Lymph Node Imaging Techniques
Imaging evaluation of lymph nodes forms an integral component of staging of various malignancies, including lymphomas, and is also helpful in the evaluation of certain infective and inflammatory processes within the abdomen. This has special relevance in the abdomen because the lymph node system in this region is not readily accessible for clinical examination or tissue sampling. Therefore, accurate identification and characterization of abnormalities of the lymph nodes in the abdomen and pelvis are crucial to achieve optimal diagnosis and plan treatment strategies. A wide range of imaging techniques is available for evaluation of the lymph nodes in the abdomen and pelvis. Lymphangiography was once considered the preferred imaging modality for evaluation of nodal disease. However, with the advent of imaging techniques such as ultrasonography, computed tomography (CT), magnetic resonance imaging (MRI), positron emission tomography (PET), and PET/CT, the role of lymphangiography has now faded and has only a few valid indications. The superior soft tissue resolution of CT and MRI has improved the detection and assessment of even small lymph nodes. However, reliance on size criteria for characterization has limited the accuracy of these modalities in nodal staging of malignancies. Fluorodeoxyglucose (FDG)-labeled PET has further revolutionized oncologic imaging by recognizing benign and malignant nodes based on their FDG uptake. Development of investigational lymphotropic nanoparticles for the diagnosis of lymph node metastases has added a new dimension to the scope of MRI of lymph nodes as well. This chapter will discuss the various imaging modalities and describe their role in imaging of lymph nodes of the abdomen and pelvis. Additionally, various clinical manifestations of lymphadenopathy in the abdomen and pelvis will be individually discussed.
Normal Anatomy
The lymphatic system is a complex network of lymph nodes connected by lymphatic capillaries and ducts that play an important role in the immune system. It provides protection against organisms, particulate matter, and neoplastic cells by the processes of phagocytosis, cell-mediated and antibody-mediated immune complexes, production of B and T lymphocytes, and antibody-producing plasma cells. There are approximately 230 nodes in the abdomen and pelvis and 400 to 500 lymph nodes in the entire adult body.
The lymphatics of the abdomen have been divided into parietal and visceral systems composed of the lymphatic channels and nodes that drain the walls and contents of the abdominal cavity. The parietal lymphatic system has a superficial component that drains the skin and subcutaneous tissue and a deep component draining the muscles and fasciae of the abdominal wall. The visceral lymphatics initially drain into nodes located close to the viscera, then into intermediate nodes situated along the peritoneal ligaments and mesenteries, and finally into the larger group of nodes situated along the major branches of the abdominal aorta. The lymphatics follow the course of the parietal and visceral branches of the abdominal aorta and ultimately drain into the venous stream via the thoracic duct. The thoracic duct is the final lymphatic pathway for the entire pelvis, retroperitoneum, and peritoneal cavity.
Lymph nodes within the abdomen can be broadly divided into those related to the abdominal viscera and retroperitoneal nodes ( Figures 61-1 to 61-3 ). The lymph nodes associated with the abdominal viscera are situated along the distribution of the celiac, superior mesenteric, and inferior mesenteric arteries. The lymph nodes of the abdominal viscera ultimately drain into the retroperitoneum and into the cisterna chyli. Lymph nodes within the retroperitoneum are named and grouped according to their relation to the inferior vena cava and the abdominal aorta: paracaval, precaval, retrocaval, aortocaval, preaortic, and para-aortic. The preaortic group comprises the celiac, superior mesenteric, and inferior mesenteric nodes that drain the lymphatics of the intestinal tract. The para-aortic, paracaval, and aortocaval nodes form the lateral aortic group, which is the main terminal group for lymphatic drainage of all the abdominal and pelvic viscera of the urogenital system. The retroperitoneal nodes finally drain into the cisterna chyli, which empties into the thoracic duct.
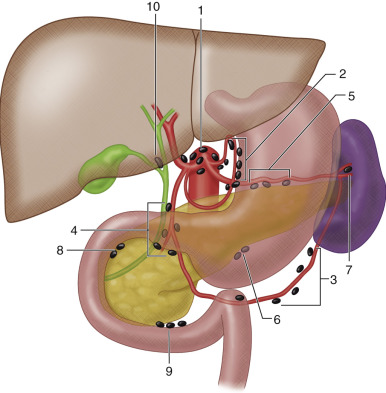

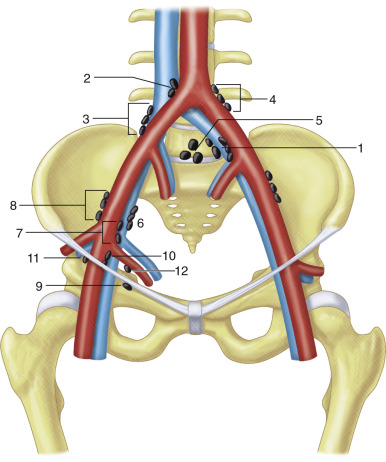
The lymphatic drainage of the pelvis has been categorized into the parietal and visceral channels as well ( Figures 61-4 and 61-5 ). The parietal channels drain the skin and superficial fascia, and the visceral channels drain the urinary tract, reproductive organs, rectum, perineum, and external genitalia. The lymphatic drainage of the pelvis occurs through nodes situated along the pelvic sidewall: common iliac, external iliac (including obturator), internal iliac or hypogastric (along the internal iliac vessels), and presacral. The external iliac nodes receive lymphatic drainage from the inguinal nodes and the obturator lymphatics, and the internal iliac nodes receive drainage from the visceral pelvic organs. The internal iliac and external iliac nodes drain into the common iliac nodes, which finally drain into the retroperitoneal para-aortic nodes along the aorta and inferior vena cava. The nodes at the junction of internal and external iliac vessels have been referred to as junctional nodes. Lymphatics from the pelvic viscera also can have drainage into the inguinal nodes externally. The pelvic nodal chains drain directly into the retroperitoneal system, and there are numerous interconnections with the celiac and mesenteric nodal systems as well.

Retrocrural Nodes
Retrocrural nodes are situated within the retrocrural space connecting the posterior mediastinum to the retroperitoneum and contain the aorta, thoracic duct, azygos vein, hemiazygos vein, and retrocrural lymph nodes ( Figure 61-6, A ). The lymphatic drainage from the diaphragm, posterior mediastinum, and upper lumbar region occurs into the retrocrural nodes. The retrocrural space can act as a conduit for communication of diseases originating above or below the diaphragm. Retrocrural lymph nodes are considered to be enlarged when they exceed 6 mm.


Gastric Nodes
The gastric nodes consist of multiple groups of nodes situated along the lesser and greater curvature of the stomach (see Figure 61-6, B ). These include the right and left gastric nodes, which are located within the lesser omentum along the lesser curvature of stomach, and the right and left gastroepiploic nodes, which lie within the greater omentum along the lower part of the greater curvature of the stomach. Also included are the pyloric nodes, which receive drainage from the right gastroepiploic nodes and from the first part of duodenum and head of pancreas. The pyloric nodes ultimately drain into the celiac nodes.
Gastrohepatic Ligament Nodes
The gastrohepatic ligament (see Figure 61-6, C ), which suspends the stomach from the liver, constitutes the superior segment of the lesser omentum. This ligament contains the left gastric artery and coronary vein and merges into the fissure of ligamentum venosum, which acts as a landmark on CT. Gastrohepatic ligament nodes are considered to be enlarged when they exceed 8 mm. Potential diagnostic pitfalls, which can mimic mild lymphadenopathy in this region, include coronary varices of the upper margin of the pancreas and transverse colon.
Portohepatic Nodes
Portal nodes, as the name suggests, lie within the porta hepatis and extend down the hepatoduodenal ligament, thereby interconnecting with the gastrohepatic ligament nodes (see Figure 61-6, D ). These nodes drain into the celiac nodes. One of these nodes around the hilum of the liver at the junction of the cystic and common bile ducts near the neck of the gallbladder has been named the Quenu’s cystic node. The portal nodes lie around the portal vein and can completely surround and obliterate the portal vein when enlarged. Hence, adequate enhancement with intravenous contrast material is essential for diagnosis and to rule out portal vein involvement. Portal nodes are abnormal if greater than 7 mm in size.
Pancreaticoduodenal Nodes
Pancreaticoduodenal nodes lie between the duodenal sweep and pancreatic head, anterior to the inferior vena cava (see Figure 61-6, E ). The pancreaticoduodenal nodes are often grouped with the pericaval and superior mesenteric artery nodes into a larger category termed the peripancreatic nodes. Nodes in this location exceeding 10 mm are considered enlarged.
Perisplenic Nodes
Perisplenic nodes are often located around the splenic hilum, and their drainage area includes the spleen, greater curvature of the stomach, and tail of the pancreas. These nodes ultimately drain into the celiac group via the pancreaticosplenic chain of nodes, which lie along the pancreas. The upper limit of normal for these nodes is 10 mm.
Mesenteric Nodes
The small bowel mesentery contains a large number of nodes that accompany the branches of the superior mesentery and vein (see Figure 61-6, G ). These are located within the mesenteric fat and are formed of three distinct groups. The most distal group formed by the juxtaintestinal nodes is situated close to the intestinal walls between the terminal jejunal and ileal arteries. The intermediate group is situated between the distal group, and the last group is the central mesenteric nodes and is situated near the mesenteric root. The eventual lymphatic drainage is to the superior mesenteric artery nodes at the base of the mesentery and from there to the retroperitoneal nodes.
Celiac, Superior Mesenteric, and Inferior Mesenteric Nodes
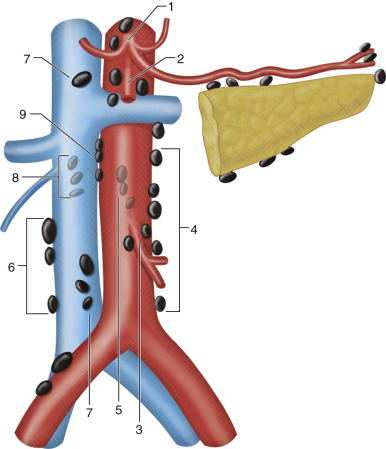
The celiac and superior mesenteric artery nodes, along with the nodes at the base of the inferior mesenteric artery, are the preaortic nodes (see Figures 61-6, H and I ). The celiac (at T12 vertebra) and superior mesenteric (at L1 vertebra) nodes are grouped around the origins of their respective vessels and are easily distinguished. However, it is difficult to distinguish the inferior mesenteric nodes because the artery often is not visualized as a discrete structure on CT. The celiac and superior mesenteric groups are the terminal nodes of the gastrointestinal tract. The celiac nodes receive lymph from intermediate nodes such as the gastrohepatic ligament, portohepatic, pancreaticoduodenal, and perisplenic nodes draining the stomach, duodenum, and hepatobiliary system. The superior mesenteric group receives lymph from the mesenteric, ileocolic, and colic nodal chains extending from the ligament of Treitz to the splenic flexure. These nodes have interconnections with the portosplenic nodes and the retroperitoneal periaortic nodes. The inferior mesenteric nodes (at L3 vertebra) receive drainage from the nodes along the inferior mesenteric artery, sigmoid artery, and superior rectal vessels. The colic nodal chains are composed of epicolic nodes embedded in the walls of the colon, paracolic nodes along the mesenteric borders of the colon, and intermediate colic nodes located along the middle and left colic arteries.
Para-Aortic Nodes
The para-aortic nodes can be divided into eight subgroups on the basis of their relationship with the aorta and inferior vena cava (see Figure 61-6, J and K ). Surrounding the aorta are the bilateral lateral aortic, preaortic, and retroaortic subgroups. The right lateral aortic subgroup is further divided into aortocaval, laterocaval, precaval, retrocaval, and paracaval subgroups to reflect the relationship with the inferior vena cava.
External Iliac Nodes
The external iliac nodal group is composed of three chains: the lateral, middle, and medial. The nodes distributed along and lateral to the external iliac artery form the lateral chain (see Figure 61-6, L ), and the middle nodal chain is composed of nodes located between the external iliac artery and vein. The medial chain is formed of the nodes lying medial and posterior to the external iliac vein, and they are often named obturator nodes because they accompany the obturator vessels (see Figure 61-6, N ).
Internal Iliac Nodes
The internal iliac (hypogastric) nodes encompass several groups of lymph nodes that are located along the visceral branches of the internal iliac artery. Differentiation of these lymph nodes is difficult on imaging because they are often clustered close to each other. The lymph nodes are identified based on the branches of the internal iliac vessels they accompany, for example, the uterine artery, the inferior vesical artery, the middle hemorrhoidal arteries, the superior and inferior gluteal arteries, and the internal pudendal artery (see Figure 61-6, M ). Two specific groups of lymph nodes that can often be identified include the anterior nodes, located anterior to the internal iliac vessels at the origin of the umbilical and obturator arteries, and the lateral sacral nodes, which lie anterior to the first two sacral foramina along the lateral sacral arteries.
Common Iliac Nodes
The common iliac nodal group consists of three groups: the medial, the middle, and the lateral groups ( Figure 61-6, O ). The medial group of lymph nodes is located in a central triangular area between the common iliac arteries, extending from the aortic bifurcation to the origin of the external and internal iliac arteries. The nodes at the sacral promontory are grouped with the medial chain. The middle group of lymph nodes occupies the region of the lumbosacral fossa, bordered by the sacral ala posteriorly, the common iliac vessels anteriorly, the psoas muscle laterally, and the lumbosacral vertebrae medially. The lateral groups of lymph nodes lie lateral to the common iliac artery and extend lower as the lateral external lymph nodal chain. The common iliac nodes ultimately drain into the para-aortic nodes.
Inguinal Lymph Nodes
The inguinal nodal group his subdivided into two categories: superficial and deep. The superficial group of nodes (see Figure 61-6, P ) lies anterior to the inguinal ligament in the subcutaneous tissue and can be identified as they accompany the superficial femoral and saphenous veins. The nodes at the saphenofemoral junction form an important group in the superficial inguinal nodal chain. The deep group of inguinal nodes (see Figure 61-6, P ) is located deeper within the femoral sheath along the common femoral vessels medial to the femoral vein. The fascial planes that separate the superficial from the deep group of lymph nodes are not clearly identified on CT. The deep group of nodes drains into the medial chain of the external iliac nodes. A useful landmark to separate the deep inguinal nodes from the medial chain of the external iliac nodes is the inguinal ligament and the origins of the inferior epigastric and iliac circumflex vessels.
Computed Tomography
CT is considered the initial examination of choice for the evaluation of abdominal and pelvic lymph nodes. It is also the most commonly performed examination for the evaluation of abdominal lymph nodes, owing to its capability to concurrently characterize solid organ and hollow visceral abnormalities. The advent of multidetector CT (MDCT) scanners that acquire isotropic volume data and the advances in imaging workstations that allow multiplanar and three-dimensional (3D) evaluation of these isotropic data sets have broadened the function of MDCT in the evaluation of abdominal and pelvic lymph nodes. Thin collimation and higher spatial resolution of MDCT now allow detection of small lymph nodes and enhanced characterization of lymph node morphology.
Technique
No particular technique is recommended for lymph node evaluation because their evaluation forms a part of the routine abdominal and pelvic CT examination. However, it is important to adhere to certain essential practices to maintain diagnostic accuracy. Chief among these is the administration of positive oral contrast material (1% to 2% dilute barium/Gastrografin) in particular to avoid misinterpretation of unopacified bowel loops as enlarged lymph nodes, especially in the retroperitoneum and mesentery. Intravenous administration of a contrast agent is routinely recommended because it is an integral component of lymph node evaluation on CT. Its benefits are threefold, as follows:
- 1.
It permits differentiation of small lymph nodes from tortuous vessels, particularly in the perigastric region and pelvis.
- 2.
Contrast-enhanced scans are invaluable in the assessment of associated pathologic processes affecting the abdominal organs.
- 3.
Lymph node enhancement patterns give important clues pertaining to the pathologic process involved.
A routine abdomen and pelvis scan obtained 60 to 75 seconds after the intravenous injection of a contrast agent is sufficient and is ideal with a volumetric acquisition with a slice thickness of at least 5 mm. This technique will provide good vascular opacification throughout the examination, allowing differentiation of enlarged nodes from adjacent vascular structures. A dedicated examination for pelvic lymph nodes requires more intense venous enhancement compared with abdominal and retroperitoneal lymph nodes. Optimal enhancement of the pelvic veins occurs at 3 minutes after initiation of the bolus injection of contrast material. Use of N-butyl scopolamine (Buscopan) is optional because it helps reduce artifacts from bowel peristalsis. However, it is also important to tailor the CT examination according to the clinical scenario in question, because of concerns about the dose of radiation particularly in those patients whose disease is being observed after treatment.
Normal Anatomy
On CT, normal nodes are ovoid and are of soft tissue density ( Figure 61-7 ). Normal lymph nodes show mild to moderate enhancement after contrast agent administration. The excellent contrast and spatial resolution of MDCT and the presence of surrounding fat allows routine visualization of normal retroperitoneal and mesenteric lymph nodes. Routine visualization of pelvic and perivisceral lymph nodes is less often possible owing to multiple adjacent vessels. Asymmetry of pelvic vessels in healthy subjects is a pitfall in the diagnosis of lymph nodes with unenhanced CT.

Measurement of Nodal Size.
Lymph node size is obtained by measuring the maximum short-axis diameter, which helps minimize the errors resulting from node orientation ( Figure 61-8 ). The size of normal nodes varies depending on the anatomic location in the body. In the abdomen, the upper limit of the maximum short-axis diameter of normal nodes varies between 6 and 10 mm and increases in size caudally.

Mimics of Abnormal Lymph Nodes on Computed Tomography.
Unopacified bowel loops and prominent normal vascular structures such as gonadal veins and iliac vessels can mimic lymphadenopathy. Vascular anomalies including a left-sided or duplicated inferior vena cava and varices resulting from portal hypertension can mimic lymph nodes. The papillary process of the caudate lobe or bulbous scalloped diaphragmatic crus may simulate lymphadenopathy in the portocaval space or retrocrural region ( Figure 61-9 ). An accessory spleen or normal ovarian tissue also can mimic lymph nodes (see Figure 61-6, D ).

Pathophysiology and Pathology
The only widely accepted criterion for the diagnosis of abnormal lymph nodes on CT is nodal size because CT cannot display abnormal architecture in a normal-sized node. CT is also unable to differentiate between reactive hyperplasia and metastases in enlarged lymph nodes. This drawback is the reason for the majority of false-negative and false-positive results from CT examinations. The secondary criteria used for characterization of nodal pathologic processes on CT are shape, margin, attenuation, and enhancement.
Ultrasonography
Ultrasonography is widely used for diagnosing lymph node disorders of the abdomen. In contrast to other imaging modalities, such as CT, which primarily rely on size criteria to diagnose malignant invasion, ultrasonography also permits analysis of various echo features of the visualized lymph nodes to predict malignant invasion. The detection and characterization of abdominal and pelvic lymph nodes can be done by either the transabdominal approach or the endoluminal route. Transabdominal ultrasonography is performed by using a standard 3.5- to 5-MHz convex array transducer, whereas endoscopic ultrasonography is performed with a radial scanning echoendoscope at 7.5 and 12 MHz. Conventional transabdominal ultrasonography has a limited role in the evaluation of abdominopelvic lymphadenopathy owing to several factors. Endoscopic ultrasonography permits closer evaluation of the intra-abdominal lymph nodes and allows more reliable assessment of their echo structure, margin, and shape than conventional ultrasonography. As a result, endoscopic ultrasonography is being increasingly used for detecting lymph nodes in the vicinity of the gastrointestinal tract as part of the staging evaluation of gastrointestinal and pancreaticobiliary malignancies. Endoscopic ultrasonographic elastography is a promising method that allows characterization and differentiation of benign and malignant lymph nodes with a high sensitivity, specificity, and accuracy, offering complementary information to conventional endoscopic ultrasound imaging. Contrast-enhanced ultrasound examination has been found to be useful in the differentiation of benign and malignant lymph nodes. On contrast-enhanced ultrasonography, benign lymph nodes have been shown to display uniform enhancement, whereas malignant lymph nodes demonstrate a defect in their enhancement.
Normal Anatomy
Normal lymph nodes can be either isoechoic or hyperechoic in echotexture. They are usually oval or polygonal ( Figure 61-10 ). A characteristic feature of a benign lymph node is the presence of a distinctive fatty echogenic hilum, called the “visible hilus sign.” Ultrasonography has a lower sensitivity in the detection of para-aortic lymph nodes owing to interference by mesenteric fat and abdominal gas.

Pathophysiology and Pathology
Features that suggest abnormality include enlargement, rounded shape, loss of central hilum, intense hypoechogenicity, and presence of altered nodal contours ( Figure 61-11 ). The features considered to be predictive of malignant lymph node invasion include hypoechoic structure, sharply demarcated borders, rounded contour, and a size greater than 10 mm. If the lymph node is less than 10 mm, is not round, is hyperechoic, and has indistinct or fuzzy margins, it is considered benign.

Magnetic Resonance Imaging
MRI is fast emerging as a mainstay in the evaluation of abdominal and pelvic lymph nodes, particularly as a part of staging for abdominopelvic malignancies. The inherent high soft tissue contrast resolution of MRI improves the detection of lymph nodes, particularly in malignancies of the rectum, urinary bladder, prostate, uterine cervix, and endometrium. The introduction of high field strength MR scanners (3.0 tesla) with increased signal-to-noise ratio (SNR) and improved spatial and temporal resolution have amplified the role of MRI in lymph node detection and characterization. The sequences particularly useful for MRI of lymph nodes include axial T1-weighted spin echo/gradient echo and axial T2-weighted fast spin echo and gadolinium-enhanced dynamic gradient echo sequences. Use of breath-hold sequences has been recommended for T2-weighted sequences because it causes minimum respiratory misregistration owing to respiratory motion during data acquisition. T1-weighted images are generally obtained for detection of enlarged retroperitoneal lymph nodes because the lymph nodes in the upper abdomen can be clearly distinguished against a background of retroperitoneal fat. Use of T2-weighted images is preferred in the lower abdomen and pelvis because lymph nodes can be distinguished from adjacent muscles such as the iliopsoas as lymph nodes have a higher signal intensity. MR oral contrast agents are advocated at times to distinguish between enlarged lymph nodes and adjacent bowel. Administration of gadolinium improves the conspicuity of lymph nodes, and the pattern of enhancement seen after gadolinium injection gives important clues to the type of lymph node pathology.
Diffusion Weighted Imaging
Diffusion weighted imaging has been advocated to improve the detection of nodal metastases in abdominal malignancies. This technique uses pulse sequences and techniques that are sensitive to microscopic motion of water protons. Single-shot echoplanar diffusion weighted imaging provides very rapid imaging sensitive to Brownian motion of water molecules, and areas with restricted water diffusion are displayed as areas of high signal intensity. Diffusion weighted imaging helps in detection of lymph nodes missed on conventional MR images and depicts more sites of nodal metastases in lymphoma and other abdominal malignancies (e.g., ovarian, pancreatic, colorectal, and hepatocellular cancer).
Magnetic Resonance Lymphangiography
MR lymphangiography helps in the differentiation of benign and malignant lymph nodes. This technique has been found to be very accurate in detecting microscopic malignant deposits within normal-sized nodes (i.e., micrometastases, which refers to malignant foci within lymph nodes <2 mm). MR lymphangiography is performed with ultrasmall superparamagnetic iron oxide particles (USPIOs; ferumoxtran-10), a reticuloendothelial system–specific contrast agent. Ferumoxtran-10 consists of a monocrystalline, superparamagnetic iron oxide core (2 to 3 nm or 4.3 to 6.0 nm) coated with a low molecular weight dextran. The lymphotropic nanoparticles first enter the interstitial space after intravenous administration and then reach the lymph nodes through the lymphatics. These nanoparticles are phagocytized by the intranodal macrophages. The nanoparticles accumulate in benign lymph nodes owing to abundance of macrophages, whereas malignant lymph nodes are unable to take up these particles because of neoplastic destruction of the macrophages.
This technique involves a baseline MR examination that is performed before the injection of the contrast agent for identification and localization of lymph nodes. After the intravenous injection of the contrast agent (2.6 mg of iron per kg of body weight reconstituted in 100 mL saline and given over a period of 20 to 25 minutes) a postcontrast scan is performed after 24 to 36 hours. Thin-section MRI with high resolution is performed for accurate characterization and detection of small metastatic foci within the nodes. The changes in signal intensity are best demonstrated on T2*-weighted gradient echo sequences. Malignant lymph nodes appear bright on these sequences owing to destroyed macrophages, whereas normal and benign lymph nodes show a drop in signal intensity as a result of susceptibility effects of the iron oxide particles within the intact macrophages. It is absolutely essential to time the postcontrast scan optimally because early scanning may result in inadequate lymph nodal localization of nanoparticles and false-positive diagnosis of benign lymph nodes as malignant.
Normal Anatomy
Lymph nodes on MRI appear round to ovoid and have an intermediate signal intensity compared with the very low signal intensity of the flowing blood in the aorta and inferior vena cava and the high-intensity MR signal of the retroperitoneal fat. Because of the high contrast between the lymphadenopathy and the surrounding fat, enlarged nodes are visualized on both T1- and T2-weighted images. Although the T1-weighted images provide optimum contrast for lymph node identification, T2-weighted images are valuable as an adjunct. Increased signal of the lymph nodes on T2 weighting compared to T1 weighting helps confirm their identity. The signal intensity of lymph nodes is lower than that of fat but higher than muscle on T1-weighted images ( Figure 61-12, A ). On T2-weighted images, the signal intensity of lymph nodes is closer to that of fat and higher than that of muscle (see Figure 61-12, B ). The presence of many venous tributaries particularly in the hypogastric region makes it difficult to detect the nodes in the pelvis owing to close apposition of the lymph nodes to the pelvic neurovascular bundle. Differentiation of lymph nodes from a vessel is made on the basis that vascular structures have low signal intensity on T1-weighted spin echo sequences and appear as tubular structures on adjacent images. After gadolinium administration, normal lymph nodes demonstrate homogeneous enhancement.

On MR lymphangiography, normal lymph nodes show a homogeneous decrease in signal intensity after the administration of USPIOs, which is suggestive of normal contrast delivery and normal macrophage activity within the nodes.
Pathophysiology and Pathology
Like CT, the diagnosis of abnormal lymph nodes on MRI is based on widely accepted size criteria. Signal intensity changes within lymph nodes also have been suggested as signs of abnormality on MRI. Malignant infiltration of lymph nodes can lead to heterogeneity of signal on T2-weighted MR images. The presence of necrosis secondary to either metastasis or infection leads to a low signal intensity on T1-weighted images and a high signal intensity on T2-weighted images. After treatment of malignancy, T2-weighted images can help in the detection of residual tumor within lymph nodes. Residual tumors usually show high signal on T2-weighted images, whereas successfully treated tumors show reduced signal intensity because of the presence of fibrosis. However, caution is required because successfully treated lymph nodes may be hyperintense for up to 1 year and because increased signal on T2-weighted images sometimes can be seen with inflammatory involvement of lymph nodes.
After gadolinium administration, the pattern of enhancement gives clues to the etiologic factor. Although homogeneous enhancement cannot substantially differentiate between normal and abnormal lymph nodes, the presence of heterogeneous or peripheral rim enhancement with central necrosis should raise suspicion. Gadolinium-enhanced MRI may be useful in differentiating between normal lymph nodes and normal-sized lymph nodes with metastatic involvement. Lymph nodes with malignant infiltration show rapid enhancement compared with uninvolved lymph nodes, and the pattern of enhancement may be similar to the primary tumor.
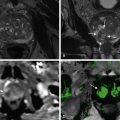
On diffusion weighted imaging, lymph nodes with malignant infiltration will appear hyperintense owing to restricted water diffusion ( Figure 61-13 ). Although this sign is quite specific for metastases, some overlap does occur when there is inflammatory involvement of the lymph nodes.

On MR lymphangiography, malignant lymph nodes appear hyperintense and do not show a reduction in signal intensity on T2-weighted spin echo or gradient echo sequences. The absence of signal loss could be either homogeneous or heterogeneous. In the latter situation, the malignant lymph nodes could either have a mottled appearance (suggesting the presence of partial infiltration of lymph node by malignant cells and areas of preserved architecture) or have an area of central hyperintensity with a peripheral rim of reduced signal.
Positron Emission Tomography
PET is a rapidly emerging innovative imaging technique that provides a 3D image or map of the functional processes in the body. This nuclear medicine imaging technique has found great application in oncology, especially in staging and posttreatment follow-up of malignancies. PET involves intravenous injection of a short-lived radioactive tracer isotope such as fluorine coupled with a metabolically active molecule such as glucose. The radioactive tracer decays by the emission of positrons, which is quantified by the imaging scanner.
The molecule for oncologic imaging is FDG, which is a combination of 18-fluorine and deoxyglucose. FDG as a glucose analog competes with glucose for transport sites on the cell membrane and within the cell for various enzymatic activities. Once inside the cell, it gets trapped by getting phosphorylated by the enzyme hexokinase to FDG phosphate because it cannot be metabolized further by metabolic pathways. This trapped FDG becomes the source for positrons and lights up tumors when imaged by the PET scanner. Malignant tissues, because of their increased glycolytic activity, accumulate FDG at a higher rate than normal tissues, making FDG a very suitable analog for oncologic purposes. The differential uptake between tumoral and nontumoral tissues also creates a good target-to-background ratio. FDG-PET thus provides metabolic information on glucose uptake of tumors and is found to be useful in management of patients with malignancies. PET has the potential to enhance the accuracy of lymph node detection by depicting metastatic deposits within lymph nodes and ruling out malignant infiltration in benign enlarged lymph nodes. It has been reported to be very sensitive and specific for the detection of lymph node metastases.
Because metabolic changes precede structural changes associated with any disease process, PET has the advantage of depicting changes in tumor function before conventional imaging modalities such as CT and MRI can demonstrate change in tumor size. This ability of PET can be used to monitor tumor response to therapy (chemotherapy/irradiation) and in the detection of recurrence.
A major limitation in imaging with PET is the low anatomic resolution of the images obtained that hampers accurate localization of the area of increased glucose metabolism. However, this has largely been addressed by the implementation of PET/CT fusion, which combines the anatomic data of CT with the functional information provided by PET. The combination of PET/CT has several benefits because it combines the high spatial resolution of CT images with the functional information provided by PET.
Normal Anatomy
Areas that show normal physiologic uptake and thus interfere with interpretation include the kidney, urinary tract, liver, spleen, stomach, small intestine, and colon, which shows the highest uptake, particularly in the region of the cecum and rectosigmoid region.
Pathophysiology and Pathology
Malignant lymph nodes that have higher than normal levels of glucose uptake show as bright spots on PET images ( Figure 61-14 ). The degree of brightness on PET images depends on the different levels of tissue or organ metabolic activity. A quantitative estimate of the metabolic activity of cells is made using a standardized uptake value (SUV). Tracking the SUV values within a lesion over time offers a standardized approach for measuring the response of tumors to therapy. Increased FDG uptake indicates increased glucose uptake. Thus, FDG is not a cancer-specific agent and increased uptake can occur in several inflammatory lesions, including sarcoidosis, tuberculosis, fungal infection, and abscesses.

Clinical Role of Lymph Node Imaging
A wide variety of disease processes, both benign and malignant, can result in abdominal and pelvic lymphadenopathy. The technical advancements in cross-sectional imaging such as MDCT and MRI have brought about profound changes in the detection and characterization of lymph nodes. The emergence of functional imaging techniques such as PET and the development of lymphotropic MR contrast agents have allowed detection of malignant lymph nodes with higher accuracy even in small lymph nodes. It is important for radiologists to be aware of the complex pattern of intercommunications between regional lymph node groups and also be well versed in the common manifestations of benign and malignant lymphadenopathy.
Benign Diseases of Lymph Nodes
Infective and Noninfective Lymphadenitis
Infective Lymphadenitis
Suppurative Lymphadenitis.
Suppurative lymphadenitis is a rare occurrence in the abdomen. It commonly results from Yersinia enterocolitica infection involving the mesenteric nodes in the right iliac fossa. Suppurative iliac lymphadenitis can develop secondary to infection in the lower extremities, and the organism most commonly implicated is Staphylococcus aureus. Metastatic S. aureus suppurative lymphadenitis also has been reported in the retroperitoneal nodes with spread from acute bacterial endocarditis. Retroperitoneal abscesses usually complicate untreated suppurative lymphadenitis and can lead to various intra-abdominal complications.
Tuberculosis.
The abdomen is involved in 11% to 16% of patients with extrapulmonary tuberculosis, and lymphadenopathy is the most common manifestation. Primary nodal involvement without extranodal diseases occurs in 55% of patients with abdominal tuberculosis. Tuberculous involvement of abdominal lymph nodes can occur either through infection of the gastrointestinal tract, which is the most common route, through hematogenous spread, or through direct spread from adjacent organs.
Human Immunodeficiency Virus Infection.
Lymphadenopathy is the most common abnormal CT finding in the abdomen in patients with acquired immunodeficiency syndrome (AIDS), occurring in both infective and neoplastic conditions. Imaging, particularly CT, plays a valuable role in the evaluation of suspected opportunistic infection or neoplasia in patients with AIDS. Infection accounts for 64% of cases of abdominal adenopathy in the population positive for human immunodeficiency virus (HIV) infection and is more common than HIV-related neoplasia. Mycobacterium tuberculosis (Mtb) infection is the most common infectious cause of adenopathy and accounts for 50% of cases. Overall, infection with Mtb or Mycobacterium avium-intracellulare complex (MAC) accounts for up to 85% of infectious causes of adenopathy in AIDS. Viral and protozoal infections do not cause significant lymphadenopathy in patients with AIDS. Fungal infections such as histoplasmosis can manifest as retroperitoneal and mesenteric adenopathy, and these nodes either have soft tissue attenuation or are hypoattenuating. Kaposi’s sarcoma and non-Hodgkin’s lymphoma are the common malignant conditions causing adenopathy in patients with AIDS.
Whipple’s Disease.
Whipple’s disease is a chronic systemic illness characterized by infiltration of the small bowel mucosa and submucosa with foamy macrophages that contain glycoprotein granules that are positive for periodic acid–Schiff. It is caused by Tropheryma whippelii, and patients usually present with malabsorption. Lymph node involvement is a common occurrence in Whipple’s disease. Lymphadenopathy commonly responds to antibiotic therapy.
Noninfective Lymphadenitis
Inflammatory Bowel Disease.
Ulcerative colitis and Crohn’s disease, collectively known as inflammatory bowel diseases, can involve regional lymph nodes in the abdomen, and these lymph nodes are frequently well depicted on cross-sectional imaging.
Diffuse Liver Disease.
Chronic diffuse liver diseases, particularly cirrhosis, and viral hepatitis–related disorders lead to involvement of lymph nodes in the hepatoduodenal ligament (39%). The highest frequency of lymph nodes is seen in primary biliary cirrhosis (85%), followed by hepatitis C (HCV)-associated liver disease (42%), HCV/hepatitis B (HBV)-associated liver disease (41%), and alcoholic and idiopathic liver disease (9.5%). HBV infection alone is less likely to cause lymph node involvement compared with HCV infection. In HCV-associated liver diseases, nodal frequency is related to the severity of liver involvement. In HBV and HCV infections, the identification of enlarged nodes usually suggests a chronic active stage of disease. Nonalcoholic steatohepatitis and hemochromatosis do not frequently have lymph node involvement.
Castleman’s Disease.
Castleman’s disease, also known as angiofollicular lymph node hyperplasia or giant lymph node hyperplasia, is a rare benign process of unknown cause characterized pathologically by hyperplasia of both lymphatic tissue and small blood vessels. It is very rare in the abdomen and pelvis, with its description in the literature limited to only a few case reports. Castleman’s disease has been divided into two clinical types, localized and diffuse, according to the extent and spread of the disease. Pathologically, Castleman’s disease is categorized into three types: plasma cell type, vascular, and mixed type.
Cavitating Mesenteric Lymph Node Syndrome.
Cavitating mesenteric lymph node syndrome is an uncommon and poorly understood complication of celiac disease and is characterized by cavitary changes confined to the mesenteric nodal chain, without evidence of malignancy or mycobacterial infection. The involved lymph nodes have a low CT attenuation value owing to cavitation of the involved nodes. These nodes regress on treatment of the celiac disease with a gluten-free diet. This finding helps in the management because celiac disease is associated with a high incidence of lymphoma, and the presence of cavitation within lymph nodes is reassuring.
Lymphangioleiomyomatosis.
Lymphangioleiomyomatosis is a rare disease of unknown cause affecting women in the reproductive age group and is characterized by progressive dyspnea, chylothorax, and chylous ascites. It is characterized by proliferation of immature-appearing smooth muscle cells in the axial lymphatics of the thorax and abdomen. The four major abdominopelvic abnormalities in patients with lymphangioleiomyomatosis include renal angiomyolipomas, retroperitoneal and pelvic lymphadenopathy, lymphangioma, and chylous ascites. The severity of lung disease is correlated with the presence of abdominal adenopathy.
Sarcoidosis.
Sarcoidosis is a multiorgan chronic granulomatous disease characterized by the presence of noncaseating granulomas in the affected organs. This disease most commonly affects the lung, and occasional abdominal involvement is noted. Abdominal lymphadenopathy is a common feature, and it mimics other systemic diseases such as lymphoma, diffuse metastatic disease, and granulomatous or mycobacterial infection.
Pathophysiology and Pathology
Suppurative Lymphadenitis.
CT is the modality of choice for the detection and characterization of suppurative lymph nodes. The affected lymph nodes typically show a low-density center with thin or irregular peripheral rim enhancement. Because of acute inflammatory changes, the involved lymph nodes show significant surrounding perinodal soft tissue fat stranding.
Tuberculosis.
The mechanism of involvement of abdominal lymph nodes by tuberculosis determines the site of involvement. Extensive involvement of the lower para-aortic region usually favors a hematogenous dissemination, whereas nonhematogeneous spread seldom involves the lower para-aortic region. The other sites of involvement such as the hepatoduodenal ligament, the portocaval space, the root of mesentery, the peripancreatic region, the periceliac region, the gastrohepatic ligament, the greater omentum, and the upper para-aortic region can occur with both hematogenous and nonhematogeneous routes. Enlarged tuberculous lymph nodes, although reaching large sizes, are usually not responsible for invasion or obstruction of the common bile duct, blood vessels, and the urinary or gastrointestinal tract.
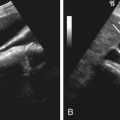
The most characteristic features of tubercular lymph nodes on contrast-enhanced CT are circular or ovoid lesions showing peripheral rim enhancement with central hypodensity. The peripheral enhancing portion corresponds to perinodal vascular inflammatory response or granulation tissue, whereas the central nonenhancing portion corresponds to caseation or liquefactive necrosis within the nodes ( Figure 61-15, A and B ). Heterogeneous and homogeneous enhancement also have been described within the involved nodes. Another specific finding is the multilocular appearance that results from the conglomeration of more than three adjacent enlarged lymph nodes. The involved tubercular lymph nodes usually measure more than 1 cm but are rarely larger than 4 cm in diameter, keeping with the self-limiting growth pattern of tuberculosis.
Calcification of the involved lymph nodes can occasionally occur. However, calcification within lymph nodes is a nonspecific feature and can occur in a wide variety of other conditions.
On T1-weighted MR images, the lymph nodes are isointense to hypointense and show increased signal intensity on T2-weighted images. The T2 hyperintensity corresponds to liquefactive necrosis. However, the central areas can occasionally be hypointense on T2-weighted images owing to release of paramagnetic free radicals from the active phagocytic cells. Obliteration of perinodal fat, which shows increased signal intensity on T2-weighted images, is also common. The contrast enhancement is predominantly peripheral, which can be uniform, thin or thick, and complete or incomplete. The affected lymph nodes when conglomerated often manifest as central and peripheral areas of heterogeneous enhancement (see Figure 61-15, C and E ).
Tuberculosis Versus Lymphoma.
Tuberculous lymphadenopathy can be easily confused with lymphoma affecting abdominal lymph nodes, and the differentiation is crucial owing to obvious difference in therapeutic management. The different characteristics of anatomic distribution, size, attenuation, and enhancement patterns displayed on contrast-enhanced CT are helpful in distinguishing tuberculosis from lymphoma. Whereas tubercular lymph nodes show a multilocular appearance with rim enhancement, lymphomatous nodes enhance homogeneously. Tuberculous nodes mostly measure less than 4 cm, whereas in lymphoma the nodes often exceed 4 cm. Distinction also can be made based on involvement of the para-aortic region, but this is relevant only when the tubercular involvement is secondary to nonhematogeneous spread. Nonhematogeneous disseminated tuberculosis predominantly affects the upper para-aortic region, whereas lymphoma involves both the upper and lower para-aortic nodes. Hematogenously disseminated tuberculosis, however, involves both the upper and lower para-aortic region, making its distinction from lymphoma more difficult. Disseminated tuberculosis has other differentiating features, such as the presence of miliary nodules in the lungs, hepatomegaly, and splenomegaly with inhomogeneous densities.
Human Immunodeficiency Virus Infection
Lymphadenopathy resulting from Mtb and MAC in HIV infection can be distinguished on CT depending on the presence of central low density or necrosis within the nodes. Central hypodensity within nodes is more common in Mtb (93%) than MAC (14%) infections, with most nodes in MAC showing homogeneous soft tissue attenuation because the immune system is not capable of granuloma formation. Nodes in MAC infection are smaller than in MTB, and clusters of normal-sized nodes are more frequently present. In Mtb, the lymph nodes are larger and frequently seen in the mesentery, retroperitoneum, and splenic hilum. An important distinction is the involvement of peritoneum and omentum, with resultant peritonitis and peritoneal implants in Mtb infection that do not occur in MAC infection. The retroperitoneal and mesenteric adenopathy in fungal infections such as histoplasmosis are often either of soft tissue attenuation or low attenuation. Other features include large hypoattenuating hepatosplenic lesions and severe immunocompromised status.
In nodal involvement secondary to Kaposi’s sarcoma, the characteristic finding on contrast-enhanced CT is the presence of hyperattenuating lymph nodes. The lymph nodes are primarily seen in the retroperitoneum, mesentery, and groin. These nodes are greater in attenuation than muscle and enhance similar to adjacent vessels. Isoattenuating and hypoattenuating lymph nodes also can be seen; however, central hypodensity is rare. Kaposi’s sarcoma accounts for 85% of cases of hyperattenuating lymph nodes in patients with AIDS. Less frequent causes of hyperattenuating nodes on CT in HIV include mycobacterial infection (9%), AIDS lymphoma (3%), angioimmunoblastic lymphoma (3%), and bacillary angiomatosis. Patients with AIDS-related lymphoma present with extensive retroperitoneal, mesenteric, and pelvic adenopathy that accounts for 56% of cases. The lymph nodes are large and bulky, with the majority of them showing soft tissue attenuation. Massive adenopathy, with nodes larger than 3 cm, is due to AIDS-related lymphoma in up to 90% of cases.
Whipple’s Disease.
The most common imaging finding in Whipple’s disease is thickening of small bowel folds with no dilatation. But the characteristic feature on CT is the presence of low-density nodes in the mesentery and retroperitoneum with hepatosplenomegaly and ascites. The lymph nodes have a high fat content, giving a low CT attenuation value of 10 to 20 Hounsfield units ( Figure 61-16 ).
Inflammatory Lymphadenitis
Inflammatory Bowel Disease.
Mesenteric lymphadenopathy is commonly seen in patients with inflammatory bowel diseases, particularly in Crohn’s disease. The nodes are found at the mesenteric root, mesenteric periphery, the right lower quadrant, or in the pericolic region ( Figure 61-17 ). The nodes range in size from 3 to 8 mm, are of soft tissue attenuation, and demonstrate homogeneous enhancement. Associated changes in the small or large bowel are usually, but not always, present. If the lymph nodes are larger than 10 mm, lymphoma and carcinoma must be excluded.
Diffuse Hepatic Diseases.
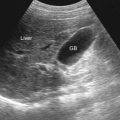
Normal nodes in the hepatoduodenal ligament are routinely seen clustered around the common hepatic artery and have been referred to as “daisy chain” nodes. The normal nodes are usually isoechoic or slightly hyperechoic with respect to liver on ultrasonography. Small hepatoduodenal ligament nodes (<5 mm) are not uncommon in patients without hepatobiliary disease. However, larger and more prominent nodes (>5 mm) in the hepatoduodenal ligament (porta hepatis) should prompt a careful evaluation of the liver for the possibility of chronic liver diseases ( Figure 61-18 ).
Castleman’s Disease
Localized Type.
The most characteristic feature of localized Castleman’s disease on CT is a well-defined, homogeneous, single intra-abdominal mass of soft tissue attenuation with typical early strong homogeneous enhancement and delayed washout on dynamic scanning ( Figure 61-19 ). These masses are circular, oval, or fusiform and range from 3.5 to 8 cm. On MRI, the lesions are either hypointense or isointense on T1-weighted images and hyperintense on T2-weighted images. Intense enhancement occurs after gadolinium administration. Large masses can show central areas of low density with distinct nonenhancing radial or fissured bands. These areas usually show low signal intensity on T1- and T2-weighted images. The low-density areas are seen mainly in the early phase of contrast enhancement and correspond to the parallel fibrous tissues seen on histopathologic examination. Another distinguishing feature seen in Castleman’s disease is the lack of cystic necrotic degeneration within the tumor. Calcifications have been reported in this disease and can be either coarse or punctate. Several daughter foci or satellite nodules can be seen around the primary lesion in the abdomen. The differential diagnosis for localized Castleman’s disease includes ectopic pheochromocytoma, accessory spleen, tuberculosis of the mesentery, and hypervascular metastatic lymph nodes.
Diffuse Type.
The diffuse type of Castleman’s disease manifests as multiple lymph node enlargement with mild to moderate and occasionally intense enhancement. Nonspecific signs such as organomegaly can be seen in certain cases. The diffuse and plasma cell variants have atypical imaging manifestations, and preoperative diagnosis is difficult. The common differential diagnosis is lymphoma, which also shows as multiple enlarged nodes with clear margins, uniform density, and mild to moderate enhancement.
Cavitating Mesenteric Lymph Node Syndrome.
In cavitating mesenteric lymph node syndrome, multiple cystic mesenteric masses are identified on CT that have central low attenuation, indicating fluid and occasionally fatty material. Masses range from 2 to 7 cm in diameter. Splenic atrophy is a common and characteristic feature. On ultrasonography, the mesenteric masses appear cystic. Imaging is remarkable for the absence of mediastinal, retroperitoneal, or inguinal lymphadenopathy. On MRI, a fat-fluid layer is seen within the lesions on both T1- and T2-weighted images, with signal loss at the fat-fluid interface occurring owing to the chemical shift artifact on out-of-phase steady-state spoiled gradient recalled echo acquisitions.
Lymphangioleiomyomatosis.
The affected retroperitoneal and pelvic lymph nodes in lymphangioleiomyomatosis typically show fat attenuation. The low Hounsfield unit values within the nodes are presumed to be due to chylous lymph collections or fat within the nodes. The adenopathy may be extensive, with nodes measuring up to 4 cm.
Mesenteric Lymphadenopathy.
The frequent use of MDCT scanners with thin collimation that allows improved spatial resolution has led to more frequent visualization of mesenteric lymph nodes in healthy adults. Normal mesenteric lymph nodes measuring up to 4 to 5 mm in short-axis diameter frequently can be seen in up to 39% of healthy adults. When detected, these nodes are usually multiple, with nearly half of these patients having five or more nodes. The nodes are routinely identified at the mesenteric root or throughout the mesentery. The current definition of mesenteric adenitis is the presence of a cluster of three or more lymph nodes with short-axis diameter of 5 mm or greater ( Figure 61-20 ). This definition has been described for adult patients but has limited value in children because mesenteric lymph nodes with a short-axis diameter of 5 to 10 mm are frequently found in the CT examination of children with low likelihood of mesenteric lymphadenopathy.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree