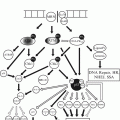
Emetogenic potential
Typical agents
Definition (no CINV prevention)
High
Cisplatin
Emesis in nearly all patients
Dacarbazine
Melphalan (high dose)
Nitrogen mustard
Cyclophosphamide plus an Anthracycline
Moderate
Anthracyclines
Emesis in >70 % of patients
Carboplatin
Carmustine (high dose)
Cyclophosphamide
Ifosfamide
Irinotecan
Methotrexate (high dose)
Oxaliplatin
Topotecan
Low
Etoposide
Emesis in 10–70 % of patients
5-Fluorouracil
Gemcitabine
Mitoxantrone
Taxanes
Vinblastine
Vinorelbine
Minimal
Bortezomib
Emesis in <10 % of patients
Hormones
Vinca alkaloids
Bleomycin
Table 37.2
Patient-related risk factors for emesis following chemotherapy
Major factors | Minor factors |
|---|---|
Female | History of motion sickness |
Age <50 years | Emesis during past pregnancy |
History of low prior chronic alcohol intake (<1 oz of alcohol/day) | |
History of previous chemotherapy-induced emesis |
The use of 5-hydroxytryptamine-3 (5-HT3) receptor antagonists plus dexamethasone has improved the control of CINV [5, 6]. Recent studies have demonstrated some improvement in the control of CINV with the use of three new agents, palonosetron, a second generation 5-HT3 receptor antagonist [5, 6], aprepitant, the first agent available in the drug class of neurokinin-1 (NK-1) receptor antagonists [7, 8], and olanzapine, an antipsychotic which blocks multiple neurotransmitters in the central nervous system [9–12].
The primary endpoint used for studies evaluating various agents for the control of CINV has been complete response (no emesis, no use of rescue medication) over the acute (24 h post-chemotherapy), delayed (24–120 h), and overall (0–120 h) periods [3, 4]. Recent studies have shown that the combination of a 5-HT3 receptor antagonist, dexamethasone, and a NK-1 receptor antagonist have improved the control of emesis in patients receiving either highly emetogenic chemotherapy (HEC) or moderately emetogenic chemotherapy (MEC) over a 120 h period following chemotherapy administration [7, 8]. Many of these same studies have measured nausea as a secondary endpoint and have demonstrated that nausea has not been well controlled [13].
Emesis is a well defined event which is easily measured, but nausea may be more subjective and more difficult to measure. There are, however, two well defined measures of nausea which appear to be effective measurement tools which are reproducible: the Visual Analogue Scale (VAS) and the Likert Scale [14]. The VAS is a scale from 0 to 10 or 0 to 100 with zero representing no nausea and 10 or 100 representing maximal nausea. The Likert Scale asks patients to rate nausea as None, Mild, Moderate or Severe. Many studies have reported the secondary endpoint of “no significant nausea” or “only mild nausea” [3–8]. Studies that have reported “no nausea” may be more useful in identifying the most effective available antinausea agents [14].
Despite the introduction of more effective antiemetic agents, emesis and nausea remain a significant complication of chemotherapy. The purpose of this chapter is to evaluate the clinical agents available for the prevention and treatment of chemotherapy induced emesis and nausea. The use of these agents in various clinical settings is described using the recently established guidelines from the Multinational Association of Supportive Care in Cancer (MASCC) and the European Society of Medical Oncology (ESMO) [15], the American Society of Clinical Oncology (ASCO) [16] and the National Comprehensive Cancer Network (NCCN) [17]. The literature cited in the report consists of the primary clinical trials used for the U.S. FDA approval of the various agents as well as recent comprehensive reviews.
37.1.1 Pathophysiology of Nausea and Vomiting
The sensation of nausea and the act of vomiting are protective reflexes that rid the intestine and stomach of toxic substances. The experience of nausea is subjective, and nausea may be considered a prodromal phase to the act of vomiting [14] although significant nausea may occur without vomiting. Vomiting consists of a pre-ejection phase, retching, and ejection and is accompanied by shivering and salivation. Vomiting is triggered when afferent impulses from the cerebral cortex, chemoreceptor trigger zone (CTZ), pharynx, and vagal afferent fibers of the gastrointestinal (GI) tract travel to the vomiting center (VC), located in the medulla (Fig. 37.1). Efferent impulses then travel from the vomiting center to the abdominal muscles, salivation center, cranial nerves, and respiratory center, causing vomiting. It is thought that chemotherapeutic agents cause vomiting by activating neurotransmitter receptors located in the CTZ, GI tract, and vomiting center [14].
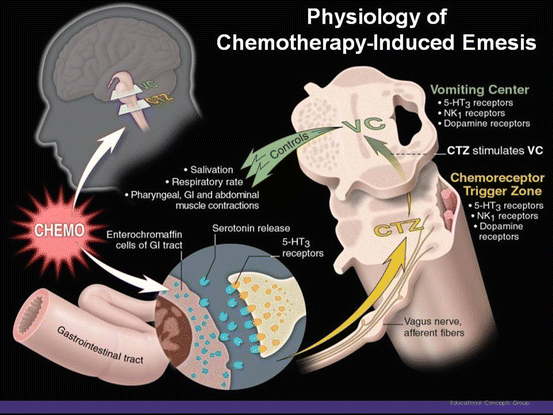

Fig. 37.1
Proposed pathways of chemotherapy-induced emesis
The mechanisms of emesis are not well defined, but investigations suggest that emesis may be primarily mediated through neurotransmitters (serotonin, dopamine, substance P) in the GI tract and the central nervous system [14]. Figure 37.1 shows that chemotherapy agents may directly affect areas in the cerebral cortex, the medulla oblongata, or may stimulate the small intestine of the GI tract via the vagus nerve. A VC, termed the “central pattern generator” by some authors [18], appears to be located in the lateral reticular formation of the medulla, which coordinates the mechanism of nausea and vomiting. An additional important area, also located in the medulla, is the CTZ in the area postrema near the fourth ventricle [18]. It is strongly suspected that the nucleus tractus solitarius (NTS) neurons lying ventrally to the area postrema initiate emesis [19]. This medullary area is a convergence point for projections arising from the area postrema and the vestibular and vagal afferents [19]. The NTS is a good candidate for the site of action of centrally acting antiemetics.
The main approach to the control of emesis has been to identify the active neurotransmitters and their receptors in the central nervous system and the GI tract that mediate the afferent inputs to the VC (Fig. 37.2). Agents that may block these neurotransmitter receptors in the CTZ, the VC, or the GI tract may be useful in preventing or controlling emesis (Table 37.3).
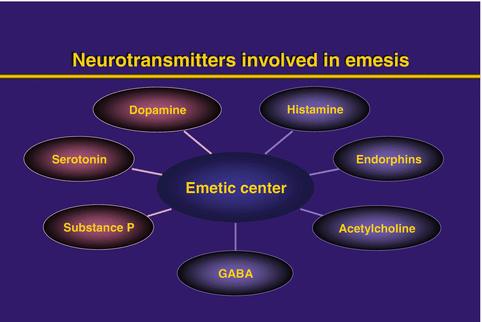

Fig. 37.2
Neurotransmitters involved in emesis
Table 37.3
Antiemetic receptor antagonists
Dopamine receptor antagonists | 5-HT3 receptor antagonists | Dopa-5-HT3 receptor antagonists | NK-1 receptor antagonists |
|---|---|---|---|
Butyrophenones | Azasetron | Metoclopramide | Aprepitant (MK-869) |
Olanzapine | Dolasetron (not recommended for use per FDA) | Fosaprepitant | |
Phenothiazines | Granisetron | Casopitant | |
Olanzapine | Netupitant | ||
Ondansetron (intravenous dose restriction per FDA) | Rolapitant | ||
Palonosetron | |||
Ramosetron | |||
Tropisetron |
Nausea is a difficult-to-describe, sick or queasy sensation, usually perceived as being in the stomach that is sometimes followed by emesis [14]. The experience of nausea is difficult to describe in another person. Nausea and emesis are not necessarily on a continuum. One can experience nausea without emesis and one can have sudden emesis without nausea. Nausea has been assumed to be the conscious awareness of unusual sensations in the VC of the brainstem (Fig. 37.1), but the existence of such a center and its relationship to nausea remain controversial [14].
The study of the receptors that are illustrated in Fig. 37.2 has guided the development of the antagonists to the serotonin and the substance-P receptors with relative success in controlling emesis. It is not clear whether the serotonin and/or the substance P receptors are important in the control of nausea. Other receptors such as dopaminergic, histaminic and muscarinic may be the dominant receptors in the control of nausea [3, 4, 13].
37.1.2 Types of CINV
Five categories are used to classify CINV: acute, delayed, anticipatory, breakthrough, and refractory. Nausea and vomiting may occur any time after the administration of chemotherapy, but the mechanisms appear different for CINV occurring in the first 24 h after chemotherapy in contrast to that which occurs in the period of 1–5 days after chemotherapy. In order to differentiate these mechanisms, the term acute-onset CINV refers to nausea and/or vomiting occurring within 24 h of chemotherapy administration [3, 4]. The incidence of acute emesis and/or nausea reflects several treatment-related factors, including the environment in which chemotherapy is administered, the emetogenicity of the chemotherapy, the dosage of the emetogenic agents, and patient-related factors [3, 4, 20].
Nausea and/or vomiting that develop more than 24 h after chemotherapy administration is known as delayed emesis and/or nausea. Typically occurring with administration of cisplatin, doxorubicin, or cyclophosphamide, delayed emesis/nausea is more common in those who experience acute emesis/nausea. Other predictive factors include the dose and the emetogenicity of the chemotherapeutic agent, patient gender and age, and protection against nausea and vomiting in previous cycles of chemotherapy [1, 3, 4, 20]. For cisplatin, which has been most extensively studied, delayed emesis reaches peak intensity 2–3 days subsequent to chemotherapy administration and can last up to a week [1, 3, 4, 15–17, 20].
If patients experience CINV, they may develop a conditioned response known as anticipatory nausea and/or vomiting which occurs prior to the administration of chemotherapy in future chemotherapy cycles and is attributed to the adverse memory of prior CINV. Incidence rates for this type of nausea and vomiting range from 10 % to 45 %, with nausea occurring more frequently [1, 3, 4, 20].
Vomiting and/or nausea that occurs within 5 days after prophylactic use of antiemetic agents or requires “rescue” is called breakthrough emesis [21]. Vomiting and/or nausea occurring after chemotherapy in subsequent chemotherapy cycles when antiemetic prophylaxis and/or rescue have failed in earlier cycles is known as refractory emesis [1, 3, 4, 15–17, 20].
37.2 Antiemetic Agents
37.2.1 Dopamine Receptor Antagonists
Dopamine receptors are known to exist in the CTZ, and this is the main area of activity of the dopamine antagonists, such as the phenothiazines and the butyrophenones (droperidol, haloperidol). A high level of blockade of the dopamine receptors, however, results in extrapyramidal reactions, as well as disorientation and sedation, limiting the clinical use of these agents. Their current use is primarily to treat established nausea and emesis and not for CINV prophylaxis [17].
37.2.2 Serotonin (5-HT3) Receptor Antagonists
Serotonin receptors, specifically the 5-HT3 receptors, exist in the central nervous system and in the GI tract. The 5-HT3 receptor antagonists appear to act through both the central nervous system and the GI tract via the vagus and splanchnic nerves. The main toxicities of these 5-HT3 receptor antagonists consist only of a mild headache and occasional diarrhea [22, 23].
The introduction of 5-HT3 receptor antagonists for the prevention of chemotherapy-induced nausea and emesis, as well as post-operative and radiotherapy- induced nausea and vomiting, has resulted in an improvement in supportive care [22, 23]. Treatment guidelines for the prevention of CINV recommended by a number of international groups [15–17] suggest the use of a 5-HT3 receptor antagonist and dexamethasone prechemotherapy for the prevention of acute CINV and the use of dexamethasone following chemotherapy for the prevention of delayed nausea and vomiting.
37.2.2.1 First Generation Serotonin (5HT3) Receptor Antagonists
Table 37.4 shows the 5-HT3 receptor antagonists currently in use. The first generation serotonin (5-HT3) receptor antagonists, dolasetron, granisetron, and ondansetron, tropisetron [24], azasetron [25] and ramosetron [26], are equivalent in efficacy and toxicities when used in the recommended doses and compete only on an economic basis [27]. The most commonly reported adverse events being mild headache, constipation, and occasionally mild diarrhea [3, 4]. Azasetron and ramosetron are not available in North America and Europe and have not been compared extensively to the other 5-HT3 receptor antagonists. They are marketed primarily in southeast Asia.
Table 37.4
Serotonin antagonists and dosage before chemotherapy
Antiemetic | Route | Dosage |
|---|---|---|
Azasetron | IV | 10 mg |
Dolasetron (not recommended for use per FDA) | IV | 100 mg or 1.8 mg/kg |
PO | 100 mg | |
Granisetron | IV | 10 μg/kg or 1 mg |
PO | 2 mg (or 1 mg twice daily) | |
Ondansetron | IV | 8 mg (restricted to <16 mg) |
PO | 24 mg | |
Palonosetron | IV | 0.25 mg |
PO | 0.50 mg | |
Ramosetron | IV | 0.30 mg |
Tropisetron | IV or PO | 5 mg |
A prolongation of cardiac conduction intervals has been reported for this class of compounds with dolasetron being more extensively studied than granisetron and ondansetron [28]. In 2006, Canada issued a drug alert for dolasetron, due to the potential of serious cardiovascular adverse events (cardiac arrhythmias) [29], stating that dolasetron was not indicated for use in children, but only for prevention of CINV in adults [29]. Subsequently, in 2010, the U.S. FDA announced that the intravenous form of dolasetron should no longer be used to prevent CINV in any patient. New data suggests that dolasetron injection can increase the risk of developing a prolongation of the QTc interval which may potentially precipitate life threatening ventricular arrhythmias [30, 31].
In 2012, the U.S. FDA placed a restriction on the doses of intravenous ondansetron due to the risk of prolongation of the QTc interval [32]. Patients who may be at particular risk for QT prolongation with ondansetron are those with congenital long QT syndrome, congestive heart failure, bradyarrhythmias, or patients taking concomitant medications that prolong the QTc interval. New information indicates that QT prolongation occurs in a dose-dependent manner, and specifically at a single intravenous dose of 32 mg. The lower dose intravenous regimen of 0.15 mg/kg every 4 h for three doses may be used in adults with CINV. However, no single intravenous dose of ondansetron should exceed 16 mg due to the risk of QT prolongation. The new information does not change any of the recommended oral dosing regimens for ondansetron, including the single oral dose of 24 mg for CINV [32].
The first generation 5-HT3 receptor antagonists have not been as effective against delayed emesis as they are against acute CINV [33–35]. The first generation 5-HT3 receptor antagonists alone do not add significant efficacy to that obtained by dexamethasone in the control of delayed emesis [34]. Hickok et al. [35] reported that the first generation 5-HT3s used in the delayed period were no more effective than prochlorperazine in controlling nausea. The antiemetic effects of prochlorperazine can be attributed to postsynaptic dopamine receptor blockade in the CTZ. A meta analysis [34] showed that there was neither clinical evidence nor considerations of cost effectiveness to justify using the first generation 5-HT3 antagonists beyond 24 h after chemotherapy for the prevention of delayed emesis.
A number of recent studies have demonstrated that there has been poor control of delayed nausea by the first generation 5-HT3 receptor antagonists in patients receiving HEC or MEC [10, 13, 36, 37] (Table 37.5). The use of granisetron and dexamethasone in patients receiving HEC resulted in “no nausea” in 25–27 % of patients [36]. The use of ondansetron plus dexamethasone in patients receiving MEC resulted in “no nausea” in 33 % of patients and “no significant nausea” in 56 % of patients [37].
Table 37.5
Phase II and III trials of various agents for the treatment of chemotherapy induced nausea
Study | Chemotherapy | Phase II or III | No. patients | No nausea, delayed (%) | No nausea, overall (%) |
|---|---|---|---|---|---|
Saito et al. [36] | HEC | III | 1,114 | Palo+Dex: 38* | Palo+Dex: 32* |
Gran+Dex: 27 | Gran+Dex: 25 | ||||
Hesketh et al. [38] | HEC | III | 1,043 | No data | Women: |
Aprepitant: 46 | |||||
Control: 38 | |||||
Men: | |||||
Aprepitant: 50 | |||||
Control: 44 | |||||
Warr et al. [39] | Aprepitant 52* | Aprepitant 48* | |||
Control 44 | Control 42 | ||||
– | |||||
Warr et al. [37] | Cyclo+Doxo/Epi | III | 866 | Aprepitant: 37 | Aprepitant: 33 |
Control: 36 | Control: 33 | ||||
Grote et al. [40] | MEC | II | 58 | APD: 31 | APD: 30 |
Celio et al. [41] | MEC | III | 334 | Palo+Dex1: 57 | Palo+Dex1: 52 |
Palo+Dex3: 62 | Palo+Dex3: 57 | ||||
Aapro et al. [42] | Cyclo+Doxo/Epi | III | 300 | Palo+Dex1: 50 | Palo+Dex1: 47 |
Palo+Dex3: 55 | Palo+Dex3: 50 | ||||
Navari et al. [9] | MEC | II | 32 | OPD: 78 | OPD: 78 |
Tan et al. [10] | MEC | III | 229 | OAD: 83* | OAD: 83* |
HEC | III | AD: 58 | AD: 56 | ||
OAD: 70* | OAD: 70* | ||||
AD: 30 | AD: 28 | ||||
Navari et al. [11] | HEC | III | 257 | OPD: 69* | OPD: 69* |
APD: 38 | APD: 38 | ||||
Cruz et al. [43] | HEC | III | 80 | Gabapentin: 72 | Gabapentin: 62 |
Control: 52 | Control: 45 | ||||
Meiri et al. [44] | MEC, HEC | III | 61 | No difference between dronabinol or ondansetron | Not reported |
37.2.2.2 Second Generation Serotonin (5-HT3) Receptor Antagonist: Palonosetron
Palonosetron is a second generation 5-HT3 receptor antagonist which has antiemetic activity at both central and GI sites. In comparison to the first generation 5- HT3 receptor antagonists, it has a higher potency, a significantly longer half-life, and a different molecular interaction with 5-HT3 receptors [5, 6, 45, 46]. Palonosetron has been approved for clinical use, and studies suggest that it may have some efficacy in controlling delayed CINV compared to the first generation 5-HT3 receptor antagonists.
Palonosetron demonstrated a 5-HT3 receptor binding affinity at least 30-fold higher than other 5-HT3 receptor antagonists [45]. Rojas et al. [46] recently reported that palonosetron exhibited allosteric binding and positive cooperativity when binding to the 5-HT3 receptor compared to simple bimolecular binding for both granisetron and ondansetron. Additional studies by Rojas et al. [46] suggested that palonosetron triggers 5-HT3 receptor internalization and causes prolonged inhibition of receptor function. Differences in binding and effects on receptor function may explain some differences between palonosetron and the first generation 5-HT3 receptor antagonists [5, 6]. These differences may explain palonosetron’s efficacy in delayed CINV compared to the first generation receptor antagonists [5, 6].
Phase III comparative studies [47–49] suggest that the use of palonosetron alone improves the complete response rate of acute and delayed emesis, when compared with the use of the first generation 5-HT3 receptor antagonists alone in patients receiving MEC [48, 49]. In patients receiving HEC, palonosetron was as effective as ondansetron in the prevention of acute CINV and with dexamethasone pre-treatment, palonosetron was significantly better than ondansetron in the overall 120-h post-treatment period [47].
In patients receiving HEC, a recent study showed that palonosetron plus dexamethasone was significantly better than granisetron and dexamethasone in delayed complete response and control of nausea, but there was a low number of patients with no nausea with either regimen (no nausea, overall period: 31.9 % palonosetron group; 25.0 % granisetron group) [36] (Table 37.5).
Two recent studies reported that palonosetron plus 1 day of dexamethasone was as effective as palonosetron plus 3 days of dexamethasone in the prevention of acute and delayed CINV in patients receiving MEC [41, 42]. Boccia et al. recently demonstrated that oral palonosetron had similar efficacy and safety as intravenous palonosetron for the prevention of acute CINV in patients receiving MEC [50].
In a systematic review and meta-analysis of all randomized controlled trials comparing a single dose of palonosetron with other 5-HT3 receptor antagonists, Borrel et al. [51] concluded that palonosetron was more effective than the first generation receptor antagonists in preventing acute and delayed CINV in patients receiving MEC or HEC, regardless of the use of concomitant corticosteroids. In an additional systematic review of the medical literature, Fabi and Malaguti [52], reported that palonosetron was the only serotonin receptor antagonist approved for the prevention of delayed CINV caused by MEC.
The safety and tolerability of palonosetron has been well documented in multiple, large phase III trials. There were no clinically relevant differences seen among palonosetron, ondansetron, or dolasetron in laboratory, electrocardiographic, or vital sign changes over multiple cycles of chemotherapy [48, 49, 51–55]. The adverse reactions reported were the most common reactions reported for the 5-HT3 receptor antagonist drug class. There have been no reports of any adverse cardiac events with palonosetron, specifically no prolongation of the QTc interval in healthy volunteers or patients receiving repeated cycles of emetogenic chemotherapy [5, 6, 53–55].
Based on the clinical studies, palonosetron is highly effective in controlling acute and delayed CINV in patients receiving either MEC or HEC. Compared to the first generation 5-HT3 receptor antagonists, palonosetron has equivalent efficacy in controlling acute CINV and is more effective in controlling delayed CINV.
The published clinical studies on palonosetron have prompted the international guideline groups to recommend palonoseteron as the preferred 5-HT3 receptor antagonist for the prevention of acute nausea and vomiting for patients receiving HEC and for the prevention of delayed nausea and vomiting for patients receiving MEC [17].
Two recent studies have reported that the complete response rates for both acute and delayed CINV were maintained with the single intravenous dose of palonosetron in patients receiving repeated courses of HEC [53, 54].
Despite the use of both first generation and second generation 5-HT3 receptor antagonists, the control of acute CINV, and especially delayed nausea and vomiting, is sub-optimal with the agents listed in Table 37.4. There is considerable opportunity for improvement with either the addition or substitution of new agents in current regimens [23, 35, 56].
37.2.3 Dopamine-Serotonin Receptor Antagonists
Metoclopramide has antiemetic properties both in low doses as a dopamine receptor antagonist and in high doses as a serotonin receptor antagonist. The use of metoclopramide may be somewhat efficacious in relatively high doses (≥20 mg orally, three times/day) in the delayed period, but may result in sedation and extrapyramidal side effects [22, 23]. Metoclopramide has been used both as a preventative agent for CINV [23] as well as a treatment for breakthrough CINV [17].
37.2.4 Neurokinin-1 (NK-1) Receptor Antagonists
Substance P is a mammalian tachykinin that is found in vagal afferent neurons innervating the brainstem NTS, which sends impulses to the VC [57]. Substance P induces vomiting and binds to NK-1 receptors in the abdominal vagus, the NTS, and the area postrema [57]. Compounds that block NK-1 receptors lessen emesis after cisplatin, ipecac, apomorphine, and radiation therapy [49]. These observations have recently led to the development of NK-1 receptor antagonists and the study of the role they may play in controlling chemotherapy-induced nausea and emesis.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree