Pankaj Sharma, Mohit Tayal, Udit Chauhan, Dheeraj Kumar Tiwari, Mathew Cherian, Kaurabhi Zade From open surgeries to microinvasive techniques, treatments have seen huge developments in the 20th century and refinements in the 21st century. Use of image guidance revolutionized the way grave, and serious diseases were once looked upon. It has resulted in significantly reduced morbidity related to open procedures and at the same time has reduced hospital stay, costs incurred and improved posthospital stay. From the use of X-rays, the image guidance now finds newer horizons with ultrasound and magnetic resonance guidance. The field has interested researchers around the world as we witness modern more precise hardware and medications for use. Whether it is an ambiguous lesion on routine imaging or a challenging deep-seated retroperitoneal lesion otherwise requiring major open surgery. Interventional radiology (IR) has established its roots through fine needle aspiration and biopsy techniques. Nonvascular procedures have frequently been able to avert the need of impending operations. It has always been difficult for surgeons to operate upon morbidly ill patients falling into various contraindications. Vascular procedures have emerged as a viable alternative to all those difficult situations where control of haemorrhage is the primary goal. Revascularization of the occluded vessel or chemoembolization of a tumour, all have been made possible by this novel field of science that is sure to witness more development in time to come. IR is a fascinating subspecialty of radiology, in which we not only diagnose lesions but also treat diseases using minimally invasive techniques, with advantage of doing procedures under image guidance. Before the advent of IR, treatment of disease was mainly done by surgery, with associated morbidity and complications. But, with introduction of IR, we could treat the same disease with minimally invasive route, with associated advantages such as early mobility, less morbidity and better acceptability of IR procedure by patients. The term IR was coined in 1967 by Margulis AR. First published literature for arteriogram was in 1924 by Brooks B and this arteriogram was done by surgeons by direct cutdown. Forssmann W performed first human catheterization on himself, using urinary catheter at young age of 25 years in Refs. Two advancements which were milestone in history of IR are Seldinger technique, described by Svan Ivar Seldinger and first angioplasty was done inadvertently by Charles T Dotter in 1963, using Seldinger technique. In 1953, Swedish radiologist, Svan Ivar Seldinger introduced famous technique known as “Seldinger technique” for introducing catheter into the vascular system, following needle access. Technique described by Svan Ivar Seldinger was double-wall puncture technique, wherein both walls of vessel were punctured with access needle without image guidance and then access needle was slowly withdrawn, until gush of blood was seen. This gush of blood was proof of access needle being within vessel lumen. Thereafter, guidewire was introduced into vessel lumen through needle, and needle was withdrawn over guidewire. Compression was applied at point where access needle had first pierced vessel wall. Thereafter, catheter was secured in vessel lumen over guidewire. Nowadays, we use “modified Seldinger technique”. In modified Seldinger technique, we use ultrasound (USG) guidance to access vessel lumen, and only single-wall puncture is done. Advantage of modified Seldinger technique is that we use in-plane view and we know preciously, when access needle has entered vessel lumen, and this ultimately results in almost no blood loss and reduces complications rate drastically. Modified Seldinger technique is also used nowadays, using image guidance, for various nonvascular procedures such as for aspiration and drainage of abscesses and collections in chest, abdomen and pelvis. Dr Charles T Dotter is credited with many new innovations in field of IR and considered father of IR. Charles T Dotter first published paper describing technique of angioplasty using Seldinger technique in Refs. First angioplasty was done accidentally by Charles T Dotter, when he crossed iliac artery stenosis, while performing aortogram in a patient with renal artery stenosis. He dilated iliac artery stenosis with coaxial dilators of size 8 and 12 F. This coaxial dilator was developed by Charles T Dotter, along with Bill Cook. First intentional angioplasty was done by Charles T Dotter along with his trainee Melvin Judkins in 1964, in an 82-year-old woman with femoral artery stenosis. Thereafter, Charles T Dotter popularized technique of percutaneous transluminal angioplasty (PTA) and trained many interventional radiologists. This ushered a new era, wherein Seldinger technique was used to access vessels, to diagnose and treat diseases endovascularly throughout human body. Charles T Dotter published his experience of opacifying coronary artery, by occluding aorta with balloon, followed by contrast injection. In 1959, Sones FM inadvertently did first coronary angiography and published his experience of cine-coronary arteriography. Thereafter, Charles T Dotter sent his trainee Melvin Judkins to learn technique of coronary catheterization from Sones FM. Melvin Judkins thereafter learned technique of cardiac catheterization and developed specialized catheters for cardiac catheterization, which were later known as “Judkins catheter”. In 1963, Fogarty TJ et al. published their technique for extraction of arterial emboli and thrombi using Fogarty balloon in Refs. However, these Fogarty balloons were inserted by surgical arteriotomy only. One person, whose name, we will like to mention specially here, is that of Andreas Grüentzig. Andreas Grüentzig had high regard for Charles T Dotter, for popularizing technique of PTA and he developed a balloon catheter in 1974, capable of dilating peripheral arteries. Andreas Grüentzig used to regularly conduct live training IR sessions, and Charles T Dotter was attendee in few of these training sessions. As a mark of respect, Andreas Grüentzig gifted a ring to Charles T Dotter, in one of his training sessions. It is due to efforts of Andreas Grüentzig that Charles T Dotter and his technique found wide acceptance among physicians, almost 15 years after Charles T Dotter did first PTA. Balloon angioplasty has now become the most frequently used treatment in vascular emergencies of the heart and the peripheral circulation. Charles T Dotter is credited with many new innovations, including development of rapid film changer and development of electronic switch for improved visualization of cardiac motion. Most important contribution of Charles T Dotter were his numerous public awareness lectures and publications, wherein he described his observations including findings in syphilitic aortitis, angiocardiography findings in myxoma, complications of angiocardiography, percutaneous catheterization using spring guide, flow-guided cardiac catheterization, fluoroscopic guidance in femoral artery puncture, coronary radiography and endarterectomy, use of stents, streptokinase in the treatment of thromboembolic disease, embolization using isobutyl 2-cyanoacrylate, description of coaxial technique and foreign body retrieval techniques. Dr Charles T Dotter was a very intelligent man, and at young age of 30, he became Faculty at Cornell Medical School. At age of 32 in 1952, he became Chairman of the School of Medicine, Department of Diagnostic Radiology, Oregon Health Sciences University, and he headed this department until his death in 1985. Here, I would like to highlight one incident. Dr Charles T Dotter published his experience, where he received a request for angiogram of left superficial femoral artery with instructions “Visualize but do not try to fix”. Thereafter, Dr Charles T Dotter did left femoral arteriogram. Then, he analyzed angiogram and found stenosis in both left superficial femoral artery and left deep femoral artery. Obeying physician’s instructions, he left superficial femoral artery and did angioplasty of left deep femoral artery. Charles T Dotter was fondly called “Crazy Charlie” by his friends and Europeans used the term “dottering” for describing technique popularized by Charles T Dotter. Charles T Dotter was in fact first one to realize true potential of IR, not only for diagnosis but also for patient treatment and management. In field of Nonvascular IR, one person whose name needs to be mentioned specifically is Constantin Cope. Constantine Cope collaborated with Cook Medical, and his innovations include development of retaining loop catheter, used in nephrostomy, biliary and abscess drainage catheters, micropuncture introducer set, gastrointestinal (GI) suture anchor set, Cope catheter introduction system and transitionless introducers. Technique of lymphangiography was first described in 1961 by Wallace S et al.. In 1964, Abrahms HI published his paper, in which he described that he could distinguish between malignant and normal vessels, based on angiographic findings. Until 1964, IR was considered only a diagnostic speciality, with role of IR only as angiographer to diagnose diseases. But in 1964, role of IR changed from being a diagnostic angiographer to therapeutic IR. Baum S et al. described clinical use of selective abdominal arteriography with its value in diagnosis of intraabdominal bleeding. IR became important part of team, to manage patients with GI bleeding. They started taking hospital rounds, doing emergency call duty and writing orders in patients charts. Selective vasopressin infusion was described by Nusbaum M et al. in 1968, wherein they described control of portal hypertension by selective mesenteric arterial infusion of vasopressin. Until this time, superselective catheterization was not possible, and to control GI bleeding, either vasopressin or embolic agents were used, with inherent risk of infarction of gut due to proximal embolization. Until 1970, Seldinger technique was well accepted in Europe. But in the United States, this technique was well accepted only in 1980. In 1980, 17 articles on transluminal angioplasty were published in a single issue of American Journal of Roentgenology. Development of IR as subspeciality of radiology was gradual between 1970 and 1980. Developments which took place in this era (1970–80) include development of occlusive coils, gelfoam embolization, selective arterial embolization for GI bleeding, embolization for pelvic trauma, development of balloon catheter by Andreas Grüentzig to dilate stenosis in arteries throughout body, selective arterial thrombolysis for arterial occlusions, transhepatic embolization for variceal bleeding, embolization technique for pulmonary arteriovenous malformations and varicoceles, bland and chemoembolization for treatment of hepatocellular carcinoma and liver metastasis and cryoablation for liver tumour. Developments which took place in field of IR in between 1980 and 2000 include embolization techniques for splenic trauma, biliary drainage procedures, percutaneous genitourinary procedures, transjugular intrahepatic portosystemic shunt (TIPS), development of balloon-expandable and self-expandable stents and stent grafts, percutaneous extraction of gallbladder and common bile duct stones, radiofrequency ablation for liver tumours and soft-tissue tumours, treatment of bone and kidney tumours by embolization, uterine artery embolization, abdominal aortic stent grafts, balloon-expandable coronary stents, intraarterial delivery of tumour killing viruses and gene therapy vectors to the liver, percutaneous delivery of pancreatic islet cells to the liver for transplantation to treat diabetes, hybrid imaging, endovenous laser ablation procedure to treat varicose veins and venous disease. IR as subspecialty started in India in 1970, with few radiologists practising as angiographers and doing diagnostic angiograms. Between 1980 and 1990, few luminaries of radiology started working as interventional radiologists. These interventional radiologists used to make catheters themselves, using tubes of different sizes. Malik N et al. published paper on transcatheter arterial embolization of renal tumours in 1981. In 1982, Rao VRK et al. published paper on retrieval of a broken catheter from the aorta without operation, using a self-made snare. In 1992, Roy S et al. published their experience for oesophageal stenting using a self-expandable metallic stent. In 1994, Roy S et al. published their experience of developing hybrid Gianturco expandable stent, which was modified handmade stent and less expensive than other metallic stent, available at that time. In same year, Sharma S et al. published their experience of treating life-threatening arterial haemorrhage using homemade steel coils. Most important development in history of IR in India is establishment of IR Society by name of Indian Society of Vascular and Interventional Radiology (ISVIR) IN 1997. 28 founding members from 18 institutions came together to form ISVIR. Now, ISVIR has more than 700 registered members, with Journal of Clinical Interventional Radiology as official journal of ISVIR. ISVIR has main aim of promoting IR in India, with training opportunities for residents and fellows, in the form of courses such as DM IR, which is a dedicated 3-year course. Many institutions are also running 1-year postdoctoral certificate course (PDCC) in IR. National Board of Examinations has also started postdoctoral DNB in Endovascular and Interventional Radiology, which is a 3-year course. IR has developed as a distinct specialty, with high regard from contemporary clinicians, due to hard work and foresight of many legendry luminaries, names of few of which we have mentioned in this chapter. However, to sustain this progress, interventional radiologists have to work hard and hone new skills, and aggressively market themselves. Most important is availability of IR practitioner, anytime in 24 h. IR practitioners need to audit their work regularly and, based on audit results, take steps to improve their practice. Only then, IR practitioner can dream of becoming an equal partner in patient care, with other clinical colleagues. IR is a subspecialty of radiology in which we perform minimally invasive procedures under image guidance. IR is divided broadly into two subgroups: vascular interventions and nonvascular interventions. In this chapter, we will be discussing basic principles of nonvascular interventions. Nonvascular interventions refer to use of principles of IR, to diagnose and treat diseases, without accessing vessels. This encompasses the following procedures: Technique described by Dr Svan Ivar Seldinger was double-wall puncture technique, wherein both walls of vessel were punctured with access needle and then access needle was slowly withdrawn, until gush of blood was seen. This gush of blood was proof of access needle being within vessel lumen. Thereafter, guidewire was introduced into vessel lumen through needle, and needle was withdrawn over guidewire. Compression was applied at point where access needle had first pierced vessel wall. Thereafter, catheter was secured in vessel lumen over guidewire. Nowadays, we use “modified Seldinger technique”. In modified Seldinger technique, we use ultrasound (USG) guidance to access vessel lumen and only single-wall puncture is done. Advantage of modified Seldinger technique is that we use inplane view and we know precisely, when access needle has entered vessel lumen, and this ultimately results in minimizing the blood loss and reduces complications rate significantly. Modified Seldinger technique is also used nowadays, using image guidance, for nonvascular procedures such as for aspiration and drainage of abscesses and collections in chest, abdomen and pelvis. The results of these image guiding procedures are very gratifying, as patients show dramatic improvement after placement of drainage catheters. However, there is slight change in modified Seldinger technique, used for nonvascular interventions. First of all, under image guidance, access needle is passed through safe path into abscess/collection, and once needle is inside abscess/collection, we aspirate fluid/pus and send this fluid/pus for analysis. Thereafter, guidewire is passed into collection/abscess. Then, access needle is removed over guidewire, and dilator is used over guidewire, to spread subcutaneous soft tissue, for easy passage of catheters into abscess/collection. One important point regarding use of dilators is that we use serial dilators, depending upon which size of catheters, we want to use. So, if we want to place 12F catheter into abscess/collection, then we will first start with 8F dilator and gradually upgrade dilatation to 10F and finally 12F. Secondly, we have to dilate over guidewire in same direction in which we had placed our access needle. This step is necessary to minimize trauma to adjoining structures. Once path has been adequately dilated, we remove dilator over guidewire and finally place our catheter in abscess/collection. We have to ensure that all side holes of catheter are inside abscess/collection. To reduce chance of bleeding, 21 G access needle can be used along with 0.018″ guidewire. Once guidewire is inside collection/abscess, guidewire exchange can be done to 0.035″ guidewire, and finally, desired catheter can be placed inside abscess/collection. Whenever we receive requisition for any image-guided percutaneous nonvascular IR procedure, we have to first consider whether this procedure needs to be done or not, as per guidelines. For instance, if we receive request for drainage of asymptomatic pseudocyst, then we need to talk with referring clinician and patient, as this procedure is unwarranted according to guidelines. Thereafter, we need to inform the patient about all other options that the patient has for management of this condition, so that the patient can give informed consent. We should alleviate patient’s anxiety, by answering all questions and clearing any doubt, ideally in IR clinic. We should have look at patient’s all previous investigations, including imaging and laboratory investigations, specially PT/INR. Ideally, INR should be less than 1.5. If INR is more than 1.5, then we can correct INR using fresh frozen plasma (FFP) and vitamin K (vitK). Ideally, haemoglobin level should be more than 9 g per decilitre. But, in emergency, we can do nonvascular IR procedure with the patient having haemoglobin level as low as 6 g per decilitre. But packed RBC should be transfused to the patient, if haemoglobin level is less than 6 g per decilitre. Serum potassium level should be less than 5.5 mill moles per litre. Then, we have to plan our procedure, depending on which modality we are seeing lesion properly or depending on which modality we can safely find path to reach abscess/collection. Most commonly, we use USG for image guidance. However, if collection/abscess is in pelvis/retroperitoneum/mediastinum, then we use computed tomography (CT) for safely accessing these abscess/collection. Few basic principles need to be followed, for accessing these abscess/collections: Nonvascular IR procedure can be done using three different techniques: Modified Seldinger technique has already been described in detail. But here we will like to highlight one important point that the shortest route is not always best route. For most of collections/abscess, we will be able to access using the shortest straight route, using safe passage. But, in case of some collections/abscesses such as subphrenic abscess, we may have to use longer tract, for safe passage. In subphrenic abscess, the shortest route will be posterior approach, through pleura, with inherent risk of pleural effusion/fistula. Safe passage for subphrenic abscess will be using anterior approach. However, using this approach, tract will be longer as compared with posterior approach. In tandem technique, we first access abscess/collection using small size needle. Once we have reached safely to target abscess/collection, then parallelly we place larger size needle to access abscess/collection. Usually, we use tandem technique, when we use single-step drainage catheter to drain abscess/collection using retroperitoneal or transgluteal approach. We use single-step procedure, for draining abscess/collection, when we have to use tract where lot of muscles need to be traversed, which makes the procedure painful, like transgluteal approach for drainage of pelvic collections. These single-step drainage catheters assembly contain from inside out: puncture needle, stiffener, dilator and drainage catheter. After instilling lot of local anaesthetic in drainage path, single-step assembly is safely placed using transgluteal approach, in pelvic abscess/collection. An important consideration while using transgluteal approach is to go as near sacrum as possible, and below level of piriformis muscle, to save sciatic nerve. If we use modified Seldinger technique for draining pelvic abscess/collection using transgluteal approach, then we have to do multiple steps of exchange over guidewire and each step is painful for patient, whereas in single-step procedure, patient will have pain only once. We remove drainage catheters, when drainage output is less than 10 mL per day, along with improvement in patient’s clinical condition. If drainage output remains stable or increased, along with improvement in patient’s clinical condition, then we should suspect fistula. If we detect fistula on fistulogram, then we have to drain these abscess or collection for prolonged period. In most cases, these abscess/collection with fistulous communication, will resolve after some time, after prolonged drainage. However, if these fistulous communications do not close after prolonged drainage, then we have to consider these scenarios: Many stalwarts have an important role to play in popularizing nonvascular IR procedures, starting from Svan Ivar Seldinger. Modified Seldinger technique allowed percutaneous placement of large size drains in abscesses/collection and that too without surgery. One IR whose name requires special mention is Cope C. Cope C introduced famous loop technique in 1980, which made percutaneously placed catheters stable in abscess/collection, due to locking loop at end of catheter. Cope C published technique of conversion from small (0.018″) to large (0.038″) guidewires in percutaneous drainage procedures in 1982. In 1983, Cope C published a paper on use of stiff fine needle guidewire for catheterization and drainage. In 1986, Cope C published a paper about suture anchor for visceral drainage. Sutural anchorage is important in procedures such as percutaneous gastrostomy/jejunostomy or percutaneous gallbladder stone extraction, to retract gastric/jejunal/gallbladder wall against abdominal wall, as this prevents invagination and helps in providing good seal, which in turn decreases chance of peritonitis due to leakage of gastric/jejunal/gallbladder contents. Cope C published a paper on needle endoscopy in special procedures in 1988. In 2001, Cope C published two important papers: first one related to percutaneous management of chronic pancreatic duct strictures and external fistulas with long-term results and second related to long-term patency of experimental magnetic compression gastroenteric anastomosis achieved with covered stents. Gerzof SG et al. published a paper on the role of ultrasound and CT guidance, for percutaneous catheter drainage of abdominal abscesses in 1979. Johnson RD et al. published a paper on percutaneous drainage of pyogenic liver abscesses in 1985. Percutaneous jejunostomy was first described in 1987. CT-guided drainage was published in 1991 by Van Sonnenberg E et al.. It is only due to contribution of pioneers in the field of IR that we have today reached stage, where we are ready to compete with clinicians from other specialty, be it vascular surgeons, cardiologists, interventional nephrologist, critical care specialists, pain specialists or other specialities. All this has become possible due to simultaneous development of high-quality imaging and availability of hardware, which we use for IR procedures. We have to learn tips and tricks of trade, specially how to increase our visibility to other specialities and general public. All interventional radiologists should have their specialty outpatient clinics, and all interventional radiologists should have patient admission rights with their own beds. This is basic minimum requirement to firmly root our specialty as serious competitor to other specialties. With our enormous publications, we have clearly demonstrated that percutaneous nonvascular interventions have results, which are equivalent or better than surgery. But, for the growth of IR as subspecialty, we have to continue hard work and become important member of team, responsible for patient care. This can only be achieved by honing our skills, be it regarding our technique or our knowledge about medicines, which we prescribe to our patients, before, during or after IR procedure. Angiography can be defined as imaging of blood vessels used to project the vascular details including details of the vessel wall and flow through the lumen. Conventionally the angiography is done by injecting intravascular contrast; however, magnetic resonance angiography can be performed with or without contrast medium. Angiography is used to study vessel anatomy, plaque characterization and assessment of stenosis, aneurysm or any abnormality of vessels. In addition, the cross-sectional imaging is also capable of studying the extraarterial anatomy. Broadly, we can divide the angiographic techniques into invasive and noninvasive techniques. The invasive technique includes digital subtraction angiography (DSA), whereas noninvasive techniques involve computed tomography angiography (CTA), magnetic resonance angiography (MRA) and colour Doppler (CD) imaging. In the modern era, there has been a great development in the noninvasive angiography techniques including CT, magnetic resonance imaging (MRI) and CD imaging. Where Doppler is limited to certain body parts, CTA and MRA techniques have proven highly reliable alternative to DSA. Both CTA and MRA can be used to detect arterial or venous disease in the whole body. The CTA and MRA can detect atherosclerotic and nonatherosclerotic causes of peripheral arterial diseases (PADs) such as vasculitis with great efficiency. DSA is a technique used for visualizing vessels by using a special fluoroscopic technique without interfering from the overlapping tissue. Here the radioopaque objects in view are subtracted digitally for accurate projection of vessels. DSA gives a clear view of vessels and uses less contrast dose. The modern equipment for DSA has various functions to reduce the dose and improve image quality and operate convenience. These include collimator, pulsed fluoroscopy, road map, mask, image enhancement, frame grab and last image hold, measurement and quantifications tools and the ability to change collimation without fluoroscopy. The DSA angiogram should always be performed after obtaining informed consent. Seldinger technique was described by Sven Ever Seldinger in 1953. The technique helps in obtaining the safe access in to blood vessel or any space. This is how conventional angiography started and IR evolved over a period of time. A needle is inserted in to the desired vessel. Guidewire is inserted when blood starts coming out. Then wire is sent within it over which the needle is removed with guidewire still in position. Finally, a sheath or a catheter is pushed over it and the wire is removed. Advantages: Cut is not required in this technique; hence, there is less pain, minimal trauma to the adjacent area and faster recovery. Uses: DSA, placement of drainage catheters, central lines, dialysis catheters (temporary or Permcath), PTBD, percutaneous gastrostomy/cholecystostomy, pacemaker lead and implantable cardioverter defibrillators. However, in case of risk to the life of the patient, angiogram can be performed with informed consent. Renal ischaemia secondary to contrast administration mainly induces toxicity at the level of the renal tubules, and the effect on glomerular function appears mild Contrast-induced nephropathy (CIN) is mainly observed in patients with preexisting renal compromise with or without diabetes mellitus; however, recovery from acute renal failure in diabetics with renal insufficiency following CIN is less with increased chances of having dependence on dialysis as compared with nondiabetics. Dehydration, excessive contrast and simultaneous intake of other nephrotoxic agent also contribute in the development of CIN. Heavy proteinuria and multiple myeloma also have a role in the development of CIN. The incidence of CIN in patients with normal kidney function is as low as 1%–2%. Definitive measures to reduce the chance of development of CIN following contrast administration are not conclusive; however, there appears to be a direct relation between contrast exposure time and development of CIN. Hence, one should consider minimizing contrast injection and maximize urine flow rate during and after angiogram to avoid CIN. Along with this limit, the use of contrast to less than 50–75 mL in patients with GFR of <20–30 mL/min. N-acetylcysteine (NAC) protects against CIN by scavenging reactive oxygen species. It has been observed that there is significant reduction in the levels of serum creatinine with the use of NAC and hydration. NAC should be administered in a dose of 1200 mg twice daily on the day of study and a day prior. Consensus panel recommends to take precautionary measures to avoid CIN in patients having eGFR <60 mL/min per 1.73 m2 as shown in the following figure:
1.13: Interventional radiology – basic principles
Introduction
History
Interventional radiology in India
Basics of nonvascular interventions
Prerequisites
Procedure
When to remove drainage catheters
Brief history for nonvascular interventional procedures
Basics of angiography
Digital subtraction angiography
Digital subtraction angiography technique
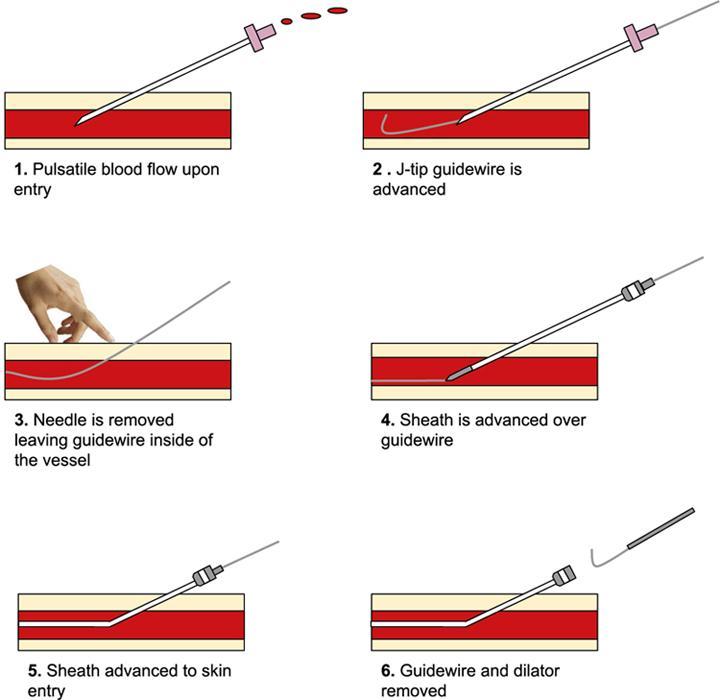
Seldinger technique
Equipment
Technique
Complication
Indications of digital subtraction angiography
Head and neck
Peripheral vascular
Cardiothoracic
Gastrointestinal
Others
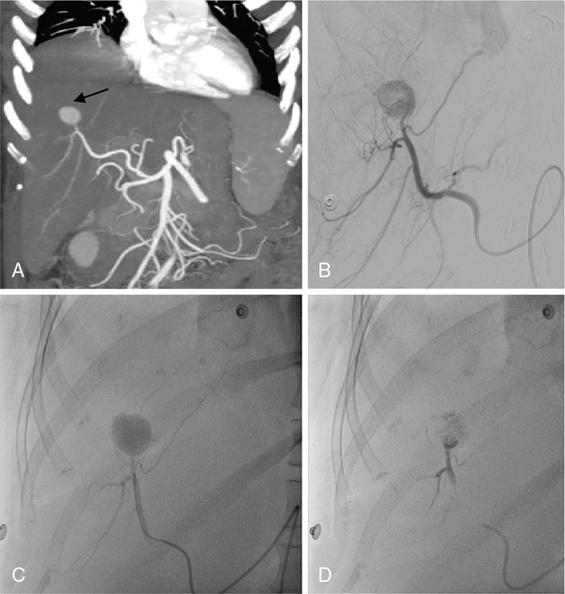
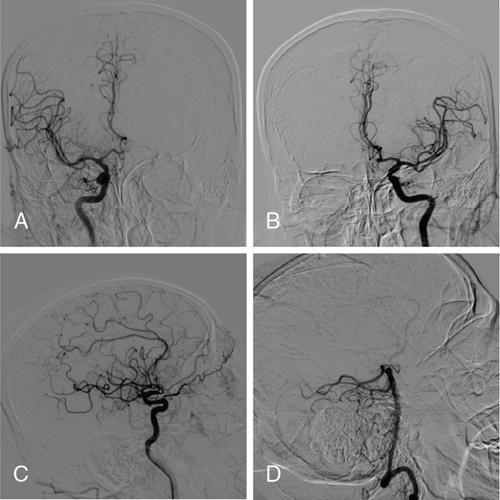
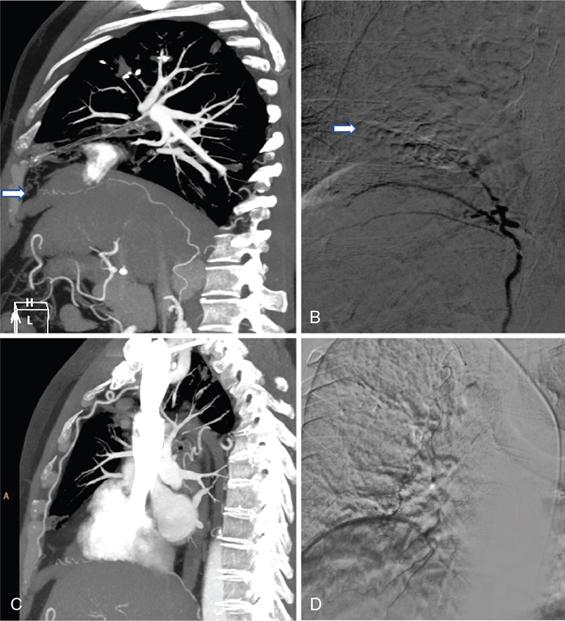
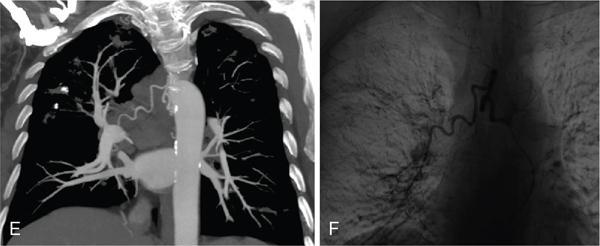
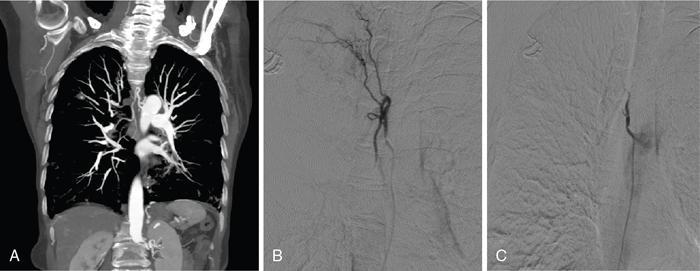
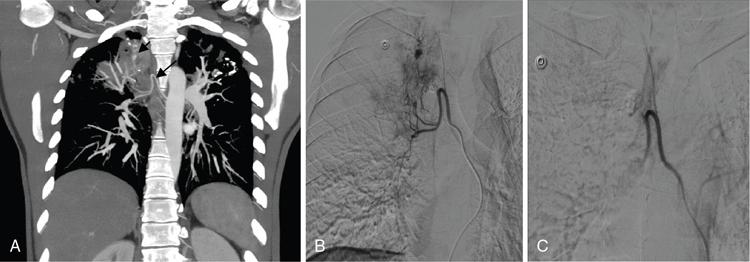
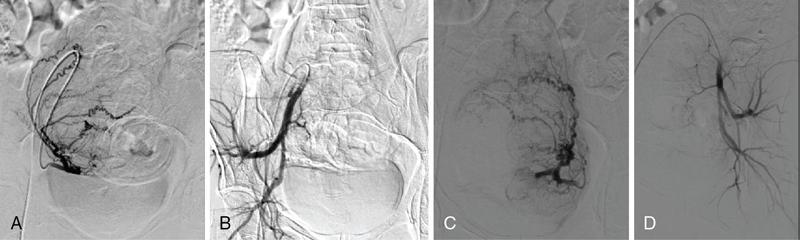
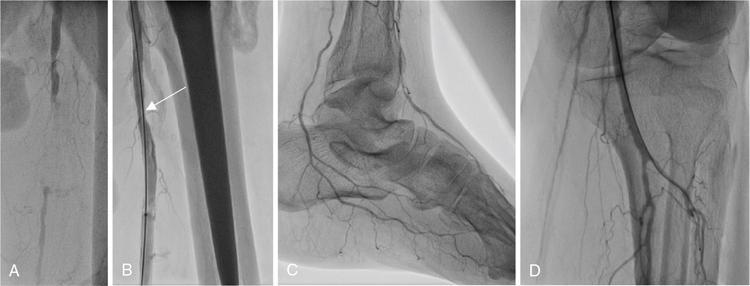
Contraindication
Complications
Major complications
Minor complications
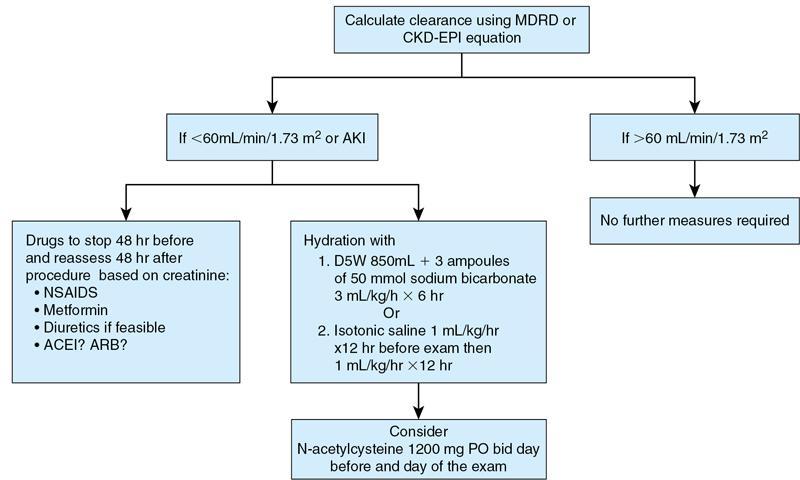
Contrast-induced nephropathy
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree