Introduction
Gustav von Schulthess
The rapid adoption of positron emission tomography (PET) within the framework of the combined use of PET and computed tomography (CT), or PET-CT, has been astounding. The main reason for this is that the imaging world has seen the limitations of anatomy-based imaging and is now prepared to venture into clinical molecular imaging methods. It should be remembered that nuclear medicine has included clinical molecular imaging right from its birth, in the application of radioactive iodine in the diagnosis and therapy of thyroid disease. The great advantage of fluorodeoxyglucose (FDG)-PET and possibly PET based on other tracers, as well as of single-photon emission computed tomography (SPECT), is that in many instances the lesions are shown with almost no interference from normal anatomic structures. As a consequence, the lesions are very conspicuous, but frequently the morphological information needed to properly locate a lesion is missing. This critical feature of nuclear medicine methods is a result of the “molecular spy” characteristics of most radiopharmaceuticals. The detection systems used are exquisitely sensitive and can locate nano- to picomolar concentrations of tracers, which is the reason nuclear methods are so uniquely useful for molecular imaging. Relevant sensitivities to extrinsic tracer concentration and current spatial resolution ranges are given in Table 1.1. The data show that nuclear imaging methods and anatomic imaging modalities are highly complementary, the former providing high contrast sensitivity, the latter spatial and temporal resolution.
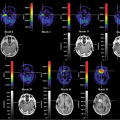
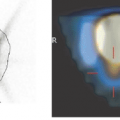
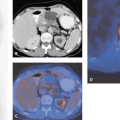
Integrated imaging with PET-CT or SPECT-CT in many ways offers the best of two worlds. This author, in many of his talks, has illustrated this using an example from Richard Scarry’s children’s books. Many readers may recall that the task in these books is to identify “Lowly the Worm,” a little creature placed among a variety of animals and structures depicted on the large pages (Fig. 1.1). This is akin to what a radiologist has to do many times a day when evaluating multiples of 200 or more CT or magnetic resonance (MR) sections to find tumor manifestations in patients who were scanned for staging or therapy follow-up. Tumor PET imaging and SPECT imaging are very different from anatomic imaging: the lesion is readily seen, but the “anatomic” backdrop may be minimal, as shown in Figure 1.2. Thus, precise lesion localization within the anatomic context, which frequently is critical, may be impossible in PET or SPECT. With a combination of the two modalities, the lesions can be identified by virtue of the high sensitivity, specificity, and contrast in PET or SPECT images (represented as the “Lowly the Worm”), and the gray scale background provided by the “hardware coregistered” CT images permits precise lesion localization (Fig. 1.3). It has been well documented over the past years that such multimodality imaging substantially increases the sensitivity and specificity of PET and SPECT examinations and thus make the integrated approach more accurate than the individual modalities by themselves.
Setting the Stage
PET and SPECT imaging are molecular imaging methods. If we define molecular imaging methods narrowly, as methods that permit the measurement of some molecular biological process in vivo, imaging with FDG and some of the SPECT tracers can certainly be classified as molecular imaging. In this narrower definition, nuclear medicine currently encompasses the only clinically useful and utilized molecular imaging methods. If we define molecular imaging more broadly to incorporate the measurement of other functional information, such as perfusion and blood pool imaging, almost all of nuclear medicine qualifies as molecular imaging. In a way, nuclear medicine imaging comprises a multitude of “imaging modalities,” because these differ from other imaging methods in being almost entirely defined by the radiopharmaceuticals used: it is not MRI or CT with contrast, it is 18F fluorodeoxyglucose PET (FDG-PET) or 111In indium octreotide SPECT. Thus, imaging with each radiopharmaceutical represents a different “imaging modality.” Very different normal structures are visible with FDG-PET, lung perfusion imaging, and myocardial perfusion imaging, in addition to the molecular imaging aspects. This is because nothing is seen on a nuclear scan if no radiopharmaceutical is introduced into the patient, and after injection only pathological structures and normal structures labeled by the specific radiopharmaceuticals are visible. A book on PET or SPECT imaging, therefore, also has to define which radiopharmaceuticals are being discussed. Only when we have defined the examinations of interest can we discuss how imaging systems should be optimally designed for them.
Table 1.1 Sensitivities to Extrinsic Tracer Concentration and Spatial Resolution Ranges | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
In PET, and this is the main topic of this book, identifying the key radiopharmaceutical is easy: it is 18F fluorodeoxyglucose. There would be no clinical PET today without the FDG molecule. The reason for the success of FDG is that it accumulates in pathological tissues, notably tumor tissues, which leads to a high lesion-to-background ratio and makes the lesions very conspicuous. This has made FDG-PET a primary method for tumor staging despite the fact that FDG accumulates not only in tumors but also in activated inflammatory cells such as granulocytes and macrophages. The biochemical properties of FDG, combined with the relatively high spatial resolution of state-of-the-art PET scanners, is responsible for the high clinical utility of FDG-PET in tumor staging. Although critics complain about the lack of specificity of FDG, its accumulation in tissues is surely more specific than the accumulation of extracellular fluid agents, such as most iodinated contrast agents and gadolinium chelates used for contrast enhancement in CT and MRI.
There are some other positron-emitting radiopharmaceuticals in clinical use, most notably labeled water (H215O), 13N ammonia (13NH3), and rubidium-82 (82Rb) for brain and heart perfusion imaging. In the last years, various other 18F-based tracers have received increasing attention, such as choline derivatives, F-ethyl-tyrosine and F-thymidine. Gallium-68 (68Ga) DOTATOC and other octreotide analogs have also been clinically evaluated. Although the use of 15O and 13N is precluded without an onsite cyclotron, the 18F-based and generator-based compounds may become more widespread in clinical practice in the coming years. All these tracers are discussed in various detail in this book. The clinical utility of choline derivatives and labeled octreotide analogs currently need to be established.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree