Central Nervous System
Hossein Jadvar
Patrick M. Colletti
LEARNING OBJECTIVES
1. Describe the major clinical applications of scintigraphy in imaging of the central nervous system.
2. Name the relevant radiotracers and general procedures that are employed in scintigraphic imaging evaluation of cerebrospinal fluid flow, brain death, epilepsy, movement disorders, and brain perfusion reserve in cerebrovascular disease.
3. Create and present parametric brain mapping.
INTRODUCTION
Imaging assessment provides important clues on central nervous system pathology when signs and symptoms suggest an abnormality referable to the brain or spinal cord. Computed tomography (CT) and magnetic resonance imaging (MRI) are mainstay imaging modalities in this clinical setting. However, scintigraphy provides valuable and clinically relevant functional information that complements the findings on anatomic imaging. Various single photon and positron emitting radiopharmaceuticals are available clinically to interrogate the pathophysiology of the central nervous system. This chapter summarizes the major clinical applications of scintigraphy in the imaging assessment of central nervous system including the relevant radiotracers and the imaging techniques.
CLINICAL APPLICATIONS
There are many clinical applications for scintigraphic evaluation of the central nervous system. The essential applications include the following indications, which will be discussed in this chapter briefly. Scintigraphic imaging of dementia and brain tumors is often performed with positron emission tomography and will be presented in Chapter 14.
Cerebrospinal fluid (CSF) flow (normal pressure hydrocephalus; leak; shunt patency)
Brain death
Epilepsy
Movement disorders
Cerebrovascular disease
Dementia
Cerebrospinal fluid flow imaging
CSF is formed by the ventricular choroid plexus as an ultrafiltrate of plasma and absorbed primarily in the arachnoid villi. The total CSF volume is about 120 mL to 150 mL. Scintigraphy provides an effective imaging method to track the CSF flow, which may be hindered in a number of pathological conditions. The major conditions include communicating hydrocephalus, CSF leaks, and assessment of shunt patency.
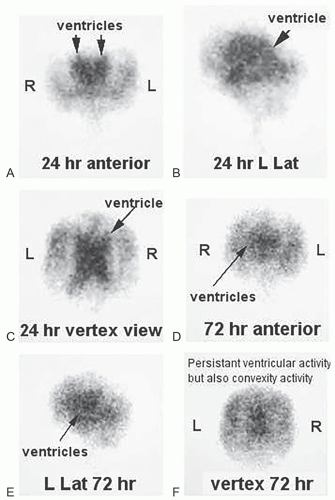
The most common relevant radiotracer for CSF flow imaging 111In-DTPA, which has a sufficiently long half-life (2.8 days) suited for serial imaging. The radiotracer (˜500 uCi) is injected into the subarachnoid space by lumbar puncture. Planar imaging is obtained serially at various intervals (2 h, 24 h, 48 h, and possibly 72 h).
In a normal scan, the activity injected via the lumbar puncture ascends the spinal canal toward the basal cisterns in a few hours (˜2-4 h). Delayed images at 24 h will demonstrate further ascent of the activity over bilateral convexities. Presence of significant activity in lateral ventricles is abnormal. If necessary, additional imaging at 48 h and 72 h can be performed to follow the CSF flow if it is relatively slow to reach the convexities.
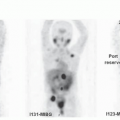
In communicating (patent foramina of Magendie and Luschka) hydrocephalus (aka. normal pressure hydrocephalus, NPH), anatomic imaging (e.g., CT) demonstrates hydrocephalus without significant cerebral atrophy (1). In rare patients, the classic triad of ataxia, dementia, and urinary incontinence may be seen clinically. Radionuclide cisternogram shows abnormal CSF flow with no transit of the activity over the convexities at 24 h (or longer) with reflux of activity into the enlarged lateral ventricles. The peripheral obstruction to CSF flow is often at the level of temporoparietal lobes (Fig. 7.1)
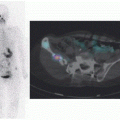
Localization of CSF leaks (e.g., CSF rhinorrhea or otorrhea) is another major clinical application for radionuclide cisternography. In the more common CSF rhinorrhea, pledgets are placed in the nostrils by otorhinolaryngologist before lumbar puncture injection of the radiotracer. Imaging is performed 1 to 3 h after tracer administration followed by removal of the pledgets at 4 to 6 h, which are then counted for radioactivity in a well counter. Contemporaneous blood sample are obtained and counted to serve as background activity. Pledget/serum activity ratio is calculated. The ratio above a selected value (typically 1.5) is considered abnormal and consistent with CSF leak. It should be noted
that there might be no imaging evidence of CSF leak despite abnormal pledget/serum ratio.
that there might be no imaging evidence of CSF leak despite abnormal pledget/serum ratio.
Obstruction of ventriculoperitoneal (VP) shunts is a relatively common complication. Scintigraphy can be helpful in localizing the site of obstruction. In this clinical setting, the radiotracer (e.g., 111In-DTPA or 99mTc-DTPA) is injected in sterile manner into the reservoir followed by serial imaging. It is important to note that while 99mTc-DTPA is superior to 111In-DTPA as an imaging agent with better counts at lower radiation exposure, 99mTc-DTPA is not food and drug administration (FDA) approved for potentially intrathecal administration due to the small risk of endotoxins in some forms of this tracer (2). In the normal nonobstructive pattern, diffuse activity reaches the peritoneal cavity in no later than 1 h. Failure in observing peritoneal activity or only focal activity at the distal end of the shunt suggests shunt obstruction. Manual occlusion of the distal limb of the shunt at the time of radiotracer injection can potentially assess the patency of the proximal limb of the shunt system. However, this should be tailored to the clinical situation and the particular CSF shunt system.
Brain death
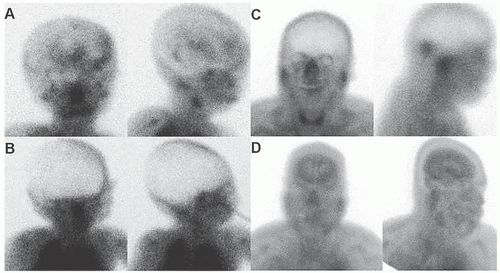
Brain death is determined by a number of clinical parameters that may include imaging cerebral perfusion scintigraphy (Fig. 7.2). Absence of brain perfusion is compatible with brain death and can be an effective visual tool in aiding difficult discussions about prognosis with patient’s family. Local guidelines may be followed for identification and declaration of brain death. Scintigraphic evaluation of cerebral blood flow is accomplished by injecting a radiotracer intravenously followed by dynamic imaging of the head and neck. Blood flow to superficial scalp vessels (external carotid artery territory) may be diminished by an elastic band place over the forehead. However, this maneuver is rarely necessary or performed. Injection of radiotracers that cross the blood-brain barrier and map the cerebral perfusion (e.g., 99mTc-HMPAO or 99mTc-ECD) is less dependent on observation of good bolus arrival in the bilateral common carotid arteries or ancillary signs such as the “hot nose” sign. Planar imaging of cerebral perfusion will include anterior-posterior and lateral views.
With single photon emission computed tomography (SPECT) imaging, the intracerebral perfusion is clearly depicted without the limitation that may be caused from superficial scalp perfusion. An “empty cranium” is typical for brain death. Occasionally, parts of the brain (e.g., cerebellum in posterior fossa) may show perfusion while there is absence of perfusion in the rest of the brain. Prognosis is poor in this clinical scenario and often repeat brain perfusion scan shows disappearance of the perfusion in entire brain in view of increased intracranial pressure. A practice guideline for brain death scintigraphy has been published (3).
With single photon emission computed tomography (SPECT) imaging, the intracerebral perfusion is clearly depicted without the limitation that may be caused from superficial scalp perfusion. An “empty cranium” is typical for brain death. Occasionally, parts of the brain (e.g., cerebellum in posterior fossa) may show perfusion while there is absence of perfusion in the rest of the brain. Prognosis is poor in this clinical scenario and often repeat brain perfusion scan shows disappearance of the perfusion in entire brain in view of increased intracranial pressure. A practice guideline for brain death scintigraphy has been published (3).
Epilepsy
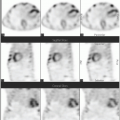
Clinical workup of epilepsy involves a number of diagnostics procedures including electroencephalography and imaging. In intractable seizures that are not well controlled with medications, localization, and resection of the epileptogenic focus can potentially be curative. MRI is often performed to detect potential morphologic abnormalities (e.g., mesial temporal sclerosis) that may explain the observed seizure activity in correlation with electroencephalography. However, there are occasions that MRI does not depict a specific relevant abnormality. Functional imaging with scintigraphy can be helpful in localization of epileptogenic focus with or without specific MRI abnormality (4,5). Brain perfusion scintigraphy with 99mTc-HMPAO or 99mTc-ECD can be performed in the inter-ictal or intra-ictal periods. In interictal period, the brain perfusion SPECT shows hypoperfusion in the region of the brain that may harbor the epileptogenic focus. Such finding can be corroborated with any MRI abnormalities at that location or with intra-ictal brain perfusion SPECT in which the same locus will demonstrate hyperperfusion (Fig. 7.3). Good quality intra-ictal demands special seizure units in which the patients are monitored actively for seizure activity. When the onset of the seizure activity is observed (clinically and/or with electroencephalography), the previously ready at-hand radiotracer is injected immediately in a previously accessed peripheral vein. The patient is then transferred to nuclear medicine clinic for imaging when the patient is clinically stable. If performed in a timely manner and properly, intra-ictal brain perfusion SPECT is more sensitive in localizing the epileptogenic focus than interictal brain perfusion SPECT.
Movement disorders
Clinical workup of patients with tremor is directed toward differential diagnosis of a variety of etiologies, which lead to different management algorithms. While some patients present with classic sign and symptoms that suggest a particular diagnosis, this is not always the case and uncertainty for an etiology may remain.
123I-Ioflupane (Iodine 123-fluoropropyl (FP)-carbomethoxy-2 beta-(4-iodophenyltropane) or DaTscan with single photon CT is approved for detecting loss of Dopamine transporters (DATs) from presynaptic dopaminergic neuron terminals in the striatum. DaTscan is therefore useful in detecting those conditions that lead to loss of striato-nigral dopamine neurons that occurs in Parkinsonian syndromes (idiopathic Parkinson’s disease, multiple system atrophy, progressive supranuclear palsy, corticobasal degeneration) and in dementia with Lewy bodies (6). However, DaTscan is unable to distinguish among these aforementioned conditions, which will require correlation with other relevant clinical information. DaTscan will show preserved striatonigral dopamine neurons in healthy individual, essential tremor,
psychogenic tremor, and drug-induced parkinsonism. Practice guideline for performing DaTscan has been published (7).
psychogenic tremor, and drug-induced parkinsonism. Practice guideline for performing DaTscan has been published (7).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree