CHAPTER 110 Inferior Vena Cava and Its Main Tributaries
INTRODUCTION
Inferior Vena Cava
Normal Anatomy and Congenital Anomalies
The IVC extends from the confluence of the common iliac veins at the level of L5 vertebral body, to the right atrium of the heart in right prevertebral location, next to the abdominal aorta and is surrounded by a rich network of lymphatic vessels (Fig. 110-1). It is partially covered anteriorly by the peritoneal membrane. The retroperitoneal space where the IVC is located can communicate with the perirenal spaces and the anterior and posterior interfascial spaces.1
Duplicated IVC: There are two IVCs below the level of the renal veins—each connected to the ipsilateral common iliac vein. The left IVC joins the left renal vein, which then crosses anterior to the abdominal aorta and drains into the right IVC (Fig. 110-2). There may be variants in this anatomy and there may be significant discrepancy in the size of the two IVCs.
Azygos continuation of IVC: The infrarenal portion of IVC receives blood from the renal veins. It passes posterior to the diaphragmatic crura, enters the thorax as the azygos vein, and then joins the superior vena cava at the azygos arch. The hepatic and right adrenal veins drain directly into the right atrium. The gonadal veins drain into the ipsilateral renal veins. The right renal artery crosses abnormally anterior to the IVC (Fig. 110-3).
Circumaortic left renal vein: There are two left renal veins. The superior-anterior renal vein receives the adrenal vein and crosses the aorta anteriorly to join the IVC. The second renal vein is approximately 1 to 2 cm more inferior and posterior. It receives the left gonadal vein and crosses posterior to the aorta to join the IVC (Fig. 110-4).
Retroaortic left renal vein: The renal vein crosses posterior to the aorta to join the IVC.
Etiology and Pathophysiology (Including any Special Anatomic Considerations)
Embryogenesis
Knowledge of the IVC embryogenesis is necessary for a better understanding of the IVC anatomic aberrations. The IVC is composed of four segments which form during the 6 to 8 weeks postconception.2,3 This is due to continuous appearance and regression of three paired embryonic veins, which include the posterior cardinal veins, the subcardinal veins, and the supracardinal veins. The first step in this complex process is the formation of the posterior supracardinal and more anterior subcardinal veins. Then, the most caudal segment of the right supracardinal vein becomes the infrarenal vena cava. The hepatic segment of IVC is derived from the vitelline vein, which conveys blood from the viscera. The suprarenal segment is formed via a subcardinal-hepatic anastomosis. The renal segment forms via anastomosis of the right supra-subcardinal and post-subcardinal veins. The infrarenal segment arises from the right supracardinal vein.
Manifestations of Disease
Clinical Presentation
With retroaortic left renal vein, an increased incidence of testicular varicoceles has been reported, presumably due to compression of the left renal vein by the abdominal aorta. With the circumcaval ureter type, there could be partial right ureteral obstruction or recurrent urinary tract infection. With the absence of infrarenal IVC or entire IVC, patients may present with venous insufficiency of the lower extremities or idiopathic deep venous thrombosis. Approximately 5% of patients younger than 30 years with idiopathic deep venous thrombosis show IVC absence on CT. Almost 10% of these patients with a coexisting thrombophilia have congenital absence of the IVC.4
Imaging Indications and Algorithm
Imaging Techniques and Findings
Reporting: Information for the Referring Physician
TUMORS OF THE IVC AND MAIN TRIBUTARIES
Prevalence and Epidemiology
Primary IVC tumors (leiomyosarcomas) are very rare with only one large series published in the literature.5 It is more common to see tumors invading the IVC through its tributary veins arising from separate abdominal organs. One of the most common causes of neoplastic invasion of the IVC lumen is the renal cell carcinoma (RCC) that can be seen invading the IVC through the renal vein in 4% to 10% of the cases. Hepatocellular carcinoma (HCC) frequently invades the portal vein but also on rare occasions can invade the IVC through the hepatic veins. Primary adrenocortical carcinoma is a rare adrenal tumor that invades the IVC. Multiple other retroperitoneal tumors can compress and invade the IVC, including lymphomas, metastasis of gonadal or uterine tumors, pheochromocytomas, and other retroperitoneal sarcomas.
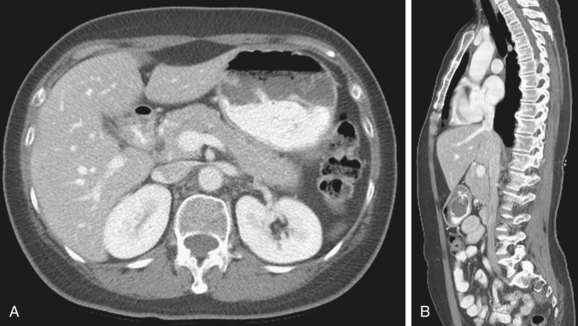
 FIGURE 110-1
FIGURE 110-1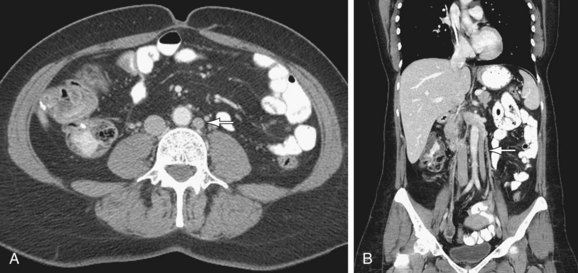
 FIGURE 110-2
FIGURE 110-2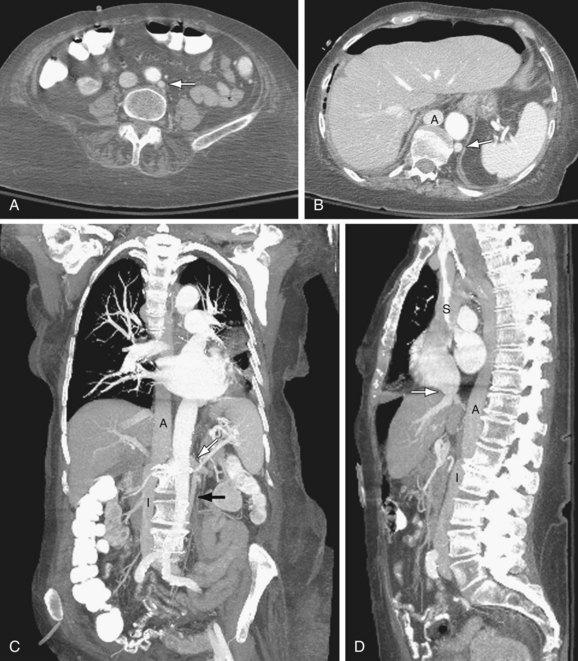
 FIGURE 110-3
FIGURE 110-3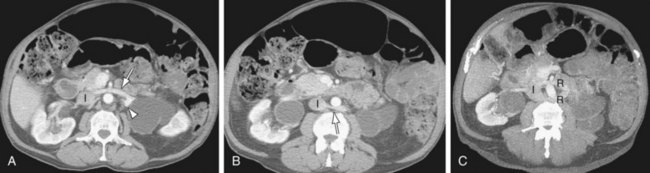
 FIGURE 110-4
FIGURE 110-4


