Chapter 15 Low back pain
Definitions
Back pain
In an effort to standardise the use of terms and to set standards of diagnostic practice, the International Association for the Study of Pain (IASP) published the second edition of its taxonomy.1 This document provides definitions of clinical terms used to describe pain and sets criteria for the diagnosis of specific entities.
With respect to presenting complaints, the taxonomy defines spinal pain topographically. It recognises ‘lumbar spinal pain’ and ‘sacral spinal pain’.1 Lumbar spinal pain is defined as pain perceived within a region bounded laterally by the lateral borders of the erector spinae, superiorly by an imaginary transverse line through the T12 spinous process, and inferiorly by a line through the S1 spinous process. Sacral spinal pain is defined as pain perceived within a region overlying the sacrum, bounded laterally by imaginary vertical lines through the posterior superior and posterior inferior iliac spines, superiorly by a transverse line through the S1 spinous process, and inferiorly by a transverse line through the posterior sacrococcygeal joints.
Somatic pain
Somatic pain is pain that results from noxious stimulation of one of the musculoskeletal components of the body.1,2 Neurophysiologically, the essential feature is that it arises as a result of the stimulation of nerve endings in a bone, ligament, joint or muscle.
Referred pain
Referred pain is pain perceived in a region innervated by nerves other than those that innervate the actual source of pain.1,2 As such, referred pain may be perceived in areas relatively remote from the source of pain, but often the distinction is blurred when the regions of local pain and referred pain are contiguous and the two pains appear to be confluent. A knowledge of the innervation of the affected regions, however, serves to make the distinction.
The physiological basis for referred pain is convergence.1 Within the spinal cord and in the thalamus, sensory neurones that subtend different peripheral sites converge onto common neurones that relay to higher centres. In the absence of any additional sensory information, the brain is unable to determine whether activity in the common neurone was initiated by one or the other of its peripheral inputs.
Clinically, the characteristic features of somatic referred pain are that it is perceived deeply, it is diffuse and hard to localise and it is aching in quality.2 Physiologically, the critical feature is that it is evoked by the stimulation of nerve endings in the structure that is the primary source of pain. The sensory nerves that innervate the region of referred pain are not activated by the primary stimulus, nor do they convey the referred pain. Referred pain occurs because of a misperception of the origin of the signal which reaches the brain by a convergent sensory pathway. These features underlie the distinction between somatic referred pain and radicular pain.
Radiculopathy
Radiculopathy is a neurological condition in which conduction is blocked in the axons of a spinal nerve or its roots.1,2 Conduction block in sensory axons results in numbness; conduction block in motor axons results in weakness. Radiculopathy can be caused by compression or ischaemia of the affected axons. Systematically, these causes are outlined in Table 15.1.
Table 15.1 The causes of radiculopathy
| Condition | Cause |
|---|---|
| Foraminal stenosis | Vertical subluxation of vertebrae3,4 |
| Osteophytes from disc5–10 | |
| Osteophytes from zygapophysial joint11–13 | |
| Buckled ligamentum flavum14 | |
| Cyst of ligamentum flavum15 | |
| Slipped inferior articular epiphysis16 | |
| Ganglion17–21 | |
| Synovial tumour22 | |
| Infections and tumours of vertebrae11,12 | |
| Paget’s disease11,23 | |
| Zygapophysial lipoma24,25 | |
| Epidural disorders | Lipoma; angioma11,12 |
| Infections11 | |
| Meningeal disorders | Cysts of the nerve root sleeve11,26–29 |
| Intradural ossification30 | |
| Neurological disorders | Diabetes31 |
| Cysts and tumours11 | |
| Infections; tabes dorsalis11 | |
| Disc herniation |
Radicular pain
Radicular pain is pain that arises as a result of irritation of a spinal nerve or its roots.1,2 Radicular pain may be associated with radiculopathy but not necessarily so. Radicular pain may occur without radiculopathy and radiculopathy may occur without radicular pain.
It was once believed that radicular pain was due to compression of nerve roots. This is patently untrue. Neurophysiological experiments have shown that compression of a nerve root does not evoke nociceptive activity; at best it evokes a brief discharge at the time of application of the compression stimulus, but thereafter the root becomes silent.32,33 It is only when dorsal root ganglia are compressed that sustained activity is evoked, but this activity occurs not only in nociceptive axons but also in β fibres.32,33 The sensation, therefore, must be more than just pain. This is borne out in clinical experiments.
Clinical experiments have shown that compressing normal nerve roots with urinary catheters evokes paraesthesia and numbness but not pain.34 Similarly, traction on a normal nerve root does not evoke pain.35 It is only when previously damaged nerve roots are squeezed by forceps36 or pulled with sutures,35 or when nerve roots are stimulated electrically,37 that a characteristic pain is evoked. The pain is shooting or lancinating in quality and travels down the lower limb along a band no more than 50 mm (2 inches) wide.35
In the face of this evidence, it is pertinent to consider the term ‘sciatica’. The implied basis for sciatica is nerve root compression or nerve root irritation, whereupon sciatica must be considered a form of radicular pain. However, the available physiological evidence dictates that radicular pain has a characteristic quality and distribution, and therefore the term sciatica should be restricted to this type of pain in the lower limb. The only type of pain that has ever been produced experimentally by stimulating nerve roots is shooting pain in a band-like distribution.2 There is no physiological evidence that constant, deep aching pain in the lower limb arises from nerve root irritation. This latter type of pain constitutes somatic referred pain and there is no justification for misrepresenting it as ‘sciatica’, or for inferring that it arises from nerve root irritation. Moreover, there is no evidence that back pain can be caused by nerve root irritation, especially in the absence of neurological signs indicative of nerve root irritation. Indeed, on clinical grounds it has been estimated that fewer than 30%, and perhaps as few as 5% or 1%, of presentations of low back pain are associated with nerve root irritation due to disc herniation.38–40 A formal survey in the USA established that no more than 12% of patients with low back pain had any clinical evidence of disc herniation.41
Disc herniation is the single most common cause of radicular pain, and there is increasing evidence that this condition causes pain by mechanisms other than simple compression. The evidence against compression is twofold. On myelography, CT or MRI, individuals can exhibit root compression by disc herniation but have no symptoms.42–45 Conversely, patients previously symptomatic can still exhibit root compression on medical imaging despite resolution of their symptoms.46,47 These observations indicate that some factor in addition to, or quite apart from, root compression operates to produce symptoms. The current evidence implicates some form of inflammation.
Inflammation was implicated initially on the grounds that surgeons have often seen signs of nerve root inflammation when operating on herniated discs.48,49 Some early post-mortem studies reported signs of inflammation around nerve roots obtained at autopsy,50 but others found no such signs.51,52 Subsequently, a variety of studies suggested that nuclear material was inflammatory49,53–55 and perhaps capable of eliciting an autoimmune response.56–62 However, clinical studies failed to reveal features of a classic autoimmune diathesis in patients with prolapsed discs.63 Nevertheless, belief in some form of inflammation has persisted and has been explored.
In animal studies, compression of lumbar nerve roots causes oedema and increased intraneural pressure,64,65 and application of nucleus pulposus to nerve roots induces inflammatory changes in the form of increased vascular permeability, oedema, and intravascular coagulation.66–69 The inflammation damages the nerve roots, blocks nerve conduction,67,70–73 and produces hyperalgesia and pain behaviour.74–76 The mediators of this inflammatory response are phospholipase A2, nitric oxide and tumour necrosis factor alpha (TNF-α).70,75–82
Studies in human patients have shown that herniated disc material attracts macrophages, fibroblasts and lymphocytes83–90; and that a variety of inflammatory chemicals are produced by these cells or the disc material itself. These chemicals include: phospholipase A2;91,92 metalloproteinases;93,94 prostaglandin E2;89,93,94 leukotriene B4 and thrombaxane;95 nitric oxide;80,81,93,94 interleukin 8;96,97 interleukin 12 and interferon γ;88 and TNF-α.96 The disc material stimulates the production of IgM and IgG antibodies,98 particularly to the glyco-sphingolipid of the nerve roots.99 These inflammatory changes are more pronounced in patients in whom disc material has penetrated the anulus fibrosus and posterior longitudinal ligament, i.e. when it has become exposed in the epidural space.
The evidence is, thus, abundant that disc material evokes a chemical inflammation. The resulting nerve root oedema causes conduction block and the features of radiculopathy. Ectopic impulses generated in the dorsal root ganglia are responsible for the radicular pain,100–102 and are probably produced by ischaemia.65
Back pain
Postulates
1 The structure should have a nerve supply, for without access to the nervous system it could not evoke pain.
2 The structure should be capable of causing pain similar to that seen clinically. Ideally, this should be demonstrated in normal volunteers, for inferences drawn from clinical studies may be compromised by observer bias or poor patient reliability.
3 The structure should be susceptible to diseases or injuries that are known to be painful. Ideally, such disorders should be evident upon investigation of the patient but this may not always be possible. Certain conditions may not be detectable using currently available imaging techniques, whereupon the next line of evidence stems from post-mortem studies or biomechanical studies which can provide at least prima facie evidence of the types of disorders or injuries that might affect the structure.
4 The structure should have been shown to be a source of pain in patients, using diagnostic techniques of known reliability and validity. From such data, a measure of the prevalence of the condition in question can be obtained to indicate whether the condition is a rarity or oddity, or a common cause of back pain.
Sources of back pain
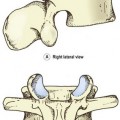
Vertebrae
There is no doubt that the vertebral bodies of the lumbar vertebrae are innervated.103–105 Nerve fibres, derived from the plexuses of the anterior longitudinal ligament and posterior longitudinal ligament, supply the periosteum of the bones and penetrate deep into the vertebral bodies where they provide a possible substrate for bone pain. However, it is not known whether the intraosseous nerves are exclusively vascular (either vasomotor or vasosensitive) or whether the bone of the vertebral body itself receives a sensory innervation.
For understandable logistic reasons, no experiments have demonstrated whether back pain can be evoked directly from bone in the vertebral body, but like periosteum in general106 the vertebral periosteum is clearly pain sensitive. Needling the periosteum in the course of procedures such as lumbar sympathetic blocks is regularly associated with pain.
The vertebral body may be affected by painful, metabolic bone diseases such as Paget’s disease107 or osteitis fibrosa,108 and it may be the site of primary or secondary tumours109,110 or infections.111,112 There is no dispute that such conditions can be painful, but how they actually cause pain is not known.
Fractures of the vertebral body may or may not be painful,113,114 and it is difficult to determine whether the pain stems from the fracture itself or arises from abnormal stresses applied to adjacent joints, muscles or ligaments as a result of the accompanying deformity. There is no evidence that fractures, anywhere in the body, are intrinsically painful, especially when stable. On the other hand, tissue deformation as a result of post-traumatic haematoma or oedema is readily viewed as a potent source of pain, particularly if this causes distension or inflammation of the periosteum. Such a model conveniently explains why acute vertebral fractures might be painful, but why old or healed fractures are not. Surrounding tissue swelling would be expected early in the history of a fracture, but in due course would subside. Persistent pain following a healed vertebral body fracture suggests a source beyond the fracture site and probably secondary to the resultant deformity.
The most common disease that affects lumbar vertebral bodies is osteoporosis but there is no evidence that this condition is painful, in the absence of fracture. The temptation is to infer that pain could arise directly from stresses applied to the weakened vertebral body, or that microfractures mechanically irritate perivascular sensory nerves within the vertebral spongiosa115 but in the absence of even remote evidence about the physiology of bone pain, such explanations are purely speculative.
Citing the literature on vertebroplasty and its claims of success, some authors have argued that the pain of osteoporosis fractures is mediated by intraosseous nerves, and is relieved by a neurolytic effect of cement injected into the fractured vertebral body.116 However, this argument is circular because it presupposes that the relief of pain reported from vertebroplasty can be attributed to a local effect of treatment at the fracture site. Others dispute this argument on the grounds that nerve fibres in the vertebral body are too irregularly present and too sparse to explain the relief of pain by vertebroplasty.117 There is also evidence that the pain of osteoporotic fractures of vertebral arises, not from the affected vertebral body but from the posterior elements behind that vertebral body.118
A revolutionary, though now not new, concept concerning vertebral pain is that of intraosseous hypertension.119,120 The notion is that, if obstructed, intraosseous veins become distended and stimulate sensory nerves in their adventitia. The cause of obstruction is suggested to be bony sclerosis, such as that which occurs in spondylosis and which narrows the bony channels through which the veins pass. This hypothesis is consistent with the presence of perivascular nerves in the vertebral bodies and is analogous to the pain of congestive venous disorders of the lower limb. However, while this is an attractive theory for the pain of spondylosis, manometric studies of vertebral intraosseous pressure have been limited in number and have not compared symptomatic with asymptomatic individuals to provide convincing statistical evidence in support of the theory.119,120
Regardless of how they might cause pain, disorders of the lumbar vertebrae are relatively easy to diagnose; they are readily apparent on radiographs and other medical imaging. Their prevalence is not explicitly known but appears to be very low. Infections, tumours and fractures of the vertebral bodies are rare amongst patients presenting with back pain under the age of 50 years.121 Even in older patients they are uncommon.
Posterior elements
Kissing spines
The lumbar spinous processes may be affected by Baastrup’s disease,122 otherwise known as ‘kissing spines’.123 This arises as a result of excessive lumbar lordosis or extension injuries to the lumbar spine in which adjacent spinous processes clash and compress the intervening interspinous ligament. The resultant pathology is perhaps best described as a periostitis of the spinous processes or inflammation of the affected ligament. Given that the periosteum of the spinous processes and the interspinous space are innervated by the medial branches of the lumbar dorsal rami,124,105,125–127 it is understandable that such a condition would constitute a source of pain.
However, clinical studies suggest that this condition has been overrated. In one study, only 11 out of 64 patients with kissing spines responded to surgical excision of the lesion.128
Lamina impaction
A condition analogous to kissing spines can affect an inferior articular process. In some lumbar motion segments, extension is limited by impaction of an inferior articular process onto the lamina below (see Ch. 8). In such segments, repeated extension injuries can result in irritation of the periosteum of the lamina and, indeed, such lesions have been demonstrated at post-mortem.129–131 This disorder is attractive as an explanation for some forms of back pain affecting athletes such as gymnasts accustomed to excessive forceful extension, but no clinical studies have yet provided evidence for its occurrence in living, symptomatic patients.
Spondylolysis
Spondylolysis was originally considered to be a defect due to failure of union of two ossification centres in the vertebral lamina. Modern evidence, however, clearly shows that it is an acquired defect caused by fatigue fracture of the pars interarticularis.132–134 Anatomically, the defect is filled with fibrous scar tissue riddled with free nerve endings and nerve fibres containing calcitonin gene-related peptide, vasoactive intestinal peptide and neuropeptide Y.135,136 Nominally, therefore, it could be a source of pain.
However, pars defects are not necessarily painful. In a survey of radiographs of 32 600 asymptomatic individuals, a pars defect was present in 7.2%.137 Amongst 936 asymptomatic adults, a unilateral defect was present in 3% and a bilateral defect in a further 7%.138 The corresponding figures in patients with back pain were 0.3% and 9%, respectively. In children aged 6 years, the prevalence of a defect was found to be 4.4%, and rose to 6% by adulthood.139
The pars defect can be infiltrated with local anaesthetic under fluoroscopic control.140 Relief of pain constitutes prima facie evidence that the defect is the source of pain. However, precautions need to be taken to ensure that the local anaesthetic has not anaesthetised an adjacent structure (such as a zygapophysial joint, which might be an alternative source of pain) and to ensure that the response to a single diagnostic injection is not due to a placebo effect. Nevertheless, relief of pain following infiltration of a pars defect is a good predictor of successful outcome following fusion of the defect and failure to obtain relief predicts poor response to fusion.140
Bone scans are used by some practitioners to diagnose symptomatic pars fractures but the relationships between positive bone scans and either radiological evidence of a fracture or pain are imperfect.141 Bone scans are most useful before a fracture has actually occurred, when the scan detects the stress reaction in the pars interarticularis. Once fracture has occurred the scan may or may not be positive, and tends to be negative in patients with chronic pain.141
Muscles
The muscles of the lumbar spine are well innervated. Quadratus lumborum and psoas are supplied by branches of the lumbar ventral rami,142 while the back muscles are supplied by the dorsal rami.124 The intertransverse muscles are variously supplied by the dorsal rami and ventral rami.143
There is no question that the back muscles can be a source of back pain and somatic referred pain. This has been demonstrated in experiments on normal volunteers in whom the back muscles were stimulated with injections of hypertonic saline.144,145 These injections produced low back pain and various patterns of somatic referred pain in the gluteal region.
Sprain
Animal studies have shown that when muscles are forcibly stretched against contraction, they characteristically fail at the myotendinous junction.146–148 The resulting lesion would presumably evoke an inflammatory repair response, which is easily accepted as a source of pain. In the case of the back muscles, such lesions could be incurred during lateral flexion or combined flexion–rotation injuries of the trunk and would be associated with tenderness near the myotendinous junctions of the affected muscles. Some of these sites are superficial and accessible to clinical examination, but others are deep. Deep sites lie near the tips of the transverse and accessory processes of the lumbar vertebrae. Accessible myotendinous junctions lie just short of the ribs near the insertions of the iliocostalis lumborum.
Conspicuously, what is lacking in the case of back muscle injuries is any direct evidence of the responsible lesion. The only data that might be invoked come from experiments in animals in which the muscles of the limbs, not the back muscles, were studied. Biopsy data in humans or imaging data have not been published. For this reason a workshop sponsored by the American Academy of Orthopedic Surgeons and the National Institutes of Health was circumspect and inconclusive in its approach to the notion that sprained back muscles were a common cause of pain.149 However, the advent of MRI may now provide a suitable non-invasive tool with which traumatic lesions of the back muscles might be identified.150
Spasm
Although myotendinous tears might underlie cases of acute back pain stemming from muscle, it is more difficult to explain chronic back pain in terms of muscle pain. A popular belief is that of muscle ‘spasm’.151 The implication is that as a result of some postural abnormality, or secondary to some articular source of pain, muscles become chronically active and therefore painful. If it occurs, such pain can only be explained on the basis of ischaemia, but the greatest liability of this model of muscular pain is that the purported evidence for its existence is inconclusive.151 Electromyographic data are inconsistent, and it is unclear what so-called muscle spasm constitutes in objective physiological terms: whether it is tonic contraction or simply hyperreflexia. Further research data are required before this notion can become more acceptable, together with an explanation of whether the pain arises as a result of ischaemia, strain on the muscle attachments or some other mechanism.
Imbalance
Another concept is that of ‘muscle imbalance’.152 It is believed that aberrations in the balance of tone between postural and phasic muscles, or between flexors and extensors, can give rise to pain. In the case of the lumbar spine, the imbalance is said to occur between the trunk extensors and psoas major on the one hand, and the trunk flexors and hip extensors on the other. While attractive to some, this theory is without proper foundation. In the first instance, it is unclear how the imbalance comes to be painful: whether the pain arises from one or other of the muscles involved or whether the imbalance somehow stresses an underlying joint. Belief in the theory is founded on the clinical detection of muscle imbalance, but the reliability and validity of the techniques used have never been determined, and so-called muscle imbalances have been pronounced as abnormal without proper comparison to normal biological variation. Objective corroborating evidence is required before this theory attracts more widespread credibility.
Trigger points
Trigger points are tender areas occurring in muscle capable of producing local and referred pain. They are characterised by points of exquisite tenderness located within palpable bands of taut muscle fibres. Clinically, they are distinguished from tender areas by the presence of the palpable band of fibres, which, when snapped, elicits a localised twitch response in the muscle, and which when pressed reproduces the patient’s referred pain.153 The pain is relieved if the trigger point is injected with local anaesthetic.
Trigger points are believed to arise as a result of acute or chronic repetitive strain of the affected muscle,153 or ‘reflexly’ as a result of underlying joint disease.154 Histological and biochemical evidence as to the nature of trigger points are incomplete or inconclusive,153 but they are believed to represent areas of hypercontracted muscle cells that deplete local energy stores and impair the function of calcium pumps, thereby perpetuating the contraction.153–155 Pain is said to occur as a result of obstruction of local blood flow and the accumulation of algogenic metabolites such as bradykinin.153
Trigger points have been reported to affect the multifidus, longissimus and iliocostalis muscles,156 and the quadratus lumborum.157 However, it is not known how often they are a cause of back pain as the diagnostic criteria are so hard to satisfy.
For the diagnosis of trigger points in the iliocostalis and longissimus muscles, the kappa scores for two observers agreeing ranged from 0.35 to 0.46, which is less than satisfying.158 Similar scores obtain for trigger points in the quadratus lumborum and gluteus medius.159 It is only if the diagnostic criteria for trigger points are relaxed, to exclude palpable band and twitch response, that acceptable kappa scores are achieved,159 but this changes the diagnosis from one of ‘trigger point’ to one of ‘tender point’ in the muscle.
Without reliable criteria for diagnosis, it is not possible to estimate the prevalence of trigger points as a cause of back pain. If classic criteria are used strictly, trigger points seem to be uncommon.159 Tenderness, on the other hand, seems to be quite common but does not constitute a diagnosis.
Thoracolumbar fascia
Compartment syndrome
At its attachment to the supraspinous ligaments, the thoracolumbar fascia is well innervated.126,127,160 However, little is known about the innervation of its central portions. There is only a mention in one study that it contains nociceptive nerve endings.161 Nevertheless, it would appear that the fascia is appropriately innervated to be a source of pain if excessively stretched.
Since the thoracolumbar fascia encloses the back muscles, it forms a compartment surrounding them, and this has attracted the proposal that the back may be affected by a compartment syndrome.162,163 The concept is that, in susceptible patients, the back muscles swell during and following activity but their expansion is restricted by the thoracolumbar fascia. Pain presumably arises as a result of excessive strain in the fascia.
The clinical marker of such a compartment syndrome would be raised intracompartmental pressure, but clinical studies have yielded mixed results. In one study, a series of 12 patients was investigated for suspected compartment syndromes. Only one patient exhibited sustained, elevated pressures in the compartment on the side of pain.164 In another study, however, seven patients were identified whose compartment pressure rose above normal upon flexion and was associated with the onset of back pain; fasciotomy reportedly relieved their pain.165 However, while offering intriguing results, this study did not rigorously report the variance of pressures in the control group and other diagnostic groups who also exhibited raised pressures. Therefore, it is not evident how unique the feature of raised pressure is to compartment syndrome.
Fat herniation
The posterior layer of thoracolumbar fascia is fenestrated to allow the transmission of the cutaneous branches of the dorsal rami. These sites may be associated with painful herniations of fat.166–173 It is unclear exactly how these herniations actually cause pain, but reportedly the pain can be relieved by infiltrating the site with local anaesthetic. In this regard, painful fat herniations resemble trigger points, but are distinguished clinically from trigger points by their extramuscular, subcutaneous location. Their prevalence is unknown.
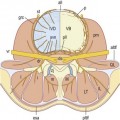
Dura mater
The dura mater is innervated by an extensive plexus derived from the lumbar sinuvertebral nerves. The plexus is dense over the ventral aspect of the dural sac and around the nerve root sleeves, but posterolaterally the innervation is sparse, and is entirely lacking over the posterior aspect of the dural sac.103,174 Clinical experiments have shown that the dura is sensitive both to mechanical and chemical stimulation.35,175 In both cases, stimulation invokes back pain and somatic referred pain into the buttock. This raises the possibility that dural irritation could be a source of back pain.
Back pain is well known in the context of neurological diseases in which the dura mater becomes inflamed in response to intrathecal blood or infection.176 This establishes that the dura can be a source of back pain. Whether it is or not, in the context of musculoskeletal diseases, is subject to conjecture.
Since it is known that disc herniation can elicit a chemical inflammation of the nerve roots and perineurial tissues,55,67,68,70,72 and that disc material contains high concentrations of phospholipase A2,91 which is highly inflammatory,92 it seems reasonable to expect that the dural sleeve of the nerve roots could be irritated chemically by this inflammation. Such irritation would elicit somatic pain, perhaps with referred pain, in addition to, and quite apart from, any pain stemming from the inflamed nerve roots. This conjecture raises the spectre that what has been traditionally interpreted as ‘root pain’ associated with disc herniation may not be purely radicular pain but a mixture of radicular and dural pain. However, no studies have yet ventured to dissect dural pain from radicular pain in cases of disc herniation.
In a similar vein, it has been proposed that the normally occurring epidural ligaments can tether nerve roots and be a source of somatic pain superimposed on radicular pain.177 However, as with dural ‘adhesions’ appropriate clinicopathological correlations have yet to be demonstrated.
Seductive evidence for dural pain comes from neurosurgical studies that report relief of post-laminectomy pain following resection of the nerves to the dura sleeve of the symptomatic nerve root.178,179 Ostensibly, the pain was due to stimulation of the nerves by fibrosis of the dura.
Epidural plexus
The epidural veins are innervated by the lumbar sinuvertebral nerves103,180 and are therefore a possible source of pain. Presumably, pain could occur if these veins became distended when flow through them was obstructed by lesions such as massive disc herniation or spinal stenosis. However, circumstantial evidence of this concept has been provided in only one published study181 and the concept has not otherwise been further explored.
Ligaments
The ligamentum flavum is poorly innervated104,125,126,182,183 and is therefore unlikely to be a source of pain. Furthermore, there are no known lesions that affect the ligamentum flavum that could render it painful, and because the ligament is elastic, it is not susceptible to sprain. It has a distensibility far in excess of that of the posterior longitudinal ligament and other collagenous ligaments of the lumbar spine.184
The so-called supraspinous ligament has been shown to consist of collagen fibres derived from the thoracolumbar fascia, the erector spinae aponeurosis and the tendons of multifidus.185,186 Technically, it is therefore a raphe rather than a ligament, but the most decisive evidence against the supraspinous ligament being a source of back pain at L4 and L5 (the most common location of low back pain) is that the ligament is totally lacking. It is consistently absent at L5, frequently so at L4, and even at L3 it is poorly developed and irregularly present.186
The posterior longitudinal ligament is innervated by the sinuvertebral nerves, and the anterior longitudinal ligament by fibres from the lumbar sympathetic trunk and grey rami communicantes.103,180,187 Reports that probing the back of a lumbar disc at operation under local anaesthetic reproduces the patient’s back pain,188,189 engender the belief that the pain naturally stems from the overlying posterior longitudinal ligaments. However, the posterior longitudinal ligament blends intimately with the anulus fibrosus of the intervertebral disc at each segmental level. Anatomically the longitudinal ligaments are not separable from the anulus fibrosus other than at a microscopic level. It is therefore not legitimate to consider disorders of the ligaments separately from those of the anulus fibrosus (see below).
Interspinous ligaments
The interspinous ligaments receive an innervation from the medial branches of the lumbar dorsal rami,44,105,124–127 and experimental stimulation of the interspinous ligament produces low back pain and referred pain in the lower limbs.190–192 This renders the interspinous ligament as an attractive source of low back pain.
Post-mortem studies have shown that the interspinous ligaments are frequently ‘degenerated’ in their central portions,186 but it is not known whether or not such lesions are painful. Otherwise, it is conceivable that interspinous ligaments might be subject to strain following excessive flexion of lumbar motion segments, but evidence of this is currently still lacking, even by way of comparing clinical history with the presence of midline interspinous tenderness and relief of pain following infiltration with local anaesthetic.
Clinical studies of the prevalence of interspinous ligament sprain are sobering. Steindler and Luck193 reported that in a heterogeneous population of 145 patients, 13 obtained complete relief of their pain following anaesthetisation of interspinous ligaments, suggesting a prevalence of less than 10%. A more recent audit of the experience of a musculoskeletal general practice found only 10 patients in a series of 230 whose pain could be relieved by anaesthetising an interspinous ligament.194 Since these injections were not controlled, the observed prevalence of 4% must be construed as a best-case estimate.
Iliolumbar ligament
The iliolumbar ligament has not explicitly been shown to have an innervation but presumably it is innervated by the dorsal rami or ventral rami of the L4 and L5 spinal nerves. Biomechanically, the iliolumbar ligament serves to resist flexion, rotation and lateral bending of the L5 vertebra,195–197 and could therefore be liable to strain during such movements. However, the evidence implicating the iliolumbar ligament as a source of back pain is inconclusive.
Some investigators have regarded tenderness over the posterior superior iliac spine as a sign of iliolumbar ligament sprain198 but this is hard to credit, for the ligament lies anterior to the ilium and is buried by the mass of the erector spinae and multifidus. Consequently, tenderness in this region cannot be explicitly ascribed to the iliolumbar ligament. Some have claimed to have relieved back pain by infiltrating the iliolumbar ligament,199 but because of the deep location of this structure, there can be no guarantee that, without radiological confirmation, the ligament was accurately or selectively infiltrated.
Other investigators have been more circumspect in interpreting tenderness near the posterior superior iliac spine, and question whether the pain stems from the iliolumbar ligament, the lumbosacral joint or the back muscles.200–202 Indeed, radiographic studies of injections made into the tender area reveal spread, not into the iliolumbar ligament but extensively along the iliac crest.198 Accordingly, the rubric – iliac crest syndrome – has been adopted to describe this entity.200,201 Others have referred to it simply as lumbosacral strain.202
What all investigators have overlooked is that the site of tenderness in iliac crest syndrome happens to overlie the site of attachment of the lumbar intermuscular aponeurosis (LIA), which constitutes a common tendon for the lumbar fibres of longissimus thoracis.203,204 The LIA attaches to the iliac crest rostromedial to the posterior superior iliac spine and exhibits a morphology not unlike that of the common extensor origin of the elbow. Thus, a basis for pain and tenderness in this region could be a tendonopathy of the LIA. On the other hand, it could be no more specific than tenderness in the posterior back muscles, which has been recognised for many years under different rubrics.202,205,206
Regardless of its underlying pathology, the putative advantage of recognising an iliac crest syndrome is that perhaps specific therapy might be applied. In this regard, if iliac crest syndrome is defined simply as tenderness over the medial part of the iliac crest, the kappa score for its diagnosis is 0.57.207 If the criteria are extended to include reproduction of typical pain, the kappa score rises to 0.66.207 These scores indicate that the syndrome can be identified. Its prevalence seems to be about 30–50%.200 However, as long as the syndrome amounts to no more than tenderness, it is not evident whether it is a unique disorder or a feature that could occur in association with other sources and causes of back pain.
Recognising the syndrome, however, has little impact on treatment. Injecting the area with local anaesthetic is significantly more effective than injecting it with normal saline, but only some 50% of patients benefit and only 30% obtain more than 80% improvement.200
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree