Histology, Formation, and Growth of Bone and Articular Cartilage
Bone: Histology, Formation, and Growth
The skeleton is made of cortical and cancellous bones, which are highly specialized forms of connective tissue. Each type of bony tissue has the same basic histologic structure (Fig. 3.1), but the cortical component has a solid, compact architecture interrupted only by narrow canals containing blood vessels (Haversian systems), while the cancellous component consists of trabeculae separated by fatty or hematopoietic marrow. A bone is a rigid calcified material and grows by the addition of new tissues to existing surfaces. The removal of unwanted bones, called simultaneous remodeling, is also a necessary component of skeletal growth. Unlike most tissues, a bone grows only by apposition on the surface of an already existing substrate, such as a bone or calcified cartilage. Cartilages, however, grow by interstitial cellular proliferation and matrix formation.
A normal bone is formed through a combination of two processes: endochondral (enchondral) ossification and intramembranous (membranous) ossification (Fig. 3.2). In general, the spongiosa develops by endochondral ossification and the cortex by intramembranous ossification. Once formed, a living bone is never metabolically at rest. Beginning in the fetal period, it constantly remodels and reappropriates its minerals along lines of mechanical stress. This process continues throughout life, accelerating during infancy and adolescence. The factors controlling bone formation and resorption are still not well understood, but one fact is clear: Bone formation and bone resorption are exquisitely balanced, coupled processes that result in net bone formation equaling net bone resorption.
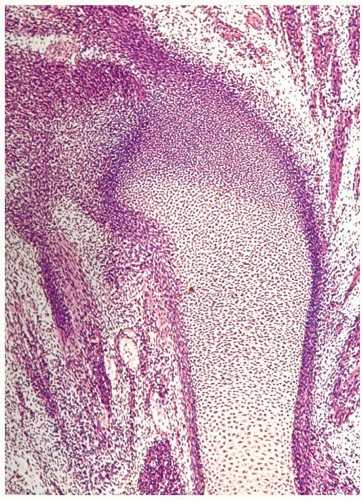
Most of the skeleton is formed by endochondral ossification (Fig. 3.3), a highly organized process that transforms a cartilage to a bone and contributes mainly to increasing the bone length. Endochondral ossification is responsible for the formation of all tubular and flat bones, vertebrae, the base of the skull, the ethmoid, and the medial and lateral ends of the clavicle. At approximately 5 weeks of embryonic life, cartilage cells (chondroblasts and chondrocytes) produce a hyaline cartilage model of the long tubular bones from the condensed mesenchymal aggregate (Fig. 3.4). The histologic events related to this process are first evident by changes in the condensation of undifferentiated mesenchymal cells, during which some of the cells inside the area of the future model become slightly larger and rounder and acquire noticeable cytoplasm. At the same time, the cells at the periphery of the models remain more spindle-shaped, less differentiated, and more condensed (Fig. 3.5). The mechanisms leading to the calcification of the cartilaginous matrix are not completely understood, but it is generally believed that the promoters of calcification are small membrane-bound vesicles known as matrix vesicles, which are present in the interstitial matrix between the cells (Fig. 3.6). At approximately the 9th week of embryonic life, peripheral capillaries penetrate the model, inducing the formation of osteoblasts. Osseous tissue is then deposited on the spicules of the calcified cartilage matrix that remain after osteoclastic resorption, thereby transforming the primary spongiosa into the secondary spongiosa (Fig. 3.7).
As this process moves rapidly toward the epiphyseal ends of the cartilage model, a loose network of bony trabeculae containing cores of calcified cartilage is left behind, creating a well-defined line of advance. This line represents the growth plate (physis) (Fig. 3.8) and the adjacent metaphysis to which the secondary spongiosa moves as it is formed. The many trabeculae of the secondary spongiosa that are resorbed soon after being formed become the marrow cavity, while other trabeculae enlarge and thicken through the apposition of a new bone, although these too eventually undergo resorption and remodeling. Others extend toward the shaft and become incorporated into the developing cortex of the bone, which is formed by intramembranous ossification. At the ends of tubular bones, a similar process is initiated. The cartilage at these sites expands by the interstitial accumulation of cartilage matrix. This appositional growth constitutes the main growth center that becomes vascularized by a network of invaginations from the perichondrium called cartilage canals (Fig. 3.9A). The cartilage surrounding these canals undergoes calcification and hypertrophy, followed by chondrocyte apoptosis, vascular invasion, and the formation of secondary ossification center in the epiphysis (Fig. 3.9B). This nucleus increases in size by the process of maturation and calcification of the cartilage surrounding the secondary center. The peripheral margin of epiphysis termed acrophysis is formed of zones of cell hypertrophy, degeneration, calcification, and ossification, similar to that of the growth plate. Endochondral bone formation is not normally observed after growth plate closure.
In intramembranous ossification, a bone is formed directly without an intervening cartilaginous stage (Fig. 3.10). Initially, condensed mesenchymal cells differentiate into osteoprogenitor cells, which then differentiate into fibroblasts that produce collagen and fibrous connective tissues and osteoblasts that produce osteoid (Fig. 3.11). Beginning at approximately the 9th week of fetal life, the fibrous membrane produced by the fibroblasts forms a periosteal collar and is replaced with osteoid by the action of the osteoblasts. Bones formed by this process include the frontal, parietal, and temporal bones and their squamae; bones of the upper face as well as the tympanic parts of the temporal bone; and the vomer and the medial pterygoid.
Intramembranous ossification also contributes to the appositional formation of periosteal bones around the shafts of the tubular bones, thus forming the cortex of the long and flat bones. This type of bone formation increases the bone width. In addition to the periosteal envelope on
the outer surface of a bone, intramembranous ossification is active in the endosteal envelope covering the inner surface of the cortex and in the haversian envelope at the internal surface of all intracortical canals (Fig. 3.12). These three envelopes are sites of potent cellular activity involving resorption and formation of bones throughout life.
the outer surface of a bone, intramembranous ossification is active in the endosteal envelope covering the inner surface of the cortex and in the haversian envelope at the internal surface of all intracortical canals (Fig. 3.12). These three envelopes are sites of potent cellular activity involving resorption and formation of bones throughout life.
 FIGURE 3.3 Endochondral bone formation. This process occurs at the ossification center, growth plate, and metaphysis. (Reprinted with permission from Anatomical Chart Company. Rapid review anatomy reference guide, 3rd ed. Philadelphia, PA: Wolters Kluwer Health; 2010, Fig. 1-11.) |
 FIGURE 3.5 Histology of early endochondral bone formation. (Continued) (C) The first histologic evidence of cartilaginous differentiation is the formation of extracellular mucopolysaccharide matrix in the condensed areas of mesenchyme. Between the lightly basophilic matrix, the nuclei of the mesenchymal cells become round and their chromatin is more vascular than that of the surrounding spindle cells. (D) In the later stages, the cells acquire lacunae, which separate the cytoplasm from the interstitial matrix. The model become histologically identical to hyaline cartilage. (Reprinted with permission from American Registry of Pathology, from Klein MJ, Bonar SF, Freemont T, et al, eds. Atlas of nontumor pathology. Non-neoplastic diseases of bones and joints. Washington, DC: American Registry of Pathology and Armed Forces Institute of Pathology; 2011: 5-6, Figs. 1.4A-C and 1.5C.) |
 FIGURE 3.6 Histology of late endochondral bone formation. Photomicrograph of the diaphysis of the long bone in a 7-week-old fetus shows calcification of the cartilage matrix (black) (von Kossa stain, ×4). (Reprinted from Bullough P. Orthopaedic pathology, 5th ed. Maryland Heights, MO: Mosby; 2009, with permission from Elsevier.)
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|