Bowel wall thickening in the hospitalized patient can be due to myriad etiologies. Familiarity with the optimal study protocols and a structured approach for evaluation are important. Understanding the pathology and knowing the imaging features of most common entities (ischemia, shock bowel, hemorrhage, infection, graft-versus-host disease, and fluid overload) enable radiologists to provide unique value to clinical management.
Key points
- •
Bowel wall thickening in the hospitalized patient is a nonspecific finding that can be encountered in a wide range of pathologic conditions. Proper evaluation starts with an optimal targeted study protocol.
- •
A pattern-based approach for image analysis consists of assessing length and sites of involvement, any changes to the fold pattern, the severity and attenuation of wall thickening, bowel contents, bowel dilation, and ancillary findings in the mesentery, vessels, and other organs.
- •
It is important for the radiologist to integrate imaging features with clinical findings and laboratory analysis to reach the most sensible diagnosis.
- •
Radiologists should be familiar with the most common entities causing bowel wall thickening in the hospitalized patients and with red flags that should be promptly brought to clinical attention.
Introduction
Hospitalized patients, especially in tertiary and quaternary care facilities, often have complex medical histories with multiple comorbidities. Due to nonspecific and variable clinical manifestations and, in many cases, detrimental consequences when diagnosis is delayed, bowel pathology should be considered during work-up of abdominal symptoms, including nausea, vomiting, gastrointestinal bleeding, and abdominal pain. Hospitalized patients may have abnormal bowel due to vascular, infectious, inflammatory, neoplastic, or other etiologies. It is important to remember the broad range of conditions effecting the bowel, particularly in critically ill patients, in whom an accurate physical examination and history may be difficult to obtain. Whether the cause for admission or not, acute conditions of the bowel can be life-threating and may be difficult to diagnose, in part due to their overlapping imaging appearances. One important manifestation of gastrointestinal disease, bowel wall thickening, is a nonspecific finding that can be seen with a variety of pathologies but also can be a valuable tool to allow radiologists to provide an appropriate differential diagnosis. With the increasing use of cross-sectional imaging in these settings, radiologists are also relied on to be familiar with the utility and limitations of different imaging modalities to evaluate the gastrointestinal tract. This article describes the characterization of bowel wall thickening in the hospitalized patient in different clinical contexts and provides pictorial examples of common relevant pathologies with the goal of aiding radiologists in diagnosing these conditions, thereby adding value in clinical management.
Normal anatomy and imaging technique
The small and large bowel is made up of multiple layers, including, from internal to external, the mucosa, submucosa, muscularis propria, and serosa (intraperitoneal) or adventitia (retroperitoneal). Retroperitoneal segments of bowel include the second and third portions of the duodenum, the ascending and descending colon, and the rectum. The histology of the wall layers is varied throughout the alimentary tract depending on function. Although the mucosa is best evaluated with direct visualization through endoscopy or less frequently with fluoroscopic examinations, computed tomography (CT), MR imaging, and ultrasound assess all layers of bowel and can show differential enhancement of the individual layers.
All imaging modalities have important uses in the abdomen, although some offer more useful information in the setting of bowel pathology than others. Considerations for choosing a modality include not only bowel visualization but also availability, radiation dose, cost, and patient stability.
Ultrasound is not often used to evaluate the bowel in hospitalized patients due to its focused nature, but it has been well established in certain populations, including pediatric patients with intussusception and pregnant patients with appendicitis. Contrast-enhanced ultrasound is gaining popularity in the work-up of inflammatory bowel disease in children, especially in Europe and Canada. Additionally, ultrasound may be used to target percutaneous procedures, including placement of tubes and potentially biopsy of exoenteric bowel wall masses. Due to its inability to visualize the entire gastrointestinal tract and its well-known operator dependency, however, ultrasound has limited use in this clinical context.
The ability to perform portable radiographic studies is an advantage for critically ill patients who are too unstable for transport. Although there are some occasions when bowel conditions have typical aunt-Minnie appearances on abdominal radiographs, such as the lead-pipe colon in ulcerative colitis ( Fig. 1 ), abdominal radiographs most often have limited utility in the setting of bowel wall thickening because of their poor resolution of superimposed structures. Fluoroscopic examinations with intraluminal contrast agents have historically been used for the evaluation of bowel pathology and remained the only way to image the bowel lumen for several decades. Similar to radiography, fluoroscopy provides limited information about submucosal and extraluminal disease and also has the added limitations of the need for patient cooperation, prolonged imaging times, and often the requirement that the radiologist obtains the images to ensure an adequate study. High-quality barium fluoroscopic examinations also may not be possible in hospitalized patients due to the need for preparation and gaseous distention as well as concern for possible leak into the peritoneal cavity in surgical patients. Thus, although the high spatial resolution and dynamic aspect of fluoroscopy make it useful for evaluation of abnormal mucosa, small fistulous tracts, and early leaks ( Fig. 2 ), these benefits do not make up for the technical challenges, and the use of fluoroscopy in evaluating bowel pathology has been declining.


Because of the limitations of fluoroscopy in the hospitalized patient, cross-sectional imaging with CT and MR imaging is often called on to provide more comprehensive information about abdominal pathology, including the bowel wall. These modalities allow for evaluation of all bowel wall layers, associated structures such as mesenteric vessels, and the remainder of the abdomen, which could suggest alternative diagnoses. MR imaging is desirable due to its superior contrast resolution and lack of ionizing radiation, which makes it a safe choice in pediatric and pregnant populations and in patients who need repetitive imaging for chronic conditions, such as inflammatory bowel disease, but these advantages need to be balanced with the need for the hospitalized patient to be still and cooperate with breathing instructions. Patients with claustrophobia, end-stage renal disease precluding contrast, and implantable devices present challenges for performing an MR imaging as well. Because of these concerns, contrast-enhanced CT of the abdomen and pelvis has emerged as the most commonly ordered modality for the work-up of bowel pathology in the hospitalized patient given its availability and speed of acquisition compared with MR imaging. Because of the above-mentioned, limitations with other imaging modalities, this article largely focuses on CT imaging for this patient population.
Imaging protocols
Protocol selection is an important consideration when performing CT to evaluate the patient with abnormal bowel. Although a noncontrast study can provide some information about bowel wall thickening, it remains a limited examination ( Fig. 3 ). The importance of administering intravenous contrast cannot be overstated, because it allows for evaluation of bowel wall hyperenhancement and hypoenhancement, mesenteric vasculature, and solid organs. Generally patients with glomerular filtration rate greater than 30 mL/min per 1.73 m 2 and no contrast allergy can safely receive contrast, and detailed guidelines are routinely updated by the American College of Radiology (ACR). For a patient with an acute abdominal presentation requiring an emergency department visit or admission to an inpatient unit, a common practice is to perform single acquisition CT with intravenous contrast in the portal venous phase. For the patient with known or highly suspected bowel pathology, however, CT protocols can be optimized to specifically evaluate for inflammatory bowel disease, bowel ischemia, and acute gastrointestinal bleed ( Table 1 ). Detailed clinical history allows a radiologist to choose the appropriate protocol. These protocols call for variable timing of one or more postcontrast phase(s). Some require initial noncontrast imaging, which can be helpful for evaluating enhancement as well as documenting the presence of hyperdense material that otherwise could be confused for contrast extravasation. Dual-energy CT can potentially obviate a separate precontrast acquisition by allowing virtual noncontrast imaging.

| Protocol | Components and Timing | Slice Thickness |
|---|---|---|
| Mesenteric ischemia/gastrointestinal bleed | Oral contrast: none Intravenous contrast rate: 4 mL/s Phases: noncontrast Arterial 35 s fixed delay or 5 s after aorta trigger Portal venous 60–90 s fixed delay | Noncontrast: axial 3 mm Arterial: axial 2 mm, coronal 3 mm Portal venous: axial 3 mm |
| Small bowel obstruction | Oral contrast: none Intravenous contrast rate: 3 mL/s Phase: portal venous 60 s fixed delay | Axial 2 mm, coronal 3 mm |
| CT enterography | Oral contrast: dilute 0.1% barium sulfate suspension Intravenous contrast rate: 3 mL/s Phase: portal venous 60 s | Axial 3 mm, coronal 3 mm |
The value added by the routine use of oral contrast in abdominal and pelvic CT, especially in the inpatient and emergency setting, is not as well-established as that of intravenous contrast and has been a controversial topic until recent ACR guidelines recommended against its routine use in the emergency setting. In the 1980s, it was widely accepted that positive (radiopaque) enteric contrast was necessary for adequate visualization of the abdominal organs and that negative (radiolucent) contrast was helpful in evaluating intraluminal disease. Over the past few decades, however, arguments against oral contrast have emerged, especially in the acute setting, including increased wait time for the patients before the scan, potential risk of aspiration of oral contrast material, and reportedly being the least pleasant portion of the CT examination for patients. The use of oral contrast likely was more valuable when CT slices were thicker and the image quality was relatively poor, but with modern computerized display and the ability to scroll through sequential images, radiologists can identify the continuity of structures to discern bowel loops from lymph nodes or soft tissue lesions. Even with the use of oral contrast, uniform opacification of the gastrointestinal tract is hard to achieve. Furthermore, with the growing overweight and obese population, many adult patients have enough visceral fat to provide spatial resolution between bowel loops and abdominal structures. A 2015 study by Alabousi and colleagues showed that omitting oral contrast for patients with body mass index greater than 25 kg/m 2 presenting with acute abdominal pain resulted in no delayed or missed diagnoses.
In the authors’ institution, CT studies are performed without oral contrast except in select scenarios. Prompt imaging is important in the acutely ill patient, and the potential value of oral contrast often does not justify a 30-minute to 90-minute imaging delay. Positive oral contrast may obscure the bowel mucosa and evaluation of enhancement. Circumstances in which the authors use intraluminal contrast include positive oral contrast for suspected extraluminal leak in postsurgical patients (fluoroscopy is an alternative study in this group) and suspected traumatic bowel injury if initial trauma CT was equivocal, negative oral contrast (such as water or dilute 0.1% wt/wt barium sulfate suspension) for CT enterography, and carbon dioxide insufflation for CT colonography. Regarding negative oral contrast agents, dilute barium sulfate has been shown to provide better distention of the stomach, small bowel, and colon and better visualization of bowel wall features compared with water alone and/or water containing methylcellulose. Additionally, the low radiographic density of neutral or negative oral contrast minimizes attenuation artifact on CT scans. Pediatric or thin patients may also require oral contrast to improve visibility of the gastrointestinal tract and separate it from fluid collections and/or masses.
Several other important factors should be considered when developing imaging protocols. Thinner slices provide better spatial resolution, but thicker slices result in better signal to noise ratio; therefore, slice thickness must be optimized to the specific anatomy in question. A 3-mm to 5-mm slice thickness typically is used. Reconstructions in coronal and sagittal planes are routinely generated and useful for following the bowel. Additional reconstructions, such as 3-dimensional volumetric images, maximum intensity projection images, curved reconstructions, and oblique and double oblique views, may be performed, particularly for assessment of the mesenteric vasculature.
Variables relating to intravenous contrast, namely contrast volume, rate of injection, and timing of image acquisition, also must be adjusted depending on the patient and clinical question. A larger volume of contrast media results in greater tissue enhancement and may be needed in larger patients. This also should be balanced, however, with potential risk for postcontrast acute kidney injury. Increasingly, dual-energy CT applications are used to artificially lower the kilovoltage after scanning to improve the conspicuity of lower quantities of iodinated contrast media. A faster injection rate (3–5 mL/s) allows for improved vascular opacification, but it results in a short temporal window for image acquisition in specific phases and usually requires a larger caliber (14–20 gauge) and more proximal (antecubital or central) intravenous access. This is needed for evaluating patients with suspected gastrointestinal (GI) bleeding or bowel ischemia, in whom vascular assessment is critical. A slower injection rate is technically easier and is sufficient for parenchymal and venous imaging because those phases last for a relatively longer time. Once contrast has been injected, images can be obtained at a fixed delay or at a time that is individualized to each patient. Individualized options are generally used for vascular imaging and for patients with compromised cardiac output and include test-bolus and bolus-tracking methods. The bolus-tracking method is generally used for abdominal imaging when contrast timing is of utmost importance. Images are obtained at a reference level (usually the aorta) and repeated after injecting the full dose of contrast. When enhancement reaches a predetermined threshold, image acquisition is triggered. Alternatively, the test-bolus method involves generating a time-enhancement curve from low radiation images obtained with a small contrast volume that is then used to calculate the appropriate imaging delay. It models a patient’s cardiovascular response and often is seen in the setting of cardiac CT angiography.
Imaging findings—approach to diagnosis
Bowel wall thickening in the hospitalized patient is a nonspecific finding that can be encountered in a wide range of pathologic conditions. This necessitates a systemic approach for evaluation of bowel wall thickening, which consists of assessing the length and sites of involvement; the pattern of enhancement; any changes to the pattern of haustration or valvulae conniventes; the severity and attenuation of wall thickening, bowel contents, bowel dilation; and any ancillary features outside the bowel, such as mesenteric findings.
Normal small and large bowel thickness is variable and depends on bowel distention. In the colon, a normal wall measures up to 6 mm to 8 mm if the lumen is decompressed and 0 mm to 2 mm with increasing distention. The small bowel wall should measure no more than 3 mm if the lumen is decompressed and 0 mm to 2 mm if the bowel is distended. Because bowel wall thickness varies with the degree of distention and because decompressed bowel loops can be difficult to delineate from one another, accurately diagnosing bowel wall thickening on imaging can pose a challenge. Many factors, including but not limited to the clinical context, distribution and severity of bowel wall thickening, pattern of enhancement, and bowel wall attenuation as well as mesenteric and other associated nonenteric findings, are important to note when evaluating bowel wall thickening. These factors provide the basis for the selection of imaging studies and protocols available for the work-up of bowel pathology.
Sites and Length of Involvement
The traditional approach to evaluating small bowel pathology using barium studies relied on the length of involvement as a major variable in the diagnostic approach to bowel wall thickening. Different disease processes tend to involve the bowel in variable patterns, with the most common patterns being segmental versus diffuse involvement. The length and site of involvement can help in narrowing the differential considerations ( Table 2 ). The more prevalent pathologies in hospitalized patients, including infectious, inflammatory, and ischemic processes, tend to involve longer segments of bowel. Exceptions include skip lesions in Crohn disease and noncontiguous ischemic bowel, such as in watershed areas. In contrast, systemic processes, such as graft-versus-host disease (GVHD), celiac disease, and lupus, and edema, generally involve long segments of the bowel.
| Length of Bowel Involvement | Differential Diagnosis |
|---|---|
| <5 cm | Adenocarcinoma |
| 5–10 cm | Diverticulitis, Crohn disease, ischemia |
| 10–30 cm | Ischemia, submucosal hemorrhage, radiation enterocolitis, infection, Crohn disease, lymphoma |
| Diffuse colonic involvement | Ulcerative colitis |
| Diffuse small and large bowel involvement | Infection, edema, lupus |
Enhancement and Fold Pattern
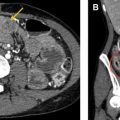
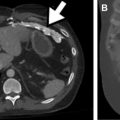
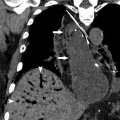
The bowel wall attenuation is one of the most useful clues for differential diagnosis and can be a reflection of the pathophysiology of the underlying process, either due to its intrinsic attenuation or that of increased or decreased enhancement ( Fig. 4 ; Table 3 ). Reliable attenuation difference is defined as at least 10 Hounsfield units and preferably 20 Hounsfield units above that of the reference structure, which is usually skeletal muscle. White attenuation, which describes full-thickness transmural enhancement of the bowel wall, can be seen with severe active inflammation or with leaky capillaries. Less commonly, diffuse hyperattenuation of the bowel wall can be secondary to acute submucosal hemorrhage and is best seen on noncontrast images. Gray enhancement reflects a lower level of enhancement than skeletal muscle, making it difficult to differentiate the layers of bowel wall. Submucosal edema of the bowel usually results in the water target sign, which describes enhancing inner (mucosa) and outer (muscularis propria and/or serosa) wall layers. Bowel wall fat and gas generally are easy to appreciate and differentiate ( Fig. 5 ).


Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree