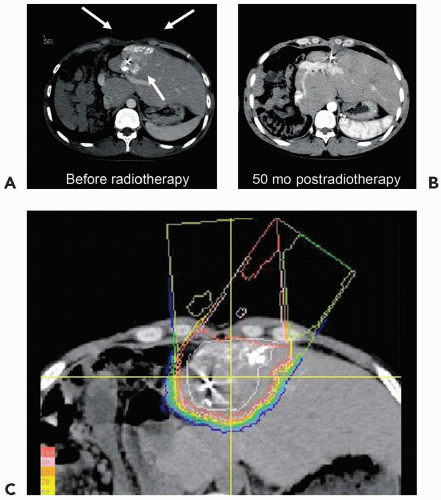
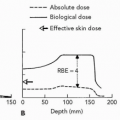
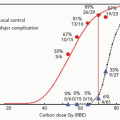
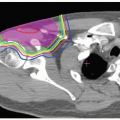
PBT for malignancies of various organs, including the liver, began at Tsukuba, Japan in 1983. One third of the 1,556 patients treated with PBT at Tsukuba as of March 2006 had HCC. Clinical experience in treating HCC patients with PBT gained at Tsukuba is presented in the following text.
Survival, Local Control, and Treatment Sequelae
From November 1985 through July 1998, physicians treated 165 patients with HCCs using a proton accelerator with maximum energy of 500 MeV at the original proton facility at Tsukuba.
8 The facility that had been used during the period was shut down in 2000. Proton therapy is currently given at a newer facility on the university campus that began treating patients in 2000. Of those 165 patients, clinical results are presented in the following text for 162 patients having 192 HCCs.
Tables 19.2 and
19.3 respectively present patient and tumor characteristics. The median total dose given was
72 Gy (range 50 to 88 Gy) with a median fraction dose being 4.5 Gy (range 2.9 to 6 Gy). The proton dose at Tsukuba has been expressed in Gy as the uncorrected physical dose rather than in the relative biological effectiveness (RBE)-corrected Gy (RBE) (generally using an RBE of 1.1) which most proton facilities have employed. Patients were treated with different fractionation regimens. For that reason, equivalent doses at 2 Gy per fraction for the several fractionation regimens employed were calculated using a linear quadratic (LQ) model with α/β ratios of 10 and 3 for early and late responding tissues (see
Table 19.4).
9
The local control rate calculated using the Kaplan-Meier method at 5 years was 86.9% for all 192 tumors among the 162 patients. The overall survival rate for all 162 patients at 5 years was 23.5%. The degree of impairment in hepatic function attributable to coexisting cirrhosis and the number of tumors affected survival in the Cox regression analysis (see
Table 19.5). Fifty patients had favorable prognostic features (Child-Pugh class A and a solitary tumor); their 5-year survival rate was 53.5%.
Treatment was generally well tolerated. The performance status (PS) of two (1.4%) patients declined from 0 before irradiation to 1 after irradiation and in another two patients (1.4%) from PS 2 before to PS 3 after irradiation. All remaining patients maintained an equivalent PS before and after irradiation.
Table 19.6 shows that very few acute reactions to the treatment were seen among the 162 patients over 185 treatment courses, aside from the transient elevation of aspartate aminotransferase (AST) and alanine aminotransferase (ALT). All acute toxicity resolved within 2 weeks and no patients discontinued treatment because of acute side effects.
Five patients had late sequelae of grade II or higher (the late radiation morbidity scoring scheme of the Radiation Therapy Oncology Group/European Organization for Research and Treatment of Cancer [RTOG/EORTC]). One patient developed fibrotic stenosis of the common bile duct within the treatment volume, noted 13 months after irradiation. Two patients developed biloma with
infection adjacent to the irradiated volume at 29 and 38 months after irradiation, respectively. One patient developed an intractable gastric ulcer in the treatment volume 4 months after irradiation and another patient had an ulcer in the ascending colon 6 months after irradiation. No patients died of treatment sequelae in this series.
From September 2001 through August 2004, the new facility treated 153 patients with HCC. Of those patients, 52 were treated with a proton dose of 60 Gy in 10 fractions over 2 weeks. The patients had Child-Pugh class A or B liver function and a solitary tumor of 10 cm or less in maximal diameter at the peripheral region of the liver. Of the 52 patients, 34 were men and 18 women; their median age was 69 years (range 26 to 85 years); 39 patients had Child-Pugh class A and 13 had B. The median tumor maximal diameter was 2.8 cm (range 0.8 to 9.3 cm). The median observed period was 22 months (1 to 47 months). Thirty-two patients were alive as of March 2006; 20 had died. The 3-year survival and local control rates were 41.9% and 94.6%, respectively. Grade 2 or higher late complications were observed in two patients—one patient suffered from radiation pneumonitis (grade 2) and rib fracture at 1 and 27 months after PBT; the other suffered from rib fracture at 8 months after PBT. This irradiation regimen of 60 Gy in 10 fractions is safe and effective when the tumor is located in the peripheral region of the liver and the liver function is reasonably preserved.