone of transition (Figs. 13-8 and 13-9). Conversely, a lesion is most likely benign if surrounded by a sclerotic rim of varying thicknesses producing a narrow zone of transition. The presence of a thick margin is always accompanied by a short zone of transition and represents an attempt of the host bone to surround and limit a lesion’s growth. However, a short zone of transition is not always accompanied by a thick margin (e.g., giant cell tumor). The zone of transition should not be used as the sole criteria for determining the aggressiveness of the lesion. Even a well-marginated lesion can prove to be malignant in selected clinical circumstances. For example, a painful lesion presenting with a narrow zone of transition in a 60-year-old patient who has a previous history of a primary tumor should be considered bone metastasis until proved otherwise.
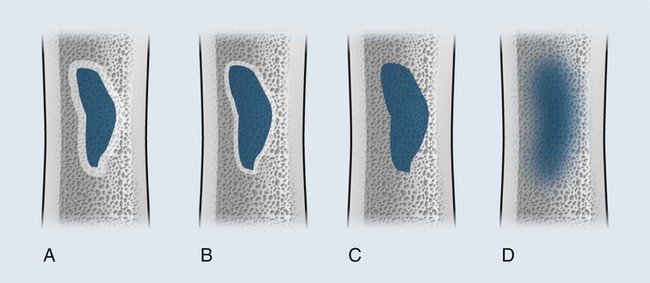
Tumor Matrix
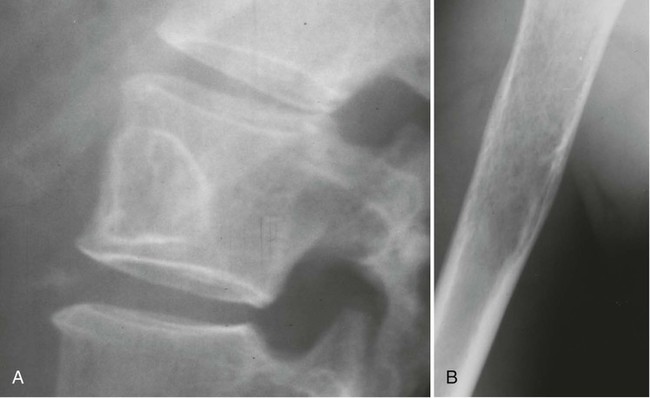
The matrix is the internal tissue, or substance, of a tumor. The radiographic appearance of the tumor’s matrix assists its categorization as primarily bone-, fibrous-, or cartilage-forming. Most bone tumors have a radiolucent matrix correlating to soft tissue. Only when the matrix is sufficiently mineralized with hydroxyapatite crystals will it become radiographically visible. Bone-producing tumors are radiodense. Highly aggressive bone-producing tumors appear less dense, with poorly formed osteoid material, than do nonaggressive bone-producing tumors. Tumors with a purely fibrous matrix appear radiolucent or slightly hazy (because of interspersed small bone fragments). Cartilage tumors usually are accompanied by irregular ring-like, flocculent, stippled, or fleck-like radiodensities within the matrix (Figs. 13-10 and 13-11).
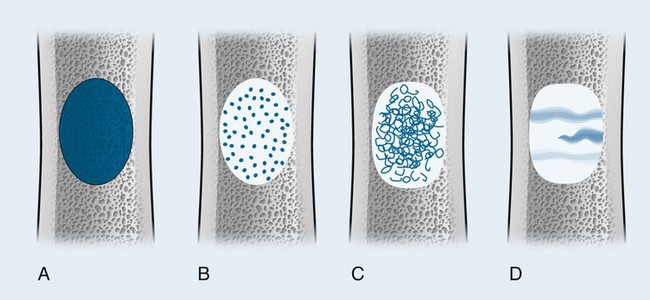
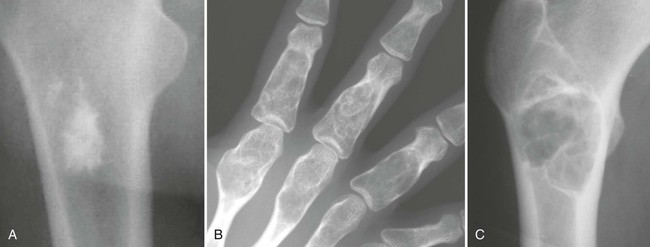
Multiplicity
The presence of multiple lesions suggests different diagnostic possibilities than does the presence of a single lesion. Generally, radionuclide bone scanning determines if multiple lesions exist. Because radionuclide imaging is sensitive but not specific, plain film radiographs are usually taken at regions of increased radionuclide uptake. Radiographs usually are directed at evaluating a lesion rather than finding it. MRI and CT are applied less commonly to resolve uncertain findings of the radionuclide bone scan and plain films. The presence of multiple lesions limits the differential diagnosis. Common aggressive polyostotic diseases include metastatic bone disease and multiple myeloma. Less-common possibilities include multicentric osteosarcoma and multifocal infections. Common nonaggressive polyostotic lesions include fibrous dysplasia, Paget disease, histocytosis, hereditary multiple exostoses (HME), multiple enchondromas (Ollier disease), and osteomyelitis.
Treatment Options
Treatment options include surgery, chemotherapy, radiation, or pain management; all may be applied alone or in combination. Surgical options extend from curettage for benign lesions to wide resection and possible amputation for highly malignant lesions. Each treatment approach is associated with complications, tumor recurrence, and varying success rates dependent on the patient’s clinical status and the type and stage of the lesions (Table 13-6). Often the treatment plan is aimed at offering the patient palliative relief alone.
TABLE 13-6
| Treatment | Description |
| Chemotherapy | Chemotherapy involves the intravenous or oral application of anticancer drugs Many agents are not selective to cancer cells, and may produce significant side effects (e.g., low blood count, vomiting, loss of appetite, loss of hair, mouth sores) |
| Complementary and alternative therapies | Complementary or alternative management strategies may be applied to reduce malignant lesions or offer palliative/symptom relief |
| Hormone therapy | The growth of some forms of cancer is enhanced by circulating hormones. For instance, estrogen promotes the growth of some breast cancers and testosterone promotes the growth of most prostate cancers. In an attempt to limit the growth of cancers, surgical removal of the ovaries or testes may be considered. More often drugs can be administered to limit the production of tumor-enhancing hormones or limit their effect on the tumor cells (e.g., tamoxifen) |
| Immunotherapy | Immunotherapy describes efforts to facilitate the patient’s immune system to recognize and destroy cancer cells by focusing on cytokines, monoclonal antibodies, and various tumor vaccines |
| Radiation therapy |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree