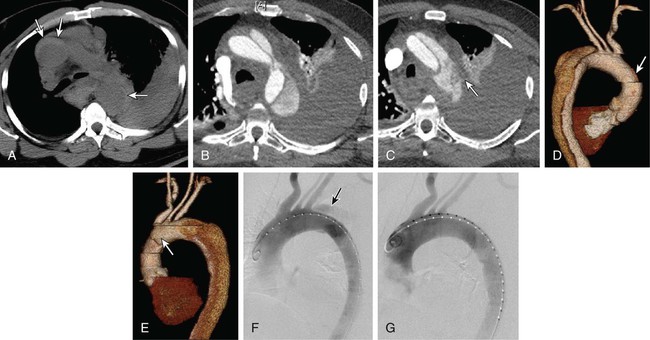
Appropriate treatment for an aortic dissection is determined by a combination of location and extent of the dissection, time from onset of symptoms, comorbid risk factors, and dissection-related conditions such as visceral malperfusion or impending rupture. Although most patients present with acute dissection, up to a third present with symptoms of more than 2 weeks’ duration.1 This distinction between acute and chronic dissection is of therapeutic significance because patients with acute aortic dissections are at highest risk for life-threatening complications. In the current era, treatment options can be divided into three main categories: medical management, surgical intervention, and endovascular intervention. Recommendations for the management of thoracic aortic dissection have been made by the 2010 American College of Cardiology Foundation/American Heart Association (ACCF/AHA) task force for the diagnosis and management of patients with thoracic aortic disease.2 Ascending aortic dissections (Stanford type A, DeBakey I, II) account for about 60% of all dissections3 and are at high risk of acute complications including cardiac tamponade (5%), myocardial ischemia (7%-19%), acute aortic regurgitation (45%), heart failure (5%), stroke (8%), and ultimately death.4 The mortality rate for acute type A dissections without intervention is estimated at 1% per hour after patients arrive in the hospital, with mortality rates of 38%, 50%, 70%, and 90% at 24 hours, 48 hours, 1 week, and 2 weeks, respectively.5,6 In general, type A dissections require emergent surgical repair. Presently, there are no U.S. Food and Drug Administration (FDA)-approved endovascular devices for the ascending aorta.2 There remains some controversy over the treatment strategy of type B aortic dissections. In 1965, DeBakey et al. published a large series of 179 aortic dissection patients treated surgically, with an operative mortality of 21% and a 5-year mortality of 50%.7 In comparison to large series of patients with aortic dissections who had undergone nonoperative treatment, DeBakey et al. concluded that all patients with aortic dissections should undergo surgical intervention.7,8 Later, Wheat et al.9 advocated a selective approach based on the observation that surgical intervention carried a 25% early mortality, whereas patients treated pharmacologically had a 16% early mortality.10 It was not until 1970 when Daily et al.11 at Stanford introduced the Stanford dissection classification that the importance of distinguishing ascending and descending aortic dissections became apparent. It was widely held that type B dissections ought to be treated medically unless life-threatening complications were present. Though operative mortality for type B dissections significantly improved over time (57%-13% in the Stanford series),10 several risk factors including visceral ischemia, aortic rupture, and older age portended dramatically increased risk with surgical repair.8,12 In the current era of endovascular stent-grafts, it seems intuitive that less invasive techniques for stabilizing complicated acute type B dissections, which carry a high risk of mortality with and without surgical intervention, could provide an opportunity to improve outcomes in this very ill patient population. Although 20% to 50% of uncomplicated type B aortic dissections demonstrate eventual aneurysmal dilatation within 3 to 5 years of diagnosis,13,14 there is a small risk of rupture in the acute setting, with a 30-day mortality of about 10% in contemporary studies. Most authors advocate medical treatment with blood pressure control and close follow-up with interval imaging.15 Many of these patients will ultimately require intervention beyond the acute setting, so some authors have suggested that asymptomatic descending aortic dissections be treated with stent-grafts to prevent late complications.16 However, long-term data comparing medical treatment and stent-grafts in uncomplicated acute type B dissections are insufficient at present to define the appropriate treatment strategy. The INSTEAD trial (Investigation of Stent-Grafts in Aortic Dissection) attempted to answer a similar debate in subacute and chronic type B dissections (within 2-52 weeks of onset) by randomizing patients to either medical therapy or stent-graft placement.17 The trial found no difference in all-cause and aorta-related mortality at 1-year follow-up, but 91% of those in the stent-graft group demonstrated evidence of aortic remodeling, compared to only 19% of those in the medical group.17 Even though the INSTEAD trial did not include acute aortic dissections (<2 weeks from onset), the trial did reveal a potential for stent-grafts to promote aortic remodeling and potentially prevent late aneurysmal degeneration. However, it is important to note that based on this trial, it appears that stent-graft treatment of patients with subacute or chronic aortic dissection offers no benefit in terms of reducing the risk of aortic rupture or enhancing life expectancy. Longer follow-up is necessary to understand the true benefit or lack thereof for stent-grafts in asymptomatic type B dissections. In contradistinction, complicated type B aortic dissections, defined by the presence of visceral or peripheral malperfusion, rupture, rapid false lumen expansion, persistent pain, or uncontrollable hypertension, often require intervention. Despite significant improvement in operative mortality for acute type B dissections, operative mortality in the presence of visceral ischemia and rupture remains as high as 70% to 80%.8,18 Addition of endovascular interventions has provided an opportunity for a less invasive, more expeditious procedure to stabilize the dissection, prevent rupture, and restore true lumen perfusion. Endovascular treatment options include aortic stent-graft placement, dissection flap fenestration, and branch vessel stenting.19 Each technique aims to achieve one or both of the two major goals in treating acute aortic dissection: prevent aortic rupture and restore end-organ perfusion. Current indications for intervention encompass the life-threatening complications of acute aortic dissection, hence the term “complicated.” As mentioned previously, complicated dissections are defined by visceral or limb malperfusion, aortic rupture or impending rupture (e.g., rapid false lumen expansion), or evidence of an unstable dissection plane (e.g., persistent pain, uncontrollable hypertension). In type A dissection, aortic branch vessel obstruction is usually corrected concomitantly with surgical management and repair of the ascending aorta.20 In type B dissection, branch vessel malperfusion is an indication for intervention. Recently, stent-graft placement over the primary entry tear has been increasingly performed as an alternative to open surgical distal fenestration of the intimal flap, percutaneous radiologic balloon fenestration of the aortic septum, operative replacement of the diseased aorta, or bypass graft reperfusion of ischemic vessels.21 The concept of endovascular stent-graft repair is predicated on successful device placement over the primary entry tear to obliterate blood flow into the aortic false lumen (Fig. 51-1). The intent is to mimic the effect of operative repair by isolating the false lumen from the circulation and redirecting all blood flow into the true lumen. As demonstrated in experimental models of dissection, coverage of the primary entry tear is the optimal method of relieving true lumen collapse, restoring perfusion to branch arteries off the true lumen, and promoting thrombosis of the aortic false lumen.22 Interestingly, dissections with a completely thrombosed aortic false lumen are associated with a favorable prognosis.23–25 In contrast, false lumen patency contributes to progressive aortic dilation and is a predictor of late mortality.26
Endovascular Treatment of Dissection of the Aorta and Its Branches
Indications for Intervention

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Endovascular Treatment of Dissection of the Aorta and Its Branches