Chapter Outline
Anatomic Classification of Malabsorption
Diagnosis of Malabsorption: Clinical Perspective
Lymphatic Obstruction and Lymphangiectasia
Malabsorption is caused by a variety of diseases originating in the liver and biliary tree, pancreas, and small intestine. Normal digestion and absorption are presented as a background for understanding intestinal malfunction. A description of diseases of the small intestine that result in malabsorption is then presented.
Digestion and Absorption
The components of most foods (carbohydrates, proteins, and fats) cannot be used in their natural state. Foods must first be digested and then absorbed by the gastrointestinal (GI) tract. Proper functioning of the stomach, liver and biliary tree, pancreas, and small bowel and, to a lesser degree, salivary glands and colon, is required for normal digestion and absorption of food.
Carbohydrate Digestion and Absorption
Carbohydrates account for 40% to 50% of daily caloric intake. Ingested carbohydrates are derived from plants, except for lactose, which originates in dairy products. Simple sugars include the monosaccharides, fructose and glucose, and the disaccharides, sucrose and lactose. Starches are soluble polymers of glucose found in the cell walls of plants. The starches are α-amylose, a linear polymer of glucose, and amylopectin, a branched form of glucose. Dietary fibers are nondigestible carbohydrates (nonstarch polysaccharides) that are structural components of plant cell walls.
The GI tract is adapted for digesting carbohydrates to a monosaccharide form that can be transported across the epithelium of the small intestine. Luminal digestion of starches is accomplished by amylase secreted by the parotid gland and pancreas. Salivary amylase is of minor importance because this enzyme is deactivated in the acidic environment of the stomach. Amylase digestion of starches is so efficient that starch digestion is more dependent on the form of the starch than on the availability of luminal amylase. Amylase deficiency is present only in cases of severe pancreatic insufficiency.
Enzymes on the apical membrane of the enterocytes are responsible for further digestion of the products of luminal digestion and breakdown of disaccharides. For example, sucrose isomaltase is an enzyme that cleaves sucrose into glucose and fructose. Brush border lactase cleaves lactose into glucose and galactose. This enzyme is critical for mammalian survival because early nutrition is provided by the mother’s milk. The monosaccharides are then brought across the epithelium by passive diffusion, facilitative transport of proteins, or sodium-coupled active transport.
Colonic bacteria digest dietary fiber that reaches the colon, producing a variety of products, including short-chain fatty acids, methane, and hydrogen. Short-chain fatty acids are rapidly absorbed in the colon, providing energy for colonic epithelial cells.
Digestion of lactose, sucrose, and starches is incomplete because digestion depends on the type of food and contact time with the brush border. Between 2% and 20% of starch is not digested. Most small bowel diseases result in global dysfunction of the intestinal mucosa, leading to carbohydrate malabsorption, with subsequent diarrhea, flatulence, and weight loss. Specific disaccharidase deficiencies result in similar symptoms. For example, trehalose is a disaccharide of glucose found in insects, yeasts, and mushrooms. As a result, people who are trehalase-deficient have severe diarrhea after ingestion of mushrooms.
Protein Digestion and Absorption
Proteins in the lumen of the GI tract derive from the diet (70-100 g/day), from salivary, gastric, pancreatic, and biliary secretions (30 g/day), and from sloughed epithelial cells (30 g/day). Protein digestion begins in the stomach. Inactive precursors (pepsinogens) are secreted from chief cells and are activated to pepsins in the acidic environment of the stomach. Proteins in the gastric lumen are digested to a mixture of large polypeptides, smaller oligopeptides, and free amino acids.
The exocrine pancreas secretes five inactive precursor zymogens into the duodenal lumen. Enteropeptidase (enterokinase) on the brush border of the duodenal enterocytes converts trypsinogen into trypsin. Trypsin then activates the remainder of the pancreatic zymogens—chymotrypsinogen, proelastase, and procarboxypeptidases A and B. These pancreatic enzymes break proteins into oligopeptides (60%-70%) and free amino acids (30%-40%). Peptidases on the brush border of the enterocytes further degrade the intraluminal oligopeptides, resulting in a mixture of tripeptides, dipeptides, and free amino acids. Distinct transport systems separately transport free amino acids and peptides of two to three amino acids into the epithelial cells. Various peptidases within the enterocytes further digest the small polypeptides. Amino acids leave the enterocytes via transport mechanisms across the basal membrane, passing into the portal circulation.
Fat Digestion and Absorption
The sources of fat include diet (120-150 g), biliary secretions (40-50 g), and sloughed intestinal cells and bacteria. Most dietary fat is composed of neutral fats, long-chain triglycerides. Phospholipids and sterols (including cholesterol) comprise only a small percentage of dietary fat.
Lipid digestion begins in the stomach with the hydrolysis of triglycerides by gastric and salivary lipase; 20% to 30% of intraluminal fat digestion occurs in the stomach. The grinding action of the antrum helps reduce triglycerides into smaller particles. The fat globules are dispersed in a stable form with a large surface area, termed emulsification. Gastric lipid digestion has an increased role in patients with cystic fibrosis or a partial gastrectomy.
After chyme reaches the small intestine, bile salts and lecithin secreted by the liver solubilize the fat, allowing the fat to be broken into smaller droplets by the agitating action of the small bowel. Lipolytic enzymes secreted by the pancreas break triglycerides, phospholipids, and sterol esters into their component monoglycerides and free fatty acids. Bile salts combine with the monoglycerides and free fatty acids to form micelles. The micelles deliver the breakdown products of fat to the microvilli of the epithelial cell brush border.
After brush border uptake, long-chain fatty acids and monoglycerides are delivered to smooth endoplasmic reticulum in the cytoplasm for resynthesis into complex lipids. Phospholipids, an important component of cell membranes, are synthesized in the rough endoplasmic reticulum. Triglycerides resynthesized in the endoplasmic reticulum of enterocytes are secreted by enterocytes as lipoproteins. These lipoproteins are multimolecular aggregates of lipid and protein, with a configuration that allows transport through aqueous intracellular fluid or plasma. Lecithin is the primary phospholipid in lipoproteins and is derived primarily from bile salts (10-20 g/day) or diet (5-10 g/day). Cholesterol is primarily derived from biliary secretions (1-2 g/day), with a minor component (0.2-0.5 g/day) from the diet.
Most dietary fat enters the lymphatic circulation through lacteals located in each villus and is then passed via mesenteric lymphatics to the thoracic duct and superior vena cava. About 25% of triglycerides are transported to the liver bound to albumin.
Digestion of fat is very efficient. Almost all dietary triglyceride is absorbed by enterocytes. No ingested triglyceride is found in the colon. The small amount of fecal fat (<7 g/day) is derived from phospholipids in sloughed intestinal membranes or intestinal bacteria. After micelle delivery, bile salts return to the small bowel lumen to solubilize more free fatty acids and monoglycerides. Eventually, conjugated bile salts (95%) are resorbed in the ileum via an active sodium-coupled process. Some bile salts are passively absorbed in the proximal small intestine. Fecal loss of bile salts is balanced by synthesis of bile in the liver.
Fluids and Electrolytes
Diarrhea and malabsorption are not synonymous. Malabsorption may occur in some diarrheal states. A large volume of fluid (8-10 L) enters the GI lumen each day, but only 100 mL of fluid is normally excreted in the feces. The daily fluid load includes about 2 L of oral intake, 0.5 to 1.5 L of saliva, 2.5 L of gastric juice, 0.5 L of bile, 1.5 L of pancreatic secretions, and 1 L of intestinal secretions. The small bowel absorbs about 7 L of fluid daily, with 1.5 to 1.9 L of fluid reaching the colon. The colon is capable of resorbing up to 4 L of fluid on a daily basis. Diarrhea therefore occurs if more than 4 L of fluid reaches the colon or if abnormally functioning colonic mucosa cannot resorb the 1.9 L of fluid that normally enters the colon each day.
Anatomic Classification of Malabsorption
In summary, carbohydrate digestion requires a functioning pancreas and small bowel brush border enzymes. Normal protein digestion requires adequate gastric and pancreatic function and small bowel brush border enzymes. Fat digestion requires normal hepatic, biliary, exocrine, pancreatic, and small bowel function. An anatomic classification of malabsorption by abnormalities of the liver, biliary tree, stomach, and small intestine aids the radiologist in understanding malabsorption ( Table 43-1 ).
| Organ | Disease or Condition | Pathophysiology |
|---|---|---|
| Stomach | Zollinger-Ellison syndrome | Pancreatic enzyme inactivation by acid |
| Postgastrectomy | Rapid transit of nutrients, dilution of pancreatic enzymes | |
| Pernicious anemia | Intrinsic factor deficiency (vitamin B 12 malabsorption) | |
| Pancreas | Chronic pancreatitis, cystic fibrosis, pancreatic cancer | Decreased pancreatic enzyme and bicarbonate secretion |
| Liver, biliary tree | Severe parenchymal liver disease | Decreased bile salt formation |
| Cholestatic liver disease (primary biliary cirrhosis, drug-induced cholestasis), bile duct obstruction (bile duct carcinoma, pancreatic cancer, gallstones, sclerosing cholangitis) | Decreased bile salt delivery to duodenum | |
| Small intestine | Jejunal diverticulosis, scleroderma, small intestinal fistulas, stricture in Crohn’s disease, diabetes, pseudo-obstruction | Stasis with bacterial overgrowth, bile salt deconjugation |
| Crohn’s disease, small intestinal resection, cholecystocolonic fistula | Increased bile salt loss | |
| Lactase deficiency, Crohn’s disease | Disaccharidase deficiency | |
| Celiac disease, tropical sprue, Whipple’s disease, eosinophilic gastroenteritis, radiation enteropathy, Crohn’s disease, intestinal ischemia, ileal resection | Loss of normal epithelial cells | |
| Abetalipoproteinemia | Nonformation of chylomicrons | |
| Lymphangiectasia, lymphoma, tuberculosis, carcinoid | Lymphatic obstruction | |
| Diabetes mellitus, giardiasis, adrenal insufficiency, hyperthyroidism, hypogammaglobulinemia, amyloidosis, AIDS | Multiple causes |
Hepatobiliary and Pancreatic Disorders
Bile salt insufficiency caused by biliary obstruction or decreased hepatic synthesis leads to mild malabsorption. Any disease that destroys pancreatic exocrine tissue decreases bicarbonate and pancreatic enzyme secretion. Maldigestion does not occur until 90% of the pancreatic exocrine tissue has been destroyed. The most common cause of pancreas-related maldigestion is alcohol-related pancreatitis. Patients with cystic fibrosis have malabsorption, but this is a less common disease.
Gastric Disorders
Diseases of the stomach may cause mild malabsorptive states. Patients with pernicious anemia have decreased production of intrinsic factor, leading to vitamin B 12 deficiency. In patients with Zollinger-Ellison syndrome, inflammation and ulceration in the duodenum and first several loops of jejunum cause diarrhea, not malabsorption. About one third of patients with Zollinger-Ellison syndrome have diarrhea related to gastric hypersecretion and intestinal mucosal damage. Mild malabsorption results from excess acid entering the duodenum, which inactivates pancreatic enzymes.
Small Bowel Disorders
Malabsorption can result from a wide variety of mechanisms beginning in the intestinal lumen and extending deep to the small mesentery (see Table 43-1 ).
Bacterial Overgrowth (Intraluminal Stasis)
Any disease that causes stasis in the lumen of the small intestine can lead to bacterial overgrowth and small bowel dysfunction ( Box 43-1 ). Disorders that cause chronic small bowel obstruction (e.g., Crohn’s disease, adhesions) may result in stagnation and bacterial overgrowth. Diseases with intrinsic small bowel hypomotility (e.g., diabetes, scleroderma, jejunal diverticulosis) can also lead to stasis and bacterial overgrowth. Stasis in surgical blind loops or the afferent loop of a gastrojejunostomy can also result in bacterial overgrowth.
* Disorders typically having a malabsorptive state are highlighted in bold.
Genetic Disorders
Familial visceral neuropathies
Familial visceral myopathies
Muscular dystrophy
Collagen-Vascular Disorders
Progressive systemic sclerosis
Dermatomyositis, polymyositis
Periarteritis nodosa
Systemic lupus erythematosus
Endocrine Disorders
Diabetes mellitus
Hypothyroidism
Hypoparathyroidism
Neurologic Diseases
Parkinson’s disease
Multiple sclerosis
Chagas’ disease
Spinal cord injury
Drugs
Narcotics
Phenothiazines
Antiparkinson medications
Ganglionic blockers
Tricyclic antidepressants
Other
Celiac disease
Radiation enteritis
Jejunal diverticulosis
Amyloidosis
Lead poisoning
In patients with bacterial overgrowth, malabsorption is related to several mechanisms, including intraluminal bacterial deconjugation of bile salts and fermentation of carbohydrates. Concomitant epithelial cell dysfunction may be present. Bacterial digestion of malabsorbed fat forms compounds that can stimulate small bowel or colonic secretion. Solutes of malabsorbed carbohydrates and deconjugated bile salts are also osmotically active, resulting in water and electrolyte loss.
Brush Border Disease
Brush border enzyme deficiencies or transport mechanism deficiencies may cause osmotic diarrhea, malabsorption, or both. The most common example of osmotic diarrhea caused by brush border disaccharidase deficiency is so-called lactase deficiency (lactose-phlorizin hydrolase deficiency) with lactose intolerance. The norm for most people is for brush border lactose-phlorizin hydrolase levels to decline in older children and adolescents to 5% to 10% of early childhood levels. As a result, lactase-phlorizin hydrolase deficiency is the norm, and the ingestion of dairy products can lead to diarrhea, flatulence, and cramps in most adults. Preservation of lactose-phlorizin hydrolase is an autosomally recessive trait, found typically in northern Europeans. Intermediate activity of this enzyme is found in heterozygotes.
Mucosal Damage
Inflammation or necrosis of crypt cells and immature villous cells causes secretory diarrhea. Blood, pus, and mucus in the intestinal lumen lead to osmotic diarrhea. Widespread epithelial damage is therefore characterized by a combination of osmotic and secretory diarrhea and nutrient malabsorption. Diseases that destroy proximal small bowel epithelium cause generalized malabsorption of fats, proteins, carbohydrates, iron, and folate, whereas diseases that damage distal ileal mucosa primarily cause malabsorption of fat because of bile salt loss and vitamin B 12 deficiency. Mucosal diseases causing malabsorption typically involve relatively long segments of small bowel. These diseases are relatively uncommon, however; they include celiac disease, tropical sprue, Whipple’s disease, and eosinophilic enteritis.
Postmucosal Disease
Any disorder that obstructs lacteals in the villi or lymphatics in the small bowel mesentery may cause fat malabsorption. In primary lymphangiectasia, for example, abnormal formation of lymphatics results in impaired absorption of chylomicrons and fat-soluble vitamins and loss of lymph into the intestinal lumen from lymphoenteric fistulas.
Diseases with Multifactorial Causes
Malabsorption is caused by several mechanisms in many diseases. In amyloidosis, for example, malabsorption can be attributed to stasis with bacterial overgrowth, mucosal destruction caused by ischemia, and disruption of nutrient absorption by amyloid deposition in the lamina propria. Fat malabsorption in hyperthyroidism is attributed to rapid small bowel transit.
Diagnosis of Malabsorption: Clinical Perspective
Malabsorption is characterized by diarrhea, steatorrhea, excessive gas, abdominal pain, and weight loss. Diarrhea is caused by decreased intestinal absorption, colonic secretion of fluid induced by hydroxy fatty acids, and osmotic overload of bile salts and fatty acids. Steatorrhea is the term used for the bulky, foul-smelling, greasy stools caused by the malabsorption of fat. Excessive gas production is related to fermentation of carbohydrates by intestinal bacteria and results in borborygmi, flatulence, and abdominal distention. Abdominal pain has a variety of causes, including pancreatic inflammation, biliary disease, small bowel obstruction, and ischemia.
Unfortunately, adults with malabsorption often have an insidious presentation. Steatorrhea may be so mild that it is unrecognized by the patient. Mild steatorrhea is typical of low-grade bile duct obstruction, chronic hepatic disease, mild to moderate pancreatic insufficiency, and even mild celiac disease. Many patients with malabsorption present with symptoms related to a specific vitamin or nutrient deficiency. For example, vitamin A deficiency may cause night blindness, vitamin K deficiency results in easy bruisability, petechiae, or hematuria, vitamin D or calcium malabsorption results in paresthesias, tetany, or bone pain, and decreased absorption of folate, vitamin B, or iron may cause pallor, glossitis, stomatitis, and cheilosis.
The clinician has a broad array of laboratory tests and biopsies to make a diagnosis of malabsorption. The reader is referred to textbooks of gastroenterology for further description of these tests. Radiologic imaging in patients with a clinical diagnosis of malabsorption is performed as an adjunct to help identify diseases (e.g., jejunoileal diverticulosis) that cause malabsorption. Radiologic imaging is also used to detect complications of diseases that cause malabsorption (e.g., T-cell lymphoma arising in celiac disease).
Bacterial Overgrowth
A wide variety of disorders may cause small bowel hypomotility with resulting stasis and bacterial overgrowth (see Box 43-1 ) or intestinal pseudo-obstruction.
Jejunoileal Diverticulosis
Small bowel diverticula are acquired protrusions of mucosa and submucosa on the mesenteric border of the small bowel, where the vasa recta pierce the muscularis propria. The diverticula are larger and more numerous in the jejunum, decreasing in size and number as they progress into the ileum. Jejunoileal diverticula develop in a heterogeneous group of disorders associated with abnormalities of smooth muscle or the myenteric plexus. Small bowel dysmotility is frequent. Some cases of jejunoileal diverticulosis may be a manifestation of systemic sclerosis or a forme fruste of isolated scleroderma.
Jejunoileal diverticulosis is not uncommon, occurring in about 2% of the population, but most people with jejunal diverticulosis are asymptomatic. In patients with numerous diverticula, stasis with bacterial overgrowth and malabsorptive symptoms may occur. Complications of jejunal diverticula, including GI obstruction or perforation, may cause abdominal symptoms.
Diverticula are readily identified on barium studies as 1- to 7-cm round, barium-filled sacs on the mesenteric border of the small bowel ( Fig. 43-1 ). The mouths of the diverticula are broad-based and the necks are variable in length. Air-fluid levels may be present on upright or cross-table lateral radiographs. If there are numerous diverticula, the course of the small bowel may be obscured by the numerous sacs. On computed tomography (CT) scans, large diverticula are easily mistaken for small intestinal loops. The best way that diverticula are detected by CT is visualization of an air-fluid level in a focal sacculation. The motor disorder seen in jejunoileal diverticulosis is manifested by decreased peristalsis, increased intraluminal fluid and gas, luminal dilation, and prolonged transit time.

Although other small bowel disorders may be associated with sacculations or diverticula, jejunoileal diverticulosis is easily differentiated from the sacculations in Crohn’s disease, scleroderma, or prior surgery, a solitary Meckel’s diverticulum, or diverticula confined to the terminal ileum.
Jejunoileal diverticulosis may be complicated by pneumatosis cystoides intestinalis or free intraperitoneal air caused by perforation or benign pneumatosis. Jejunal diverticulitis may cause abdominal pain, free perforation obstruction or abscess formation, or GI bleeding. Jejunal diverticulitis is manifested on CT as a stranding or mass in the small bowel mesentery adjacent to a diverticulum or true abscess formation. Mechanical obstruction may result from volvulus, enterolith impaction, or diverticulitis. Heterotopic tissue or neoplasms are rare complications.
Progressive Systemic Sclerosis, Visceral Neuropathies, and Myopathies
The small intestine is clinically involved in about 40% of patients with progressive systemic sclerosis (scleroderma). The smooth muscle, especially that of the circular muscle layer, degenerates and is replaced by collagen. The muscular abnormalities result in duodenal and jejunal dilation ( Fig. 43-2 ) and hypomotility, with prolonged small bowel transit time and increased intraluminal fluid. Patchy and predominant fibrosis of the circular muscle layer leads to bunching of small bowel folds, producing the so-called hidebound bowel (see Fig. 43-2 ). Despite luminal dilation, an increased number of small bowel folds per inch is usually seen. Asymmetric scarring leads to wide-mouthed sacculations, usually on the mesenteric border of the small bowel. As with any small bowel disorder characterized by hypomotility, transient intussusceptions and pneumatosis intestinalis may occur, with or without pneumoperitoneum.

Several forms of visceral myopathies and neuropathies present with malabsorption or intestinal pseudo-obstruction. These include autosomal dominant and recessive conditions or isolated cases. Other isolated cases have been caused by Epstein-Barr virus infection or abnormalities of cytoskeleton filament proteins. Visceral myopathies are characterized histologically by degeneration and fibrosis of the inner circular and outer longitudinal smooth muscle layers. Congenital neuropathies are characterized by degeneration of the myenteric plexus of the bowel. Decreased or absent peristalsis is seen in visceral myopathies and scleroderma, but large-amplitude, nonpropulsive contractions are seen in visceral neuropathies. Marked duodenal enlargement typically occurs in visceral myopathies, whereas small bowel dilation may be present in visceral myopathies and neuropathies. The increased number of folds and sacculations typical of scleroderma are not found. The colon demonstrates hypercontractility in visceral neuropathy and dilation and lack of haustration in visceral myopathy.
Epithelial Cell Damage
Celiac Disease
Celiac disease (gluten-sensitive enteropathy, or celiac sprue) is a chronic immunologic inflammatory disease in which the gliadin fraction of wheat, barley, or rye gluten incites damage to the small bowel mucosa. Removal of wheat products from the diet reverses this mucosal damage. Although the mechanism of celiac disease is not completely understood, it is postulated that there is suspension of normal immune tolerance to foreign food antigens. The gliadin fraction of ingested wheat products stimulates an immune response in the epithelium of the small intestine, leading to mucosal inflammation and destruction. Lymphocytic infiltration may also occur in the stomach (as lymphocytic gastritis) or colon.
Celiac disease occurs in about 1 in 200 whites. Most people with celiac disease ignore minor symptoms or are asymptomatic. Celiac disease resulting in severe clinical symptoms is uncommon and is usually encountered in whites, particularly northern Europeans and people from Ireland. Children may present with diarrhea, abdominal distention, weight loss, and failure to thrive. Young adults may present with diarrhea, steatorrhea, or infertility. Older adults may present with steatorrhea, anemia, weight loss, or other symptoms. Patients who develop symptoms in adulthood or who develop celiac disease de novo in adulthood may present with symptoms related to the malabsorption of specific nutrients such as vitamin A or B 12. Bleeding or purpura related to vitamin K deficiency may be present. Aphthous stomatitis, cheilosis, and glossitis are not infrequent. In adults, symptoms in celiac disease may be precipitated by pregnancy, respiratory therapy, and gastric surgery. Gluten-sensitive enteropathy is seen in patients with dermatitis herpetiformis. Celiac disease has also been associated with other skin diseases, such as psoriasis, eczema, cutaneous amyloid, and mycosis fungoides.
The diagnosis of celiac disease can be made when physicians test for serum antibodies associated with celiac disease in patients with minimal GI complaints or indicators of malabsorption, such as vitamin deficiencies. Immunoglobulin A (IgA) and IgG antibodies to gliadin are elevated in 75% to 85% of patients with celiac disease. These antibodies may also be elevated in some patients with inflammatory bowel disease or liver disease. Endomysial antibody is also elevated in a large percentage of patients with celiac disease.
The antigen for antiendomysial antibody is tissue transglutaminase (tTG). An enzyme-linked immunosorbent assay (ELISA) test for IgA anti-tTG is reported to have a sensitivity of 90% to 95% for the diagnosis of celiac disease. Other studies have shown less favorable results for antiendomysial antibody. Some patients with celiac disease have IgA deficiency, so this test will have false-negative results in these patients. Small bowel biopsies and follow-up biopsies after gluten withdrawal are usually required for a definitive diagnosis.
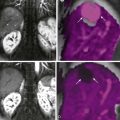
Biopsy specimens in celiac disease typically reveal a loss of intestinal villi associated with crypt hyperplasia and infiltration of the lamina propria by plasma cells and lymphocytes ( Fig. 43-3 ). Lesser degrees of villous atrophy may be present, however, ranging from mild villous flattening to partial villous atrophy or a flat mucosa. Because other diseases may cause loss of intestinal villi, biopsy-proven reversal of mucosal changes after gluten withdrawal is essential for confirming this diagnosis. Nevertheless, some patients may develop disease that is refractory to gluten withdrawal.