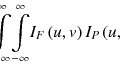Fig. 6.1
Conceptual scheme of similar-case-based radiotherapy treatment planning
6.5 Similar-Case-Based Beam Angle Optimization
The accuracy and efficiency of beam arrangement determinations could potentially be improved by combining similar-case-based treatment planning and BAO algorithms (Magome et al. 2013a, b). Figure 6.2 shows the overall scheme of the similar-case-based beam angle optimization method, which consisted of three main steps. First, cases that were similar to an objective case were automatically selected from the RTP database based on geometrical features related to structures, such as the location, size, and shape of the target and OARs. Second, the initial beam arrangements of the objective case were determined by registering similar cases to the objective case, using a linear registration technique (Burger and Burge 2007). Finally, the beam directions of the objective case were locally optimized based on the cost function, which took the radiation absorption in normal tissues and OARs into account.


Fig. 6.2
Overall scheme of similar-case-based beam angle optimization
6.5.1 Feature Extraction for Searching Similar Cases
It is very important to consider the exact meaning of “similarity” in the radiation oncology field. “Similarity” could be defined in many different ways, for example, based on the similarity of the tumor type (histological type or staging), the patient (gender, age, height, weight, etc.), or other characteristics. However, similar cases should be defined from the viewpoint of treatment planning because they are intended to be useful for the treatment planner. It should be assumed that the geometrical similarity with respect to tumor and OAR among clinical cases may be a key in the similar-case-based treatment planning. Therefore, four types of features (comprising 10 features in total) were defined for lung SBRT: the planning target volume (PTV) shape, the PTV size, the lung dimensions, and the geometrical relationship between the PTV and the spinal cord (Fig. 6.3), as assessed using the DICOM-RT structure set (Magome et al. 2013a).


Fig. 6.3
Geometrical features, which were used to search for similar cases in a lung SBRT database (Magome et al. 2013a)
The ten defined geometrical features were described as follows: PTV centroid in left-right (LR), anterior-posterior (AP), and superior-inferior (SI) directions; effective diameter of the PTV; sphericity of the PTV; lung dimension in LR, AP, and SI directions; distance between the PTV and the spinal cord in the isocenter plane; and angle from the spinal cord to the PTV in the isocenter plane. The PTV centroid was determined by registering the lung structure image of each case in the RTP database with that of a reference case, using a linear registration technique (Burger and Burge 2007). The effective diameter was defined as the diameter of a sphere with the same volume as the PTV. The sphericity was defined as the roundness of the PTV without directional dependence, and given by the ratio of the number of logical AND voxels between the PTV and its equivalent sphere with the same centroid and volume as the PTV to the number of PTV voxels. The lung dimensions were defined as the three side lengths of the circumscribed parallelepiped of the lung regions in the LR, AP, and SI directions. The distance between the PTV and spinal cord was measured between the centroid of the PTV and that of the spinal cord in the isocenter plane. The angle from the spinal cord to the PTV was defined in the two-dimensional coordinate system with the origin at the centroid of the spinal cord in the isocenter plane, and ranged from −π (clockwise) to π (counterclockwise) for a baseline of the posterior-anterior direction. Although only the PTV centroid was determined in a fixed reference coordinate system by registering the lung regions of each case in the RTP database with those of a reference case, the other features were calculated with respect to each of their original coordinate systems. The calculations were performed in this way in order to consider both the relative similarity of the tumor in the lung regions and absolute similarities, such as of the lung dimensions and spinal cord position.
6.5.2 Selection of Similar Planning Cases Using Geometrical Features
The RTP database was searched for the cases that resembled the objective case by considering the weighted Euclidean distances between the geometrical feature vector of the objective case and the geometrical feature vectors of all other cases in the RTP database (Fig. 6.4). The weighted Euclidean distance was thus regarded as a similarity measure. The weighted Euclidean distance d image was calculated using the following equation:


Fig. 6.4
Conceptual illustration of an objective case and similar cases in a n-dimensional feature space. Most similar case was defined as the case that was closest to the objective case in the feature space, as measured with a weighted Euclidean distance

(6.1)
Weights were needed for the geometrical features in order to incorporate their relative importance from the viewpoint of treatment planning. Therefore, when applying the proposed method to their own databases, each institute should determine the appropriate weights for the geometrical features based on their own philosophy or policy of treatment planning. In our investigation, the weights for geometrical features were empirically set as follows: PTV centroid (three-dimension) = 0.3, effective diameter of PTV = 0.1, sphericity of PTV = 0.1, lung dimension (three-dimension) = 0.3, distance between PTV and spinal cord = 1.0, and angle from spinal cord to PTV = 1.0.
6.5.3 Determination of Initial Beam Arrangements Based on the Linear Registration Technique
In the second step, beam arrangements for the objective case were automatically determined (Magome et al. 2013a) based on the registration of similar cases with the objective case in terms of lung regions, using a linear registration technique (i.e., affine transformation) (Burger and Burge 2007). The beam arrangement of the similar case was modified to fit the objective case with respect to the lung regions.
First, a beam angle (i.e., a beam direction with a gantry angle θ and couch angle φ) was described as a point in a Cartesian coordinate system. As shown in Fig. 6.5, the beam direction with gantry angle θ and couch angle φ can be considered as a line in a spherical polar coordinate system, with the origin as the isocenter. An arbitrary point (x θ,φ , y θ,φ , z θ,φ ) on the line is described in a Cartesian coordinate system as follows:

where r is distance from the isocenter (x iso , y iso , z iso). In this study, r has no meaning (r = 1 for simplicity) because the purpose of the analysis is registration of the beam angle.


(6.2)

Fig. 6.5
Illustration of a beam direction with gantry angle θ and couch angle φ in a spherical polar coordinate system and a Cartesian coordinate system. Here, the origin indicates an isocenter and couch angle φ is defined with respect to the patient-fixed coordinate system
Second, each beam point of the similar case in the Cartesian coordinate system was modified based on a linear registration technique (i.e., an affine transformation) (Burger and Burge 2007). Please note that the linear registration maps straight lines to straight lines, and thus the beam directions—which can be considered as points—are uniquely and automatically determined by the registration. The beam point (x θ,φ , y θ,φ , z θ,φ ) was modified to the point (x′ θ,φ , y′ θ,φ , z′ θ,φ ) by using the affine transformation matrix to register the lung regions of each similar case with those of the objective case, as follows:

where u 11 …u 34 are the transformation parameters. The isocenter point (x iso , y iso , z iso) was also modified to the point (x′iso , y′iso , z′iso) in the same manner. The affine transformation can apply a linear combination of translation, scaling, rotation, and/or shear mapping. Further details can be found in the literature (Burger and Burge 2007). The vertices of a circumscribed parallelepiped of a lung (including the left and right lung regions) were automatically obtained as feature points to calculate the parameters of the affine transformation matrix. In this study, the circumscribed parallelepiped was chosen to reduce the calculation time that was necessary to find the feature points of the lung.

(6.3)
Finally, the resulting direction vector (x′ θ,φ – x′ iso , y′ θ,φ – y′ iso , z′ θ,φ – z′ iso) in the Cartesian coordinate system was converted into the spherical polar coordinate system as gantry angle θ′ and couch angle φ′, as follows:



(6.4)

(6.5)
6.5.4 Local Optimization of Beam Arrangements
The beam directions of the objective case were locally optimized based on the cost function, which took into account the radiation absorption in normal tissues and OARs (Magome et al. 2013b). Although Meyer et al. (2005) developed the cost function for a global optimization of beam arrangements, the cost function was used for the local optimization of each beam direction in this study. The cost function C θ,φ of a beam with gantry angle θ and couch angle φ was defined as follows:

where C θ,φ (PTV) represents the dose absorption in normal tissue until the X-ray beams reach the PTV surface, C θ,φ (OAR k ) is a term for the irradiation of k-th OAR, and w k is a weight for the k-th OAR. The first term C θ,φ (PTV) was determined by the following equation:

where μ is a linear attenuation coefficient in water, and d θ,φ (PTV) is the mean distance in centimeters from the body surface to the PTV surface. The second term for the k-th OAR C θ,φ (OAR k ) was defined as follows:

where v θ,φ (OAR k ) is an irradiated fractional volume of the k-th OAR, d θ,φ (OAR k ) is the mean depth from the body surface to the k-th OAR surface, and λ is a parameter for controlling the relative significance of the first and second terms. The term exp(−μd θ,φ (OAR k )) represents the number of incident photons in the k-th OAR. Figure 6.6 presents a conceptual illustration of the cost function.


(6.6)

(6.7)

(6.8)

Fig. 6.6
Conceptual diagram of the cost function for local optimization of beam angles
Each beam direction was locally optimized in the range of ± p degrees at an interval of q degrees. The lung and spinal cord were incorporated as OARs in the cost function, and both weights (for the lung and the spinal cord) were set to 5.0. The parameters for the local optimization of the beam arrangement λ, p, and q, were set to 0.6, 4°, and 2°, respectively. Although the parameters for the local optimization of beam arrangements were set empirically based on the preferences of our institution, each institute could determine the appropriate parameters based on their own philosophy or policy of treatment planning, in resemblance with the geometrical feature weights. Each optimal beam direction was defined as the direction of the beam which had the lowest cost value among the beam directions of the local range.
6.5.5 Evaluation of Beam Arrangements Using Planning Evaluation Indices
The similar-case-based beam arrangements were evaluated by manually preparing plans based on both the beam arrangements and other planning parameters (such as nominal beam energies, collimator angles, and beam weight) derived from the treatment plans of similar cases in a radiation treatment planning system. The following 11 planning evaluation indices were used for validation.
The planning evaluation indices for the PTVs calculated in this study were the D95, homogeneity index (HI), conformity index (CI), and tumor control probability (TCP). The D95 was defined as the minimum dose in the PTV that encompassed at least 95 % of the PTV. The HI was calculated as the ratio of the maximum dose to the minimum dose in the PTV. The CI was the ratio of the treated volume to the PTV. The treated volume was defined as the tissue volume that was receiving the minimum PTV dose. The TCP was estimated based on a linear-quadratic (LQ) model according to a Poisson distribution by considering the radiosensitivity variation and nonuniform dose distribution (Sanchez-Nieto and Nahum 1999, 2000). The TCP was averaged over a population with variability in radiosensitivity, which was simulated as a Gaussian distribution of α k values with mean  and standard deviation σ α in K groups of patients (Webb and Nahum 1993; Kanai et al. 2006). Specifically, TCP was given by
and standard deviation σ α in K groups of patients (Webb and Nahum 1993; Kanai et al. 2006). Specifically, TCP was given by
![$$ \mathrm{TCP}={\displaystyle \sum_{k=1}^K\left(\frac{1}{\sqrt{2\pi}{\sigma}_{\alpha}}\right) \exp \left\{-\frac{{\left({\alpha}_k-\overline{\alpha}\right)}^2}{2{\sigma}_{\alpha}^2}\right\}}\times {\displaystyle \prod_{l=1}^L \exp \left[-{\rho}_c{v}_l\cdot \exp \left\{-{\alpha}_k{D}_l\left(1+\frac{d_l}{\alpha_k/{\beta}_k}\right)\right\}\right]}, $$](http://radiologykey.com/wp-content/uploads/2017/07/A340376_1_En_6_Chapter_Equ9.gif)
where ρ c is the number of initial clonogenic cells per volume cm3, L is the number of dose bins of the differential dose-volume histogram (DVH) in the PTV, v l is the volume (cm3) irradiated by a dose d l (Gy) per fraction in the PTV, and D l is the total dose (Gy) at v l . The α k ranged α k ± γσ α , and increased in certain intervals divided by K. The parameters for the TCP calculation were obtained from Kanai et al.’s (2006) study of patients with lung cancers.
 and standard deviation σ α in K groups of patients (Webb and Nahum 1993; Kanai et al. 2006). Specifically, TCP was given by
and standard deviation σ α in K groups of patients (Webb and Nahum 1993; Kanai et al. 2006). Specifically, TCP was given by![$$ \mathrm{TCP}={\displaystyle \sum_{k=1}^K\left(\frac{1}{\sqrt{2\pi}{\sigma}_{\alpha}}\right) \exp \left\{-\frac{{\left({\alpha}_k-\overline{\alpha}\right)}^2}{2{\sigma}_{\alpha}^2}\right\}}\times {\displaystyle \prod_{l=1}^L \exp \left[-{\rho}_c{v}_l\cdot \exp \left\{-{\alpha}_k{D}_l\left(1+\frac{d_l}{\alpha_k/{\beta}_k}\right)\right\}\right]}, $$](http://radiologykey.com/wp-content/uploads/2017/07/A340376_1_En_6_Chapter_Equ9.gif)
(6.9)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree