Atherosclerosis is a complex inflammatory process and an integral component of myocardial infarction and stroke. Atherosclerotic plaques can be detected using ultrasonography, myocardial perfusion imaging, coronary angiography, multidetector computed tomography (CT), and MR imaging. These modalities assess the luminal encroachment of the plaques or the structural features. Imaging plaque biology in concert with plaque structure may provide important insights. PET scanning using 18 F fluorodeoxyglucose. ( 18 F FDG-PET) is commonly combined with CT scanning to characterize oncological processes. This review examines the role of 18 F FDG-PET/CT imaging in the characterization of atherosclerotic plaque biology.
Key points
- •
PET with 18 F fluorodeoxyglucose. ( 18 F FDG)-PET/computed tomography (CT) is a noninvasive imaging modality that can quantify carotid inflammation.
- •
Carotid 18 F FDG uptake correlates with inflammation, predicts plaque progression and subsequent cerebrovascular events.
- •
18 F FDG-PET/CT can be used to evaluate the impact of therapies on arterial inflammation.
- •
Future studies should determine if carotid 18 F FDG-PET/CT imaging can be used to risk stratify individuals with stenotic carotid disease to decide whether to pursue optimized medical therapy or revascularization.
Introduction
Atherosclerosis, a complex inflammatory process, is an integral component of myocardial infarction and stroke and thus is implicated as the most common cause of death in developed countries. The presence of atherosclerotic plaques can be detected using several well-established techniques such as ultrasonography, myocardial perfusion imaging, coronary angiography, multidetector computed tomography (CT), and MR imaging. These modalities assess the luminal encroachment of the plaques or the structural features and do not directly report on the biological processes present within the atheroma. Because atherosclerotic disease is driven by a dynamic biological process (with inflammation as a key component) imaging plaque biology in concert with plaque structure may provide important insights. PET scanning using 18 F Fluorodeoxyglucose. ( 18 F FDG-PET) is a molecular imaging modality that is commonly combined with CT scanning to characterize oncological processes. 18 F FDG-PET imaging has been well-demonstrated to greatly improve the characterization of tumors and has transformed the imaging approach in cancer. This review examines the role of 18 F FDG-PET/CT imaging in the characterization of atherosclerotic plaque biology.
Introduction
Atherosclerosis, a complex inflammatory process, is an integral component of myocardial infarction and stroke and thus is implicated as the most common cause of death in developed countries. The presence of atherosclerotic plaques can be detected using several well-established techniques such as ultrasonography, myocardial perfusion imaging, coronary angiography, multidetector computed tomography (CT), and MR imaging. These modalities assess the luminal encroachment of the plaques or the structural features and do not directly report on the biological processes present within the atheroma. Because atherosclerotic disease is driven by a dynamic biological process (with inflammation as a key component) imaging plaque biology in concert with plaque structure may provide important insights. PET scanning using 18 F Fluorodeoxyglucose. ( 18 F FDG-PET) is a molecular imaging modality that is commonly combined with CT scanning to characterize oncological processes. 18 F FDG-PET imaging has been well-demonstrated to greatly improve the characterization of tumors and has transformed the imaging approach in cancer. This review examines the role of 18 F FDG-PET/CT imaging in the characterization of atherosclerotic plaque biology.
Pathogenesis of atherosclerosis
Atherosclerosis, although previously considered an indolent lipid storage disorder, is now well-recognized to be an active inflammatory condition. Inflammation seems to play a role in all phases of plaque development, from initiation, to plaque progression, and eventually to plaque rupture. One of the initial steps in atherosclerotic plaque development is endothelial dysfunction. Several cardiovascular risk factors are associated with heightened expression of selective adhesion molecules (vascular cell adhesion molecule-1) on the endothelial cell surface, which prompts leukocytes to bind to the arterial wall. Once monocytes attach to the wall, they migrate to the intima through diapedesis, which is stimulated by chemoattractant molecules such as monocyte chemoattractant protein-1. Once monocytes invade the arterial intima, they differentiate into proinflammatory macrophages ( Fig. 1 A, E ). There, they release inflammatory mediators, which attract additional inflammatory cells to the developing plaque, causing a vicious cycle of inflammation.
Plaque inflammation stimulates progression and growth of the atheroma and prompts the development of several features that renders the plaque more prone to rupture. First, macrophages contribute importantly to the development and enlargement of the lipid-rich necrotic core. Macrophages engulf accumulated oxidized low-density lipoprotein (LDL) molecules and turn into foam cells. Eventually, foam cells undergo apoptosis and apoptotic bodies accumulate within the core of atheromatous plaque, leading to expansion of the lipid core. Additionally, macrophages release proteolytic enzymes such as matrix metalloproteinases, which thin and weaken the fibrous cap, rendering it more susceptible to rupture. The proteolytic milieu also participates in the positive remodeling of the atheroma, another feature of rupture-prone plaques. As the inflammatory condition continues over decades, plaques progression continues, and often results in clinical manifestations owing to luminal narrowing (either from gradual encroachment or from or sudden plaque rupture).
18 F Fluorodeoxyglucose uptake as a marker of tissue inflammation
18 F FDG, an analog of glucose, enters the cells via glucose transporter protein (GLUT) and is subsequently phosphorylated to become 18 F FDG-6-phosphate by hexokinase, a glycolytic enzyme. However, in contrast to glucose-6-phosphate, 18 F FDG-6-phosphate cannot be further metabolized through the glycolytic pathway; thus, it becomes trapped inside the cell and accumulates at a rate that is proportionate to the cell’s glycolytic rate. This link to glycolysis facilitates the identification of inflamed tissues. Inflammatory cells (such as macrophages) have a relatively high rate of glycolysis at rest, relative to other tissues, and increase their glycolytic rate further upon proinflammatory stimulation. Additionally, recent studies also show a tight relationship between 18 F FDG uptake and macrophage (MØ) activation in an atheromatous environment. Uptake of radiolabeled 2-deoxyglucose by human MØ in vitro is increased by cytokine activation and by oxidized LDL. Moreover, across oxygen tensions and varied stimuli, MØ activation and glycolytic flux are tightly and linearly related, resulting in calibration of glycolysis to keep pace with proinflammatory activation (tumor necrosis factor production) via a mechanism that depends on the ubiquitous PFK-2 isoenzyme and hypoxia inducible factor-1α ( Fig. 1 B–D).
In concert with the strong associations between glycolysis and MØ activation, several studies have demonstrated that arterial 18 F FDG uptake correlates with MØ density within the artery wall. Preclinical studies reported high correlations between 18 F FDG uptake and the density of MØ in atherosclerotic lesions and that anti-inflammatory therapies can reduce the arterial 18 F FDG-PET signal. Moreover, several groups have shown in human studies that 18 F FDG uptake in the carotid artery, which can be quantified noninvasively using PET, provides and index of atherosclerotic inflammation. Rudd and colleagues demonstrated that 18 F FDG uptake in carotid plaques was higher in symptomatic carotid lesions compared with the contralateral healthy asymptomatic lesions, which was confirmed later by autoradiography of carotid plaques after carotid endarterectomy. Several groups demonstrated that 18 F FDG uptake within carotid plaque correlates with the amount of macrophages in the inflamed plaque, by staining with anti-CD68 antibodies ( Fig. 2 A–G ). Further, the inflammatory signal is reproducible, as was shown in a study in which patients with known atherosclerotic disease underwent PET/CT imaging twice (2 weeks apart).
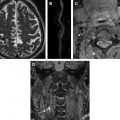
The measure of arterial inflammation correlates with risk factor prevalence and seems to predict subsequent structural changes within the artery wall. Arterial inflammation as assessed by 18 F FDG-PET/CT correlates with the Framingham Risk Score as well as with inflammatory biomarkers. In a study of 137 patients who underwent PET/CT imaging, arterial 18 F FDG uptake predicted the subsequent rate of arterial calcification within the underlying segment. Moreover, arterial 18 F FDG uptake has been repeatedly shown to provide an independent measure of risk for subsequent cardiovascular disease (CVD) events. Rominger and colleagues observed, in a retrospective study of 932 patients who underwent 18 F FDG-PET/CT imaging for cancer, that the 18 F FDG uptake (as a target-to-background ratio) predicted the risk of subsequent vascular events. In another retrospective study, Figueroa and colleagues in a study of 513 subjects who were followed for a median 4.2 years showed that arterial 18 F FDG uptake significantly and independently improved the prediction of cardiovascular events apart from Framingham Risk Score, resulting in a net reclassification improvement of approximately 27.5% ( Fig. 3 ). Furthermore, Marnane and colleagues showed in a prospective study of individuals with recent cerebrovascular accidents that carotid 18 F FDG uptake was associated with the risk of subsequent cerebrovascular accident ( Fig. 4 ). Accordingly, a growing body of retrospective as well as prospective studies shows that arterial 18 F FDG uptake independently predicts atherothrombotic events.
Evaluation of the pathophysiology of atherosclerotic diseases
Several chronic conditions are associated with both increased immune system activity and a heightened risk of atherosclerosis. PET/CT imaging is increasingly being used to examine the interplay between those inflammatory conditions and atherosclerosis. One such chronic inflammatory disorder, rheumatoid arthritis (RA), is associated with a substantially increased risk of CVD. A recent study using PET/CT imaging in individuals with RA reported that individuals with RA have substantially heightened arterial inflammation relative to carefully matched controls, and that inflammation in the artery wall correlates with the degree of inflammatory activity in the joints. Further, another study in patients with RA showed that 8 weeks of treatment with an antibody targeting tumor necrosis factor-α (used for the purpose of reducing joint inflammation), resulted in a reduction in atherosclerotic inflammation. These observations suggest a potential communication between the synovial and atherosclerotic inflammatory milieus, and supports the hypothesis that treating synovial inflammation may also benefit atherosclerosis.
Infection with the human immunodeficiency virus (HIV) is also associated with a heightened risk of atherosclerosis. Although the exact mechanisms underlying this risk have not been well-established, chronic up-regulation of immune system activity is believed to represent an important potential mechanism. To further evaluate the relationship between inflammation and HIV, Subramanian and colleagues performed 18 F FDG-PET/CT imaging in 81 subjects, including 27 with HIV infection and 54 matched controls. They found that individuals with HIV infection have increased arterial inflammation compared with noninfected individuals who were matched according to age, gender, and Framingham Risk Score. Furthermore, the arterial 18 F FDG signal was found to correlate with CD163 (a soluble marker of monocyte activation), suggesting that upregulation of the monocyte cellular niche may represent an important pathobiological mechanism of atherosclerosis in HIV. In a separate study, the same group evaluated whether arterial inflammation was associated with structural lesions within the coronary tree of individuals with HIV. They observed that individuals with increased arterial inflammation (greater than median values for aortic 18 F FDG uptake) had a 4-fold higher risk of having high-risk coronary plaques on CT (coronary atherosclerotic plaques with positive remodeling or low attenuation). Together, the findings link monocyte activation, to arterial inflammation, to the presence of high-risk coronary structural features, thus providing some mechanistic clues linking HIV to atherothrombosis.
The evaluation of arterial inflammation has provided insights into the pathobiology of milder inflammatory conditions as well. For example, asthma, which is associated with a very long duration of mild chronic inflammation, associates with atherosclerotic inflammation even after correcting for cardiovascular risk factors. Moreover, the degree of arterial inflammation correlates with the severity of the pulmonary condition. Along similar lines, another study observed a relationship between periodontal and carotid inflammation. In a later study, the same group used PET/CT imaging to study the impact of statin therapy on periodontal inflammation, and observed a substantial reduction in periodontal inflammation, which correlated significantly with changes in arterial inflammation.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree