33 Duplex Ultrasound Evaluation of the Male Genitalia
The Scrotum
Anatomy and Normal Sonograpic Features
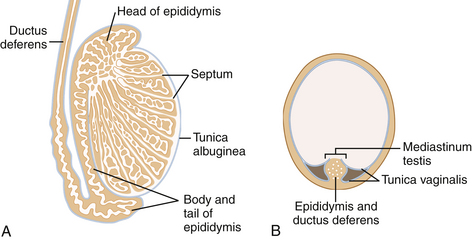
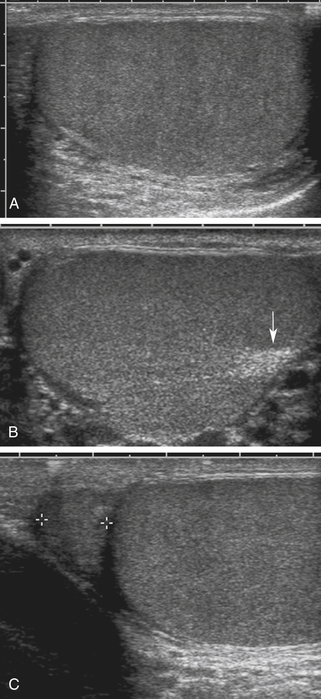
The anatomy of the scrotum, testis, and epididymis is illustrated in Figures 33-1 and 33-2. As seen with ultrasound, the normal testis is homogeneous and medium in echogenicity (Figure 33-3), with a smooth outer border but no visible capsule.1–9 In adults, each testis measures 3 to 5 cm in long axis and 2 to 3 cm in short axis. The testes are relatively hypoechoic before the age of puberty and increase in echogenicity with adulthood . The mediastinum testis is seen regularly as a strongly echogenic band running along one side of the testis. The epididymis is similar to or slightly less echogenic than the testis. Its echotexture may be somewhat heterogeneous.
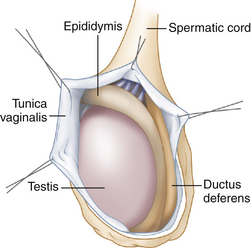
FIGURE 33-1 Scrotal anatomy. Each testis and epididymis is suspended in a sac lined by the tunica vaginalis.
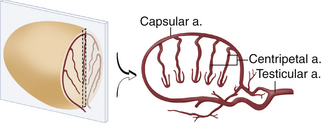
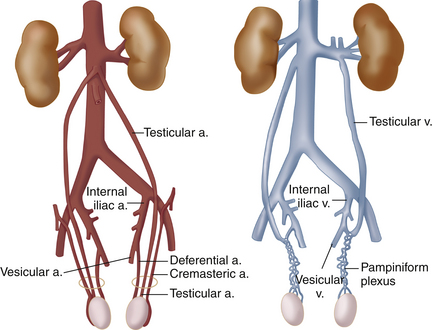
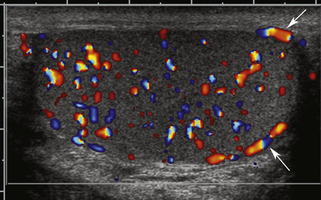
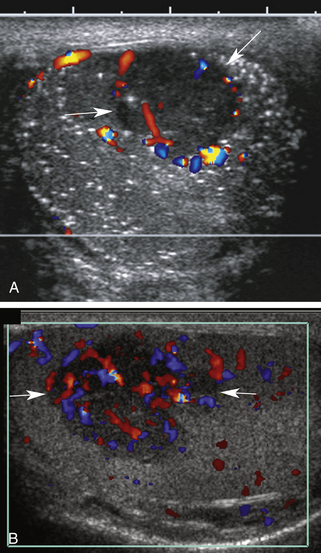
The arterial and venous anatomy of the testes is illustrated in Figures 33-4 and 33-5. In postpubertal boys and adults, blood vessels4–7,10,11 are normally visible in and about the testis with color flow sonography (Figure 33-6). The capsular arteries, which course around the periphery, and the centripetal arteries, which penetrate the parenchyma, are commonly seen. Blood flow in the centripetal arteries is from the capsule inward. Testicular veins follow the same pattern as the arteries and generally are readily visualized. Differentiation between arteries and veins is possible only with spectral Doppler. In some normal individuals, one or more large artery/vein pair(s) may traverse the testis obliquely from the mediastinum to the opposite capsule.4,5 These “transmediastinal” vessels may be visible on gray-scale imaging and should not be mistaken for pathology.
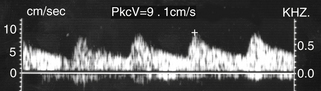
Arterial blood flow in the testis and epididymis characteristically exhibits a low-resistance pattern on Doppler spectral waveforms, including continuous flow during diastole (Figure 33-7). In contrast, a high-resistance flow pattern is seen in extragonadal arteries, which are part of the cremasteric system. These arteries are occasionally visualized along the course of the spermatic cord. It is important not to mistake extragonadal flow signals for testicular flow. Peak systolic velocity in testicular arteries ranges from 4 cm/sec to 19 cm/sec (mean, 9.7 cm/sec), and end-diastolic velocity, from 1.6 cm/sec to 6.9 cm/sec (mean, 3.6 cm/sec).11 These values permit quantitative assessment of arterial flow when a sufficiently long arterial segment is visualized with color flow, allowing for angle correction of the Doppler signal. When angle correction is not possible, spectral Doppler features are evaluated qualitatively.
Sonographic Technique
The first step in scanning the scrotal contents is to get oriented. Long- and short-axis images of each testis and epididymis, including long- and short-axis testis dimensions, should be recorded. A composite transverse view showing both testicles simultaneously is also obtained, as this view is essential for comparing testicular echogenicity and testicular vascularity. If both testes cannot be viewed simultaneously on a transverse view, separate images should be recorded side by side, using identical ultrasound settings. When pathologic findings are present, they should be portrayed in whatever image plane best documents the abnormality, but long- and short-axis views should be used whenever possible as an aid to orientation.
Scrotal Masses
Testicular Cysts
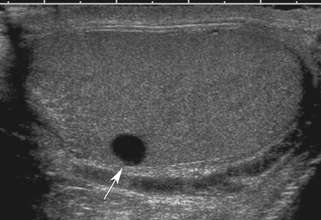
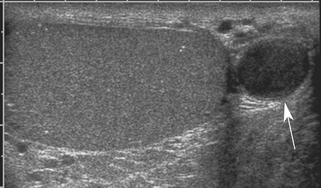
Cysts of the testis* are idiopathic and benign. They also are fairly common, seen with increasing frequency with increasing age and present in approximately 8% of adults at sonography. Most intratesticular cysts are located near the mediastinum testis and are not palpable. They are typically small, measuring less than 1 cm in diameter, and are thought to arise from the rete testes, the convergence of intratesticular tubules at the mediastinum. These cysts may be single or multiple. Sometimes, in the presence or absence of discrete cysts, a region of dilated tubules is seen in the vicinity of the mediastinum, representing a dilated rete testis.13 Cysts located on the testicular surface are almost always tunical cysts, arising in the tunica albuginea, the fibrous layer that encapsulates the testis. Tunical cysts may be palpable, prompting ultrasound investigation.
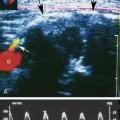
The most important point about testicular cysts and a dilated rete testis is distinguishing these benign lesions from other pathologies, including tumors and abscesses. Testicular cysts (Figure 33-8) have the following sonographic features: (1) anechoic contents, (2) sharply defined borders and invisible wall, (3) enhanced through-transmission of ultrasound, and (4) no blood flow within or surrounding the cyst (other than normal testicular vessels). Cysts meeting these criteria are benign and inconsequential and require no follow-up. A dilated rete testis appears as small serpiginous tubular structures clustered in the mediastinum (Figure 33-9).
Testicular Neoplasms
Testicular neoplasms are most often primary testicular tumors of germ cell origin (Table 33-1).1,2,14 These neoplasms occur most frequently between the ages of 25 and 35 years and almost always are malignant. The prognosis generally is excellent, however, and 5-year survival is 95%, overall, assuming timely treatment with surgery, radiation therapy, and/or chemotherapy.15 Less common testicular neoplasms arise from the stromal parenchyma and are either Sertoli or Leydig cell tumors. Rarely, nontesticular malignancies involve the testicle, including leukemia and lymphoma and metastatic disease. Testicular tumors usually present in one of two ways: as a palpable mass or with sudden pain and swelling due to hemorrhage. It is not uncommon for the latter presentation to follow minor trauma. Tumors may also present with symptoms of epididymitis. A small number of patients present with signs and symptoms resulting from metastatic testicular cancer, such as back pain from retroperitoneal disease.
TABLE 33-1 Classification of Testicular Malignant Neoplasms
| Testicular malignancies of germ cell origin (95%) Other primary testicular malignancies |
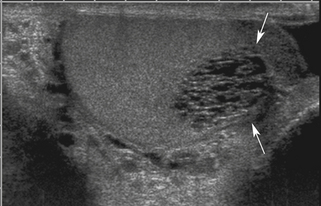
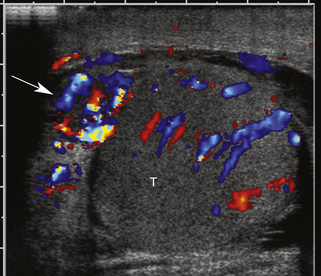
Ultrasound can distinguish intratesticular from extratesticular pathology with extremely high accuracy.1 Nevertheless, ultrasound generally cannot differentiate among histologic types of testicular tumors, nor can it generally differentiate between malignant (common) and benign (uncommon) neoplasms. Most testicular tumors are well-defined hypoechoic intratesticular masses, but some may be poorly marginated or grossly infiltrating.† They may exhibit some degree of internal heterogeneity due to hemorrhage and/or necrosis, and calcification is occasionally present. Vascularity is evident within testicular neoplasms on color Doppler images and is a very important feature used to distinguish tumors from avascular structures such as cysts, hematomas, and abscesses. Testicular tumor vascularity (Figure 33-10) is variable; however, most malignant tumors are hypervascular compared to the surrounding normal testicular parenchyma.4,9–11,14,17,18 The distribution of tumor blood vessels also is variable, with some lesions showing an orderly distribution of blood vessels and others, a chaotic distribution. Large avascular areas may be present in a testicular tumor, when necrosis or hemorrhage is present. Spectral Doppler generally shows low-resistance flow within tumor vessels, which is typical of malignant neoplasms regardless of location. Flow velocities may be elevated substantially in markedly hypervascular tumors, but flow may be in the normal range in mildly hypervascular lesions or lesions with less flow. In general, the larger the testicular tumor, the more hypervascular it will be.11
Testicular microlithiasis, scattered small calcifications in the testicular parenchyma as shown in Figure 33-10, A, has been associated with increased risk for testicular cancer in some series, but the level of risk and follow-up requirements remain controversial.17,19–21
Testicular Tumor Mimics
Lesions that can mimic the appearance of neoplasms include abscesses, inflamed areas or focal orchitis (without frank abscess formation), contusions, hematomas, and infarcts.1,7,9,14,22–24 The sonographic appearance of these lesions is nonspecific, as discussed later. Color and spectral Doppler features are of considerable importance in differentiating among these etiologies. Although blood flow is absent within abscesses, infarcts, and hematomas, blood flow signals can be detected at the periphery of abscesses.
Epididymal Cysts
Epididymal cysts1,2,7,9,14 are much more common than testicular cysts, being found in about 40% of adult males examined sonographically.1 Most are located in the epididymal head, but cysts may occur anywhere in the epididymis. They may be single, multiple, unilateral, or bilateral. In some cases they are palpable, while in others they are found incidentally. Unlike testicular cysts, epididymal cysts may be septated or even multilocular. Most are 2 to 3 mm in diameter, but larger cysts are also common, and occasionally they may be several centimeters in size. The etiology of epididymal cysts is not entirely clear, although some are spermatoceles, which are encapsulated collections of sperm.
The great majority of epididymal cysts have the same sonographic features as those described previously for testicular cysts, but there are other variations. Some may be septated or multilocular, and others may contain diffuse or dependent low-level echoes (Figure 33-11). When evaluating epididymal cysts sonographically, it is most important to document that benign epididymal cysts have no solid components, have exquisitely thin walls, and show no internal blood flow on color Doppler examination.
Other Epididymal Masses
Other than cysts, the only common mass lesions of the epididymis are hematomas, abscesses, and inflammatory masses.‡ Tumors of the epididymis are uncommon and usually benign. Their sonographic features are not well described in the medical literature. An important characteristic, however, is the presence of blood flow within the tumor, which is not present in hematomas or abscesses. Hematomas of the epididymis or spermatic cord usually occur in the context of recognized trauma but may occur spontaneously and in association with vigorous exercise. An epididymal hematoma usually presents as a hard, palpable (and possibly tender) mass that may mimic a neoplastic mass on physical examination. On ultrasound, a hematoma usually has a nonspecific, hypoechoic, or heterogeneous appearance. Most importantly, blood flow is absent in and around the lesion (except for normal vessels) on color flow examination. Abscesses and inflammatory masses are differential considerations and are discussed later.
Epididymitis and Orchitis
Infection is the most common cause of acute scrotal pain and tenderness.14 In the great majority of cases, the infection§ is caused by sexually transmitted organisms (principally Neisseria gonorrhoeae and Chlamydia trachomatis) that “ascend” through the genital tract. The tail of the epididymis is infected first, and then the infection spreads throughout the epididymis (epididymitis). The infection may then extend to the testis (orchitis) and finally to the scrotal cavity, generating an infected hydrocele.9
Ultrasound is a very useful method for confirming the diagnosis of epididymitis or orchitis and for excluding other pathologies that may cause acute scrotal pain or swelling. The key findings are enlargement and decreased echogenicity of the affected structures, accompanied by increased blood flow (hyperemia) on color and spectral Doppler examination (Figure 33-12). Because orchitis may be present by the time the patient seeks care, involvement of both the testis and epididymis may be evident sonographically; however, in some patients, epididymal involvement may predominate. A hydrocele (excess scrotal fluid) often is present, and the scrotal wall may become edematous and/or inflamed. In some cases of epididymitis/orchitis, the ultrasound findings may be dramatic. In other instances, findings are less obvious and are based solely on side-by-side comparison of epididymal size, echogenicity, and blood flow. Obviously, side-by-side comparison is not helpful in cases of symmetrical, bilateral infection; nevertheless, hyperemia of the scrotal contents may still be evident in such cases. It is noteworthy that a focal, hypoechoic area of inflammation may be seen in the periphery of the testis, adjacent to an infected epididymal head.28 This should not be mistaken for a testicular tumor but should be followed to resolution with antibiotic treatment.
(Figure 33-12). Because orchitis may be present by the time the patient seeks care, involvement of both the testis and epididymis may be evident sonographically; however, in some patients, epididymal involvement may predominate. A hydrocele (excess scrotal fluid) often is present, and the scrotal wall may become edematous and/or inflamed. In some cases of epididymitis/orchitis, the ultrasound findings may be dramatic. In other instances, findings are less obvious and are based solely on side-by-side comparison of epididymal size, echogenicity, and blood flow. Obviously, side-by-side comparison is not helpful in cases of symmetrical, bilateral infection; nevertheless, hyperemia of the scrotal contents may still be evident in such cases. It is noteworthy that a focal, hypoechoic area of inflammation may be seen in the periphery of the testis, adjacent to an infected epididymal head.28 This should not be mistaken for a testicular tumor but should be followed to resolution with antibiotic treatment.
The sonographic diagnosis of epididymo-orchitis generally is straightforward, but mimics occasionally may cause misdiagnoses; namely, hyperemia following detorsion of a testis (considered later) and diffusely infiltrating lymphoma or leukemia.29,30 The epididymis and testis usually return to a normal sonographic appearance following an episode of infection, but in severe cases, testicular atrophy or infarction may occur. Atrophy is a noteworthy complication of mumps orchitis.
Varicocele
Varicocele, or dilatation of the pampiniform venous plexus, is a common cause of a palpable epididymal mass and scrotal discomfort.¶ In some individuals, a varicocele contributes to low sperm count, decreased sperm motility, and infertility. These problems have been attributed to persistent, hyperemia-induced elevation of testicular temperature, but the real cause may be more complex and is not known with certainty.
The veins of the pampiniform plexus drain the testis and epididymis and are normally quite small, but may dilate because of valvular incompetence and/or elevated pressure, forming a tangle of enlarged veins along the course of the spermatic cord and epididymis. In unusual cases, dilated veins may even extend into the substance of the testis.33 Varicoceles are more common on the left side of the scrotum than the right, possibly due to elevated pressure in the left testicular vein. The left testicular vein inserts into the left renal vein, which drapes across the aorta and may be compressed between the aorta and superior mesenteric artery, raising venous pressure. The right testicular vein drains into the inferior vena cava directly and is not subject to compressive effects. Because of the left-side predominance of varicocele, the possibility of neoplastic testicular vein obstruction or compression (due to intra-abdominal lymphadenopathy) should be considered when a varicocele is isolated to the right cord/epididymis.
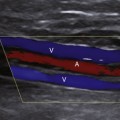
Varicocele usually is a clinical diagnosis, as the tangle of veins is easily palpated and feels like a “bunch of worms.” Ultrasound is required when the nature of the palpable mass is unclear or when pain or tenderness is present, as well as in men who experience infertility. Varicocele is diagnosed with color flow ultrasound when numerous veins of unusually large size are seen along the spermatic cord or epididymis, as shown in Figure 33-13. In all cases, the extent of the varicosities should be documented, and the largest veins should be measured, as discussed later. The presence of reflux within the veins can be investigated by having the patient perform the Valsalva maneuver during color flow observation. Normal pampiniform veins are barely detectable, so from one perspective, a varicocele can be diagnosed whenever veins of unusual size and number are readily seen. More specific diagnosis may be important, however, in men with pain or infertility, in whom a decision must be made concerning the potential benefit of therapy. In this respect, veins 2 mm in diameter or less are generally regarded as not substantially dilated, while larger veins are considered varicose, especially those with a diameter of 3 mm or larger.2,
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree