Infections in Patients with Metallic Implants: PET-CT and SPECT-CT Imaging
Katrin D. M. Stumpe
Jürg Hodler
The diagnosis of musculoskeletal infections in patients with metallic implants still remains a major challenge. Most imaging methods suffer from one or more limitations such as low sensitivity in detecing bone or soft tissue abnormalities and low specificity in differentiating infection from reparative tissue as well as implant-associated artifacts.
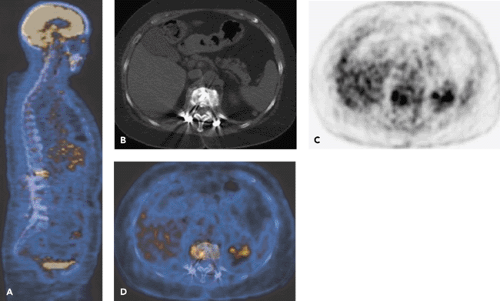
Radionuclide studies currently represent imaging methods of choice in patients with suspected infection in the presence of metallic implants. However, nonspecific tissue uptake of imaging agents and limited spatial resolution restrict their usefulness. Because of these problems, the diagnosis of implant-associated infection may be delayed. Revision surgery may have to be planned without definitive diagnosis. The interest in advanced imaging methods is, therefore, considerable. Fluorodeoxyglucose (FDG) positron emission tomography (PET) imaging is both sensitive and specific in the diagnosis of implant-associated infections in trauma patients because FDG is taken up into activated white cells only. PET–computed tomography (CT) offers additional information as it provides precise anatomic information (see Fig. 61.1) and can identify sequestered bone fragments, which is relevant for surgical planning. Whether single-photon emission computed tomography (SPECT)–CT can replace PET-CT in institutions without PET-CT remains to be seen.
In contrast to use in patients with implants used for trauma surgery, PET and PET-CT may not be as useful in the diagnosis of infection in patients with hip and knee prostheses.
General Remarks
The diagnosis of implant-associated infections in trauma and orthopedic sugery is difficult (1,2). Clinical signs and laboratory parameters may be uncharacteristic or even normal in so-called low-grade infections. Standard radiographs may demonstrate implant loosening, periosteal reaction, osteopenia, osteolysis, osteosclerosis, or sequester formation, depending on the stage of infection. However, in early and low-grade infection and in the diagnosis of abnormalities related to soft tissue, these signs may not be diagnosic. Ultrasound has its value in diagnosing abnormalities of bone surfaces and soft tissues, but is not useful in diagnosing intraosseous changes. Magnetic resonance imaging (MRI) and computed tomography (CT) tend to have degraded image quality in the presence of metallic
implants owing to susceptibility and beam-hardening artifacts, respectively. Even when metallic devices are removed, small residual metallic particles can lead to susceptibility artifacts that can limit the interpretation of MR images.
implants owing to susceptibility and beam-hardening artifacts, respectively. Even when metallic devices are removed, small residual metallic particles can lead to susceptibility artifacts that can limit the interpretation of MR images.
In light of these considerations, nuclear medicine studies play an important role. Radionuclide imaging may be the most useful imaging modality for evaluating painful prosthesis, although the differentiation of aseptically loosened from infected joint prosthesis is not always possible. Standard bone scintigraphy represents an initial screening test. A normal bone scan rules out both septic and aseptic loosening. Bone scintigraphy is more sensitive than plain radiographs or arthrography (3,4) in the diagnois of loosening. 67Gallium (67Ga) imaging combined with bone scans was originally suggested for suspected implant-associated infection. Because of different radiotracer characteristics of 67Ga citrate imaging and bone scintigraphy, each study provides complementary information (5). However, the use of gallium has substantial limitations in this setting; as uptake also is found in postoperative reactive changes, which may be difficult to differentiate from postoperative osteomyelitis. 111Indium (111In)-labeled white blood cells in combination with 99mtechnetium (99mTc) sulfur colloid marrow imaging has become the method of choice to assess infection in total joint replacement, as it allows correction for the abnormal leukocyte accumulation that may occur because of either displacement of marrow at the time of surgical implantation of the prosthesis or stimulation of the conversion of yellow to red marrow. Noncongruent radionuclide uptake in leukocyte and marrow scintigraphy with absent bone marrow uptake and accumulation of leukoctyes is the most accepted criterion of infection (6,7,8).
Labeled leukocyte imaging alone has improved specificity in comparison with other imaging methods (9). However, leukocyte imaging cannot distinguish between the leukocytes of normal hematopoietic bone marrow and activated white cells. The diagnosis of osteomyelitis may be difficult in the axial and to a lesser degree in the peripheral skeleton. Labeled leukocyte imaging combined with bone scanning or bone marrow scanning is helpful in this setting, and the combination demonstrates higher sensitivities and specificities than leukocyte imaging alone (10). Fluorodeoxyglucose (FDG) positron emission tomography (PET) also has been shown to be very promising in this setting (Fig. 61.1), particularly in patients in whom bone anatomy has been altered because of the presence of posttraumatic bone deformity and in chronic infection (11,12,13,14). However, currently no tracer is equally satisfactory for all types of infections in the musculoskeletal system.
The use of FDG-PET in this setting has the potential advantage of being a single-step procedure with imaging 1 hour after the injection; other advantages are rapid loss of radioactivity from the patient and a radiation dose comparable with that of leukocyte-imaging techniques. Initial studies indicate that FDG-PET detects both loosening and infection and differentiates between them in both hip and knee prostheses. However, recent data are inconsistent and less encouraging (8,15,16,17,18,19,20,21). Metallic artifacts and foreign
body reactions influence PET image interpretation in patients with prostheses (see also Chapter 59, e.g., Fig. 59.5).
body reactions influence PET image interpretation in patients with prostheses (see also Chapter 59, e.g., Fig. 59.5).
Metallic Implant-Associated Infections in Trauma Patients
In traumatology, osteomyelitis is a rare but serious complication after fracture stabilization with metallic implants. Despite important advances in surgical and long-term antibiotic treatment, osteomyelitis often remains refractory to therapy, leading to chronic illness. Infection is responsible for 5% to 10% of nonunions after fractures. Particularly in patients with nonunited fractures who have previously undergone surgery, the distinction between fracture instability and implant-associated infection may not be possible with the currently available imaging modalities (1,2).
Standard radiographs may demonstrate nonunion, sequestered bone, intraosseous abscess formation, and bone resorption at implant–bone interfaces in infection. They are not useful, however, for the diagnosis of early or low-grade infection. MRI sensitively demonstrates abnormalities of bone marrow and soft tissue if not hampered by susceptibility artifacts caused by metallic implants. However, reparative and infected tissue may be impossible to differentiate. This is also true for ultrasound, which is also limited to bone surfaces and soft tissue. CT more precisely demonstrates fragments and sequesters than standard radiographs, but is inferior to MRI in soft tissue and bone marrow assessment. CT also suffers from beam-hardening artifacts at the level of implants. Modified imaging techniques and protocols in MRI and CT decrease artifacts in the vicinity of metallic instrumentation (22,23,24), but do not provide perfect images.
In conventional nuclear medicine, the three-phase bone scan has been the accepted examination for the initial evaluation for osteomyelitis, but findings are affected by previous surgery and trauma and are often not specific. The limitations of spatial resolution are a relevant problem. Specificity increases with combined bone and gallium scanning. Specificities of this combined technique, however, still vary between less than 50% and 100% (1,2,5,25). 111In-labeled leukocyte combined with 99mTc methylene diphosphonate scintigraphy has been the method of choice for posttraumatic infection imaging at fracture nonunion sites (1). Sensitivities of 84% and specificities of 97% have been reported in the detection of osteomyelitis (26,27). This combined method improves the specificity of 111In-labeled leukocyte imaging alone by differentiating between soft tissue infection and bone infection at the fracture site.
In the last decade, 111In-labeled leukocyte combined with 99mTc bone marrow scintigraphy have been shown to be highly accurate for the diagnosis of various musculoskeletal infections that could alter distribution of bone marrow with sensitivities and specifities of 100% and 94%, respectively (10).
FDG-PET appears to have a sensitivity superior to that of combined leukocyte–bone marrow scintigraphy, particularly in chronic infections of the axial skeleton. FDG-PET also has practical advantages as FDG accumulates within 1 hour in the lesions, contrary to other infection-specific nuclear medicine modalities requiring multiple scans and imaging over a period of up to 24 hours because of the slow accumulation kinetics of these radiotracers. Furthermore, PET is a tomographic technique with substantially better spatial resolution than that of other scintigraphic techniques. FDG-PET may also provide a different type of imaging characteristics, as uptake of FDG in bone and bone marrow is relatively low. The available data suggest that FDG-PET better distinguishes between osseous and soft tissue infection than other techniques. This is relevant for the surgeon, as the operative approach differs greatly when osteomyelitis is present. Unlike MRI and CT (Fig. 61.2) (28,29), FDG-PET images do not show substantial implant-associated artifacts or tissue edema, which can interfere with image interpretation in patients with osteosynthetic metallic implants. These contrast with the findings in patients with hip and knee prostheses, as is discussed in the next section.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree