Chapter Outline
Colonic diverticula are acquired herniations of the mucosa and of portions of the submucosa through the muscularis propria. Diverticular disease of the colon represents a continuum from an initial, prediverticular phase of marked muscular thickening of the colon wall (myochosis) to frank outpouchings (diverticulosis) and finally to diverticular perforation (diverticulitis). Disease progression from the initial phase to the advanced phases does not necessarily occur. In this chapter, we discuss the natural history of this disease and focus on radiologic diagnosis and intervention.
Diverticulosis
Epidemiology
Diverticular disease is the most common colonic disease in the Western world. Interestingly, this disease was a pathologic curiosity before the 20th century. Its modern development is attributable to the introduction of the roller milling process in 1880, which removes most of the fiber from flour. This process results in a low-residue diet producing low-volume, tenacious stools that require a high degree of propulsive effort for expulsion.
In Western countries, colonic diverticula occur in 5% of the population by 40 years of age, in 33% to 50% of the population after 50 years of age, and in more than 50% of the population after 80 years of age. Among patients of routine risk, diverticulosis is found in 71% of patients on colonoscopy age 80 years or older. These findings contrast sharply to the prevalence rate of less than 0.2% in underdeveloped areas of Asia and Africa. In individuals from low-prevalence areas who migrate to Western communities, the frequency of diverticula increases within 10 years.
The sigmoid colon is involved in up to 95% of patients. Japan is an interesting exception to other developed countries in that patients with right-sided diverticulosis predominate at younger ages, subsequently becoming more bilateral over time. Of all patients who harbor diverticula, and 20% of those whose diverticula are clinically recognized, 4% to 5% will develop some complication, 1% to 2% will require hospitalization, and 0.5% will need surgical intervention, radiologic intervention, or both. In 2011, diverticulosis and diverticulitis were listed as the principal discharge diagnosis in 329,165 cases, accumulating over $11 billion in hospital charges in the United States. In-hospital deaths were listed as 1,858 cases (0.56%).
Causative Factors
The two fundamental factors contributing to the development of sigmoid diverticula are a pressure gradient between the lumen and serosa and areas of relative weakness in the bowel wall. The first factor can best be appreciated by considering the tendency of the colon to function not as a tube but as small compartments created by the haustra (segmentation). The sigmoid is the narrowest portion of the colon and generates the highest intrasegmental pressures, an example of the law of Laplace. In addition, stool is most dehydrated in the sigmoid colon, further increasing segmentation and motor activity.
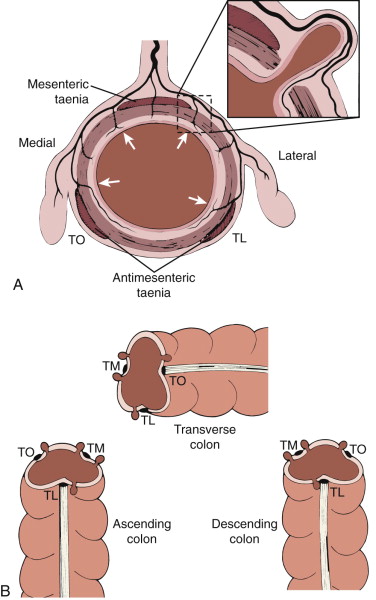
The colon wall is weakest where the intramural vasa recta penetrate to the submucosal layers. These points are on either side of the taenia mesocolica and on the mesenteric side of the taenia libera and taenia omentalis ( Fig. 55-1 ). Diverticula do not usually occur on the haustral row between the taenia omentalis and taenia libera.
Pathophysiology
Most colonic diverticula are false diverticula, or pseudodiverticula, because they contain mucosa and submucosa but not the muscularis propria ( Fig. 55-2 ). They are usually 0.5 to 1.0 cm in size and penetrate the clefts between bundles of circular muscle fibers at points where nutrient arteries pass through the submucosa. The proximity of the diverticula to the arteries is considered key in the development of hemorrhage.

In the uninflamed state, diverticula are elastic and compressible but have a tendency to empty poorly and, as a result, to fill with inspissated stool. This tendency may account for the increased number of lymphoid follicles found in the lining mucosa. Most patients with sigmoid diverticula have myochosis, a disorder in which there is thickening of the circular muscle layer, shortening of the taeniae, and narrowing of the lumen. The circular muscle is often corrugated, and its appearance has been likened to that of a concertina.
Diverticula of the rectum are rare, likely because the taeniae become fused anteriorly and posteriorly at this level, forming a completely encircling, supporting coat. Rectal diverticula are true diverticula and are usually lateral.
Clinical Findings
In most patients, diverticulosis is an incidental finding. Patients may complain of symptoms of irritable bowel syndrome, such as chronic or intermittent postprandial lower abdominal pain, which is relieved by defecation.
Radiologic Findings
Radiographic Studies
More than 50% of patients with significant diverticular disease demonstrate a distinctive bubbly appearance of the sigmoid colon on routine abdominal radiographs. When pelvic phleboliths are present, there is a statistical correlation between their number and size and the degree of diverticular disease. It is postulated that both these common disorders are caused by the long-term effects of a low-fiber diet and straining while defecating.
Barium Studies
Diverticulosis is detected more often by double-contrast studies than by single-contrast studies because of the greater colonic distention and improved visualization of intramural diverticula ( Fig. 55-3 ). When viewed en face, these immature diverticula may simulate aphthae. Their true nature is established when they are viewed in profile, when they are conical or triangular and 1 to 2 mm high.

The mature diverticulum has several characteristic appearances ( Fig. 55-4 ) that depend on the angle with which it is viewed and degree of barium filling. In profile, the diverticulum appears as a flasklike protrusion measuring several millimeters to several centimeters in length, joined to the wall by a fairly long, large neck. En face, it may appear as a ring shadow or a well-marginated barium collection or may resemble a bowler hat ( Fig. 55-5 ). The size and form of diverticula vary from one moment to the next, depending on the degree of colonic distention. The alignment of diverticula along the taeniae is usually apparent.


Diagnostic difficulties may arise if the diverticulum inverts into the lumen, and a number of diverticular polypectomies have been described in the endoscopic literature. Also, if feces obstruct the diverticulum, the diverticular orifice may bulge into the lumen, resulting in a ring shadow or filling defect. In profile, only the neck of the diverticulum may be visible. If the obstruction is incomplete, a fine linear strand of barium may penetrate the diverticulum, simulating a spur. Because this strand of barium appears outside the lumen, its presence usually allows the correct diagnosis to be made. For this reason, careful attention to the appearance of polyps and diverticula from film to film is necessary.
A diverticulum with a large neck may resemble a sessile polyp, one with a narrow neck may resemble a pedunculated polyp, and an obstructed diverticulum may resemble a polyp or adherent fecal material. In addition, both polyps and diverticula can produce the so-called bowler hat sign. If the bowler hat points toward the center of the long axis of the colon, it represents a polyp. If it points away from the center of the long axis of the bowel, it represents a diverticulum. If the bowler hat is located in the midline of the bowel, or is directly parallel to the long axis of the bowel, it cannot be confidently classified as representing a polyp or diverticulum.
Caliber and haustral abnormalities ( Fig. 55-6 ) usually accompany and often antedate the appearance of diverticula, particularly in the sigmoid colon. Haustral clefts are usually symmetric and face one another in the sigmoid; however, in diverticular or prediverticular disease, they indent alternately from one border to another, creating a staggered effect. The circular muscle is hypertonic and thickened, and the taeniae contract. Diverticula originate at the top of the haustra. With progressive disease, the circular muscle continues to undergo hypertrophy and a concertina or zigzag appearance develops. This appearance is caused by the alternated, highly compact, deep, and uniform haustra that separate and segment the haustral cul-de-sacs.

Computed Tomography
Computed tomography (CT; Fig. 55-7 ) reveals mural thickening of the colon (>4 mm) in myochosis and in most cases of diverticulosis. The diverticula appear as outpouchings that contain air, stool, and/or contrast agent.

Diverticulitis
Epidemiology
Diverticulitis is the most common complication of diverticulosis. Traditionally, this has been estimated to occur in 10% to 25% of patients with diverticulosis. The true incidence is likely less, with one study of Veterans Administration patients establishing an incidence closer to 4%. Simple, or uncomplicated diverticulitis, will occur in 75% of initial presentations. Complicated diverticulitis, defined as the presence of an abscess, fistula, obstruction, or perforation, is seen in the remaining 25%.
Pathophysiology
The stagnation of nonsterile, inspissated fecal material within the diverticulum can cause inflammatory erosion of the mucosal lining in early, subclinical stages of diverticulitis. Subsequently, the wall of the diverticulum is eroded. This perforation is the essential feature of diverticulitis ( Fig. 55-8A ).

This sequence of events can involve an intramural diverticulum, leading to the formation of an intramural abscess. Usually, it occurs extramurally within the pericolic fat, leading to fibrinous exudate, abscess formation, local adhesions, or peritonitis. During surgery and pathologic examination, many juxtacolic abscesses are small and difficult to find in the mass of indurated fat and fibrous tissue. It is important to emphasize that the inflammation begins at the apex of the diverticulum and may spread rapidly into the soft pericolic and mesocolic fat. The inflammatory changes in diverticulitis are therefore mainly pericolic, adjacent but outside the bowel wall.
The colonic mucosa is not affected in diverticulitis, except in the region of the erosion. Most inflammatory complications of clinical significance are secondary to the rupture of a diverticulum, and they occur in a pericolic location in almost all cases. Consequently, these pathologic changes may be better described as peridiverticulitis or pericolitis. True diverticulitis, with inflammation limited to the diverticular lining, was present in 10% of pathologic specimens, and muscular hypertrophy, with distortion alone and without inflammation, accounted for the remaining 25% of cases.
The consequences of diverticular rupture depend on the host response and virulence of the bacterial contamination. Most patients develop sealed-off abscesses or contained sinus tracts and fistulas. Free perforations are uncommon but can lead to localized pelvic or generalized peritonitis. Fistulas involve adjacent structures, such as the small bowel, urinary bladder, vagina, and anterior abdominal wall. Occasionally, several adjacent diverticula communicate with each other along the outer aspect of the deep muscular layers, forming an intramural fistulous tract. In other cases, fistulas extend deeply into the fatty tissue at the site of the attachment of the sigmoid mesocolon.
Clinical Findings
The classic clinical features of sigmoid diverticulitis are left lower quadrant pain, tenderness, fever, and leukocytosis. Because of this clinical constellation, sigmoid diverticulitis has been termed left-sided appendicitis . Diverticulitis occurring in other colonic locations is more difficult to diagnose clinically. In one review, the following clinical features were noted: pain in the left iliac fossa in 85% of patients; palpable mass, tenderness, and muscle guarding in 48%; fever in 30%; vomiting in less than 50%; and partial obstruction in 20%. Mild bleeding occurred in 25% of patients, and about 33% of patients had a history of recurrent episodes of similar attacks. Surgical evaluation of this group of 258 patients showed a 20% incidence of pericolic abscess, 8% incidence of fistulas, and 18% incidence of peritonitis. The initial clinical diagnosis was sigmoid carcinoma in 32% of patients, but carcinoma was found in only 2% of patients. The occurrence of malignancy was entirely coincidental, but it highlights the clinical difficulties of diagnosing and evaluating patients with suspected acute diverticulitis. These difficulties are further documented by large surgical series of patients with a clinical diagnosis of acute diverticulitis, in whom 25% to 33% of the resected surgical specimens showed no active inflammation, abscess, or fistulas.
Severe diverticulitis can occur in certain groups of patients and produce only minimal or unremarkable clinical symptoms, such as debilitated older patients, patients who have renal failure, are undergoing dialysis, or have had renal transplantation, and patients receiving corticosteroids. Disease in these patients may progress to free perforation, and the diagnosis is often made only when free intra-abdominal air is detected on abdominal films.
Patients younger than 40 years have historically been reported to have more severe forms of diverticulitis, ultimately requiring surgery. Some reports have suggested that younger patients follow the same course as older patients, and the decision for surgery should be made similarly.
The clinical management of patients with acute diverticulitis depends on the severity, type, and extent of the pericolic inflammatory changes. Mild forms of disease are managed medically with antibiotic therapy. A large recent Swedish study, however, concluded that mild acute uncomplicated diverticulitis may be successfully treated with anti-inflammatory agents, with no additional benefit from antibiotics. In more severe forms, surgical resection may be indicated, either at the time of the diagnosis or after a cooling-off interval of antibiotic therapy and percutaneous abscess drainage. One study demonstrated emergent surgical intervention in 12% of patients admitted for diverticulitis. This has decreased over time, partly because of increased use of image-guided percutaneous drainages. Emergent surgery is reserved for Hinchey 3 or 4 acute diverticulitis.
Close clinical and radiologic evaluation are crucial for the triage of patients to conservative, radiologic, or surgical management. The radiologic investigation of these patients has two objectives—to confirm the clinical suspicion of diverticulitis (and rule out other colonic or pelvic disease) and evaluate and stage the severity of the inflammatory disease. Sigmoidoscopy is usually contraindicated and serves no useful purpose except in patients who have more chronic forms of disease and present with bleeding, or in patients in whom polyps or sigmoid carcinoma is suspected.
Radiologic Findings
Radiographic Studies
Abdominal radiographs taken with the patient supine and upright are usually diagnostic only in the most severe forms of diverticulitis, in which the patient presents with free intraperitoneal air or sealed-off perforations and pelvic extraluminal air. The detection of an ill-defined, left-sided pelvic mass, localized ileus, and fluid in the pelvis suggests the diagnosis in the appropriate clinical setting. In most patients with acute diverticulitis, however, abdominal plain films are unremarkable and do not contribute to the diagnosis.
Contrast Enema Examination
Contrast enema (CE) examination had traditionally been the primary method of examining patients with suspected diverticulitis, but now has been replaced by CT as the initial diagnostic test. It superbly depicts diverticula, colonic mucosa and lumen, spasm, muscle hypertrophy, and sacculations. These findings, however, are indicative of diverticular disease but not diagnostic of acute diverticulitis. Because this procedure primarily evaluates the colonic lumen and mucosal surface, the pericolic inflammatory process present in most patients with diverticulitis can only be inferred ( Fig. 55-8B ).
A specific diagnosis of perforated diverticulitis can be made only when there is extravasation of contrast material from a diverticulum into a walled-off abscess, sinus tract, or fistula or free extravasation of contrast material into the peritoneal cavity. These extraluminal collections of contrast medium vary in size, are usually located adjacent to the colon, compress and displace the colonic wall, and are detected in less than 50% of cases of diverticular perforation. Narrowing of the sigmoid lumen, extrinsic compression, and spasm are present in more than 80% of patients, but these findings are less specific. Similarly, an altered mucosal pattern is present in 68% of patients but is not a reliable indicator of acute inflammation. Lack of mobility with fixation of the sigmoid colon and narrowing, pointing, and distortion of individual diverticula are indirect signs consistent with acute or chronic diverticulitis.
A contour defect that is smooth, well defined, and associated with adjacent sigmoid diverticula represents an intramural inflammatory mass. The contents of the intramural mass, whether pus, organized abscess, or merely mural fibrosis, are more difficult to characterize. Tracts filled with contrast material passing from ruptured diverticula into the pericolic tissues are common ( Fig. 55-9 ). They may be single or multiple, end blindly (sinuses), or connect with an adjacent hollow viscus or abdominal wall (fistulas). The most common fistulas are colovesical and coloenteric. Colocutaneous fistulas are less frequent and are clinically associated with subcutaneous abscess, emphysema, or fasciitis. Fistulas to the vagina, ureter, appendix, hip, perineum, and soft tissues of the thigh have also been reported. With CE, the fistulous tract between the colon and urinary bladder is visualized in only 20% of patients who have colovesical fistulas.

Longitudinal intramural fistulous tracts represent ruptured diverticula that communicate with each other. Although this sign has also been reported in carcinoma of the colon and Crohn’s disease, longitudinal intramural fistulous tracts most often are caused by diverticulitis unless there is a previous history or radiographic evidence of Crohn’s disease in the remainder of the colon or terminal ileum.
Ultrasonography
Although multidetector CT (MDCT) is the primary cross-sectional imaging study for patients with suspected diverticulitis, ultrasonography (US) may be the first study ordered for nondescript abdominal pain and for pregnant patients suspected of having diverticulitis. The advantages of ultrasound are the lack of ionizing radiation and relative lower cost. Ultrasound is practically and technically more difficult in obese and severely ill patients, and is operator-dependent. US should be performed with a graded compression technique. One meta-analysis has demonstrated no significant difference in the accuracy of graded compression US and CT in diagnosing diverticulitis, with CT more likely to find alternative causes of abdominal pain. Additionally, CT has been found to be more sensitive in the evaluation of complicated diverticular disease, better demonstrating abscess and free air.
The segmental concentric thickening of the gut wall commonly found in patients with diverticular disease manifests as a hypoechoic segment that reflects the predominant thickening of the muscular layer ( Fig. 55-10 ). Sonographic findings of diverticulitis include segmental wall thickening more than 4 mm, echogenic adjacent pericolic fat that is noncompressible, and inflamed diverticula. Local pain on compression is also a typical finding. Inflamed diverticula are brightly echogenic reflectors with acoustic shadowing or a ring-down artifact in or beyond the thickened gut wall. There may be adjacent fluid. Intramural sinus tracts appear as high-amplitude linear echoes that often have a ring-down artifact within the colon wall. A fecalith will be echogenic, with posterior acoustic shadowing. Abscesses appear as loculated, thick-walled fluid collections that may contain gas.

Magnetic Resonance Imaging
Mural thickening of the colon ( Fig. 55-11 ) and diverticular abscesses are well visualized on magnetic resonance imaging (MRI), particularly on gadolinium-enhanced, fat-suppressed, T1-weighted spoiled gradient-echo images and T2-weighted, single-shot echo-train spin-echo images. Inflammatory change within the adjacent fat can be seen on non–fat-suppressed and fat-suppressed images. Sinus tracts, fistulas, and the walls of abscesses enhance and are well depicted in a background of suppressed fat on gadolinium-enhanced, fat-suppressed, spoiled gradient-echo images.

The major advantage of MRI is the lack of ionizing radiation. The disadvantages of MRI are longer scan times, greater susceptibility to motion artifact from the adjacent bowel, and relative higher cost. Given the efficiency and effectiveness of CT in the assessment of diverticulitis and its complications, MRI has little role in the primary evaluation of routine patients with known or suspected diverticulitis. MRI could be considered in special situations, such as pregnant patients, because of the lack of ionizing radiation.
Computed Tomography
MDCT has dramatically improved the diagnosis and management of patients with diverticulitis. MDCT is ideally suited for evaluating the intramural component of the inflammatory process and its intraperitoneal or retroperitoneal extension. CT is particularly useful in the evaluation of patients with sepsis who present with left lower quadrant pain, fever, leukocytosis, and a tender, palpable pelvic mass. CT has a reported accuracy, specificity, and sensitivity of up to 99%. Current guidelines recommend a CT with intravenous (IV) contrast as the initial test for patients with suspected diverticulitis. Oral contrast is not practically necessary, but may help in the evaluation of fistulas and with bowel distention.
CT findings in acute diverticulitis are listed in Table 55-1 . On non–contrast-enhanced scans, the offending diverticulum can often be identified. It is hyperdense and is the nidus of the surrounding inflammation ( Fig. 55-12 ). Diverticula are identified on CT at the site of the perforation or at a site adjacent to it in more than 80% of cases of acute diverticulitis. They appear as small outpouchings filled with air, barium, and/or fecal material projecting through the colonic wall. Symmetric thickening of the colonic wall in excess of 4 mm is seen in about 70% of cases. The thickened wall has a homogeneous density, and its diameter usually measures less than 1 cm in the distended colon. When there is significant muscular hypertrophy, the wall of the colon can be up to 2 or 3 cm thick. In diverticulitis, significant hypertrophy of muscle primarily occurs in the sigmoid segment, which can mimic carcinoma of the colon. Accurate assessment of colon wall thickness is possible only with proper lumen distention.
| Finding | No. of Cases (%) |
|---|---|
| Mural thickening of the colon | 96 |
| Fat stranding | 95 |
| Adjacent diverticula | 91 |
| Fascial thickening | 50 |
| Visualization of inflamed diverticulum | 43 |
| Pericolic gas bubbles | 30 |
| Arrowhead sign | 16 |
| Abscess | 4 |
| Phelgmon | 4 |
| Intramural gas | 2 |
| Sinus tracts | 2 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








