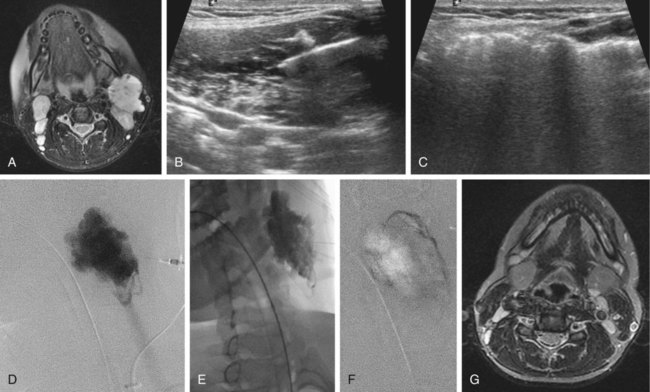
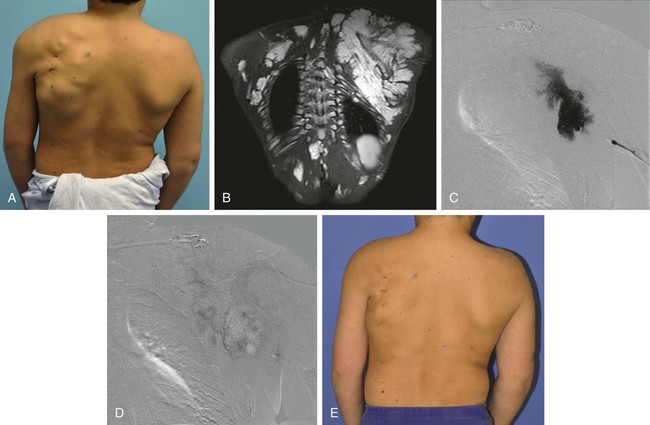
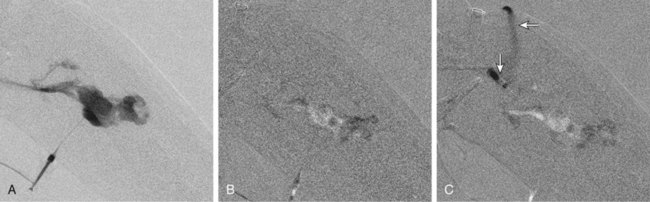
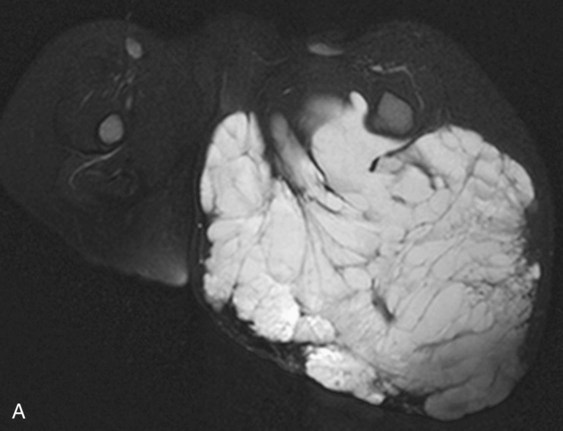
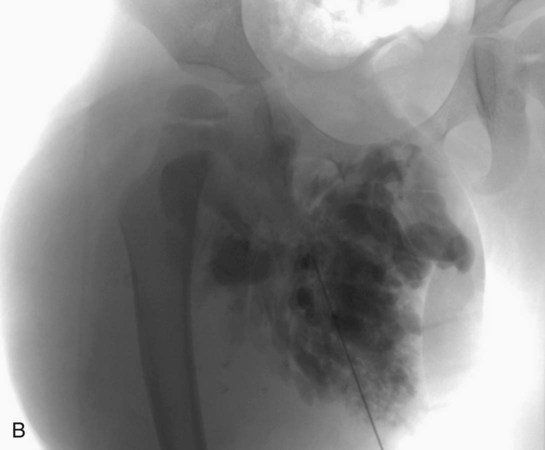
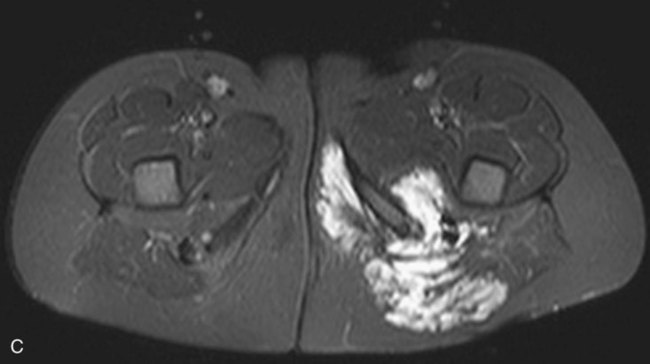
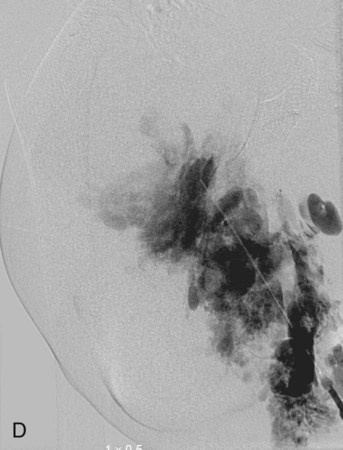
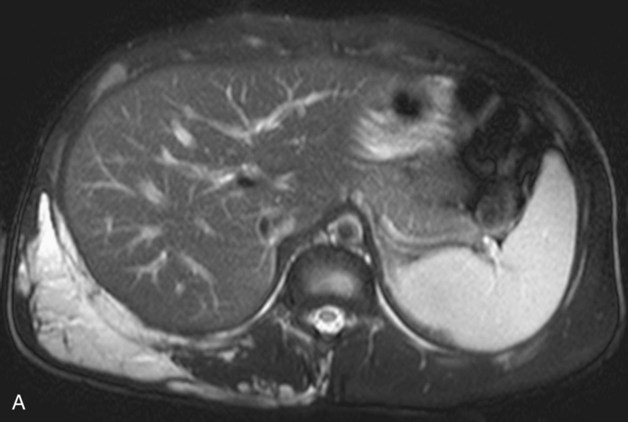
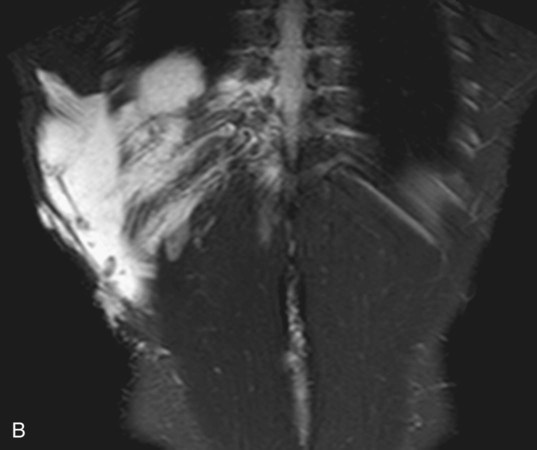
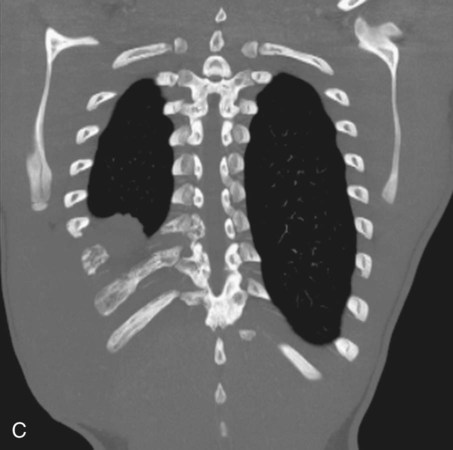
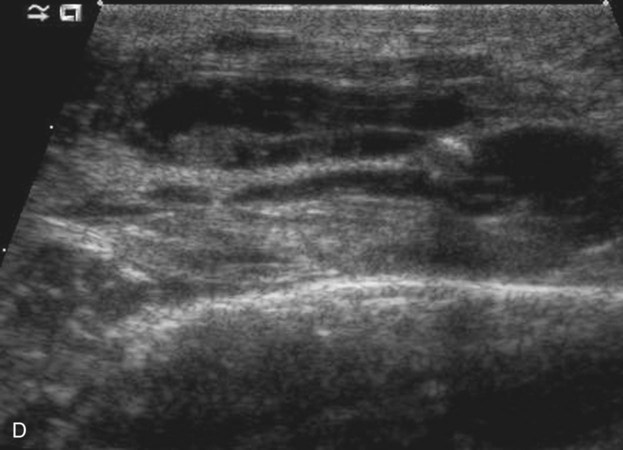
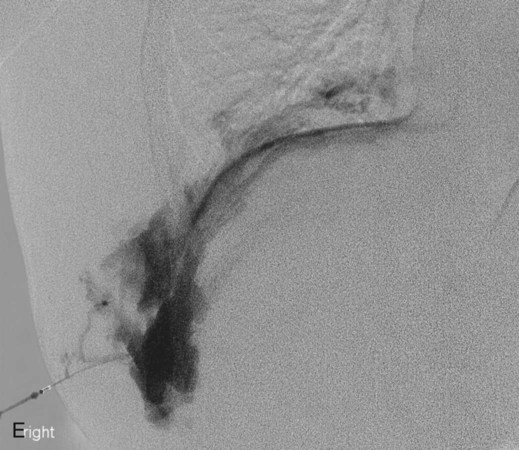
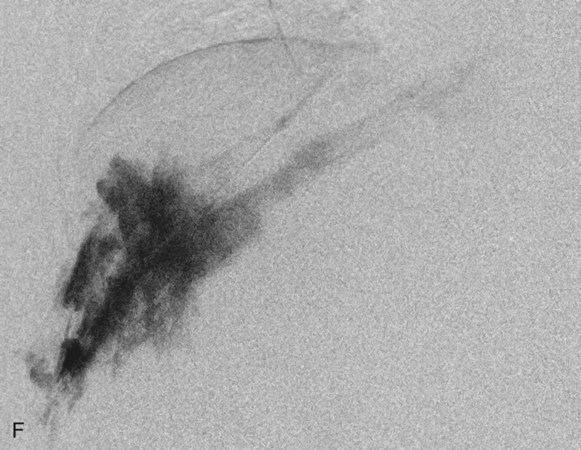
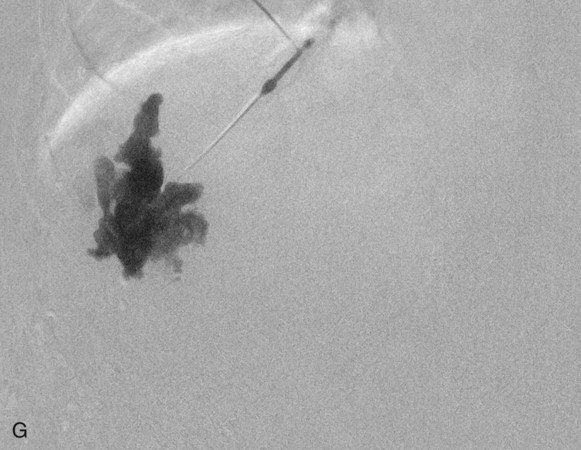
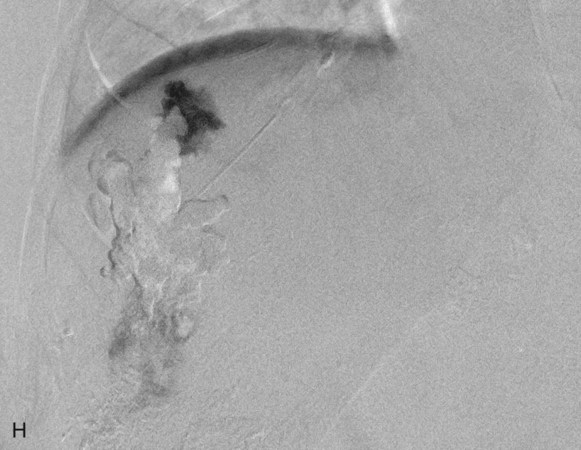
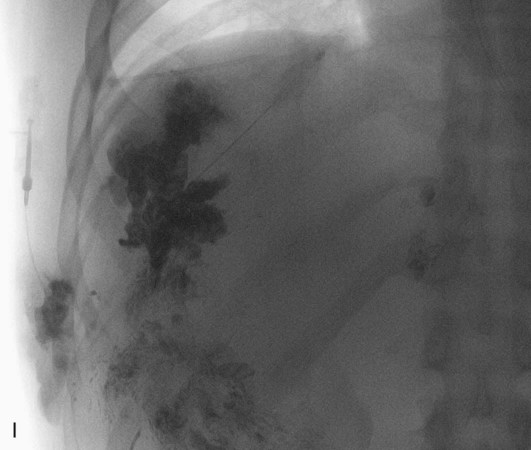
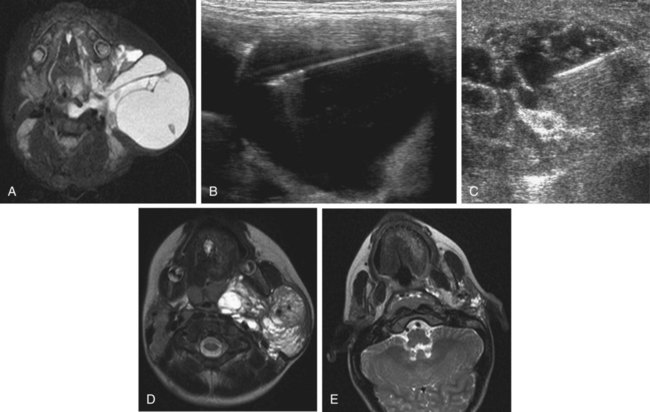
Chapter 44 The goal of sclerotherapy is to obliterate abnormal channels by damaging the endothelium, thereby resulting in subsequent inflammation and fibrosis. Venous malformations (VMs) are caused by abnormal development of the vein wall, with thinning and asymmetric disruption of the smooth muscle layer of the vein in association with endothelial cell abnormalities. This results in progressive, often asymmetric, dilation of the affected channels. Associated absence or insufficiency of valves in the conducting veins contributes to swelling. Affected channels become progressively enlarged, and the resulting stagnation of blood causes thrombosis, swelling, and pain. Most VMs undergo a continuous cycle of spontaneous thrombosis and thrombolysis.1 Calcification of thrombi results in formation of phleboliths. Symptoms and signs include blue or purple cutaneous lesions, swelling with dependency or effort, pain, deformity, and consumption coagulopathy. Pulmonary embolism can occur, especially when the conducting venous channels are malformed. The angioarchitecture of VMs includes focal (Fig. 44-1), multifocal (Fig. 44-2; also see Fig. e44-1 Diffuse VMs involve multiple tissue layers, usually including muscle, subcutaneous fat, skin, and sometimes bone. In diffuse lesions, the malformed veins are nonsequestered and communicate directly with the main conducting veins, which are frequently also abnormal.4 Diffuse VMs are difficult to treat effectively because injected sclerosant can directly enter the circulation and potentially cause deep venous thrombosis, pulmonary embolism, or systemic effects of ethanol. Recanalization is also more likely than after sclerotherapy for nonsequestered lesions.5 Some patients with diffuse VMs have focal eccentric varices that can exert a considerable mass effect on adjacent structures. These varices can be obliterated by endovascular treatment. Multifocal lesions are most commonly seen in familial forms of VM, including blue rubber bleb nevus syndrome (see Fig. 44-2 and Lymphatic malformations (LMs) result from regional maldevelopment of lymphatic channels and include cystic and channel-type anomalies. Cystic LMs are generally classified into three groups: microcystic (cystic components <1 to 2 cm diameter), macrocystic, and combined forms. Macrocystic lesions are most common in the neck, axilla, and pelvis (Fig. 44-4). LMs are frequently combined with cutaneous capillary malformations (capillary-lymphatic malformations [CLMs]) and anomalies of conducting venous channels (LM with venous dilation, lymphatic-venous malformations [LVMs], or capillary-lymphatic-venous malformations [CLVMs]). Generally, LMs manifest either as focal mass lesions (macrocystic) or as diffuse tissue swelling or overgrowth.6 Unlike VMs, they do not expand with the Valsalva maneuver but rather expand or swell intermittently, especially in association with systemic viral illness. Swelling can occur acutely as a result of infection or bleeding into the lesion. Bleeding is presumably due to either adjacent abnormal venous channels or rupture of small arteries, frequently seen in the septa. Sepsis is most frequently a problem with lesions close to the alimentary tract (e.g., face, pelvis). Cutaneous extensions of LMs are manifested as vesicles that may leak clear, bloody, or chylous fluid. Diffuse and multifocal LMs (generalized lymphatic anomaly) are composed of incompetent lymphatic channels, often associated with chylous reflux and leaks. Life-threatening chylothorax or chylous ascites may develop in patients with generalized lymphatic anomaly. Gorham syndrome (vanishing bone disease) is a type of CLM associated with increased osteoclastic activity and progressive osteolysis of affected bone, most often in the shoulder and pelvis. Magnetic resonance imaging (MRI) is the best technique to confirm the diagnosis and extent of low-flow vascular malformations.7,8 VMs and LMs are hyperintense on fluid-sensitive sequences and do not have fast-flow components on gradient-recalled echo sequences. VMs enhance inhomogeneously, whereas LMs typically show minimal or rim enhancement. Arteriography is not routinely necessary. Pulmonary hypertension and atrial septal defects are contraindications to sclerotherapy for VMs unless the malformation is completely sequestered from the venous circulation.9,10 Relative contraindications include diffuse involvement of a closed space, such as the orbit or a muscle compartment (because of the risk of compartment syndrome), confluent or nonsequestered VMs, and severe consumption coagulopathy. The latter can often be corrected or improved with use of anticoagulants for 2 weeks before treatment. In patients with vascular malformations involving or adjacent to the airway, precautions for airway protection must be taken. Patients who have had cardiovascular complications from ethanol should not receive this agent again. Those with lesion-related neuropathy may be made worse by sclerotherapy. Low-flow vascular malformations are best treated in an angiography suite with road-mapping capability and anesthesia support. Ultrasound equipment with high-frequency probes and sterile probe covers and gel is useful. An automated tourniquet system with sterile cuffs is important for control of flow in some VMs of the limbs. Sclerosants include 95% to 98% ethanol, 3% sodium tetradecyl sulfate (STS), sodium morrhuate, ethanolamine, and bleomycin for VMs, and doxycycline, OK-432, bleomycin, or ethanol for LMs. Ethiodol or Lipiodol can be used to opacify sclerosants with minimal dilution. An agent that combines ethanol and ethyl cellulose for greater viscosity has been developed but is not yet clinically available.11 Useful needles include angiocatheters, 21-gauge single-wall needles, and 25- and 27-gauge butterfly needles. Plastic connecting tubes and three-way stopcocks are useful for hand-injected imaging. If oily contrast medium is used, plastic stopcocks must be checked for compatibility (see Sclerotherapy is performed by direct percutaneous cannulation of vascular channels, usually with a simple needle.3,12–16 Previously obtained MRIs are used to determine the location and extent of the lesion and are available in the procedure room to help plan the percutaneous access. Sonographic guidance of needle placement is useful (see Fig. 44-1, B). MRI guidance is feasible and may be advantageous for deep lesions not easily seen with sonography. Small localized lesions can be injected with detergent sclerosants under regional anesthesia. Patients with extensive VMs and those undergoing ethanol sclerotherapy are anesthetized, pretreated with corticosteroids, and have a Foley catheter placed. Patients undergoing sclerotherapy for VMs must be kept well hydrated to compensate for the hemolysis caused by the sclerosant.
Management of Low-Flow Vascular Malformations
Clinical Relevance
![]() ), and diffuse forms (Fig. 44-3; also see Fig. e44-2). The degree to which the lesion communicates with adjacent conducting veins is the key factor in planning treatment.2,3 VMs with minimal communication can be considered “sequestered,” whereas those with free drainage are confluent or “nonsequestered” (see Fig. 44-3 and
), and diffuse forms (Fig. 44-3; also see Fig. e44-2). The degree to which the lesion communicates with adjacent conducting veins is the key factor in planning treatment.2,3 VMs with minimal communication can be considered “sequestered,” whereas those with free drainage are confluent or “nonsequestered” (see Fig. 44-3 and ![]() Fig. e44-2). Focal lesions may be intramuscular, cutaneous, or mucosal and usually consist of collections of abnormal interconnecting channels or spaces that are sequestered or drain through fairly small channels to normal adjacent conducting veins. This type of lesion is characterized by focal pain or a sensation of fullness with dependency, after exercise, or on arising in the morning (due to stasis). These lesions are easily and effectively treated by injection of sclerosant.
Fig. e44-2). Focal lesions may be intramuscular, cutaneous, or mucosal and usually consist of collections of abnormal interconnecting channels or spaces that are sequestered or drain through fairly small channels to normal adjacent conducting veins. This type of lesion is characterized by focal pain or a sensation of fullness with dependency, after exercise, or on arising in the morning (due to stasis). These lesions are easily and effectively treated by injection of sclerosant.
![]() Fig. e44-2), mucocutaneous familial VMs, glomuvenous malformation, and Maffucci syndrome.
Fig. e44-2), mucocutaneous familial VMs, glomuvenous malformation, and Maffucci syndrome.
Contraindications
Equipment
![]() Fig. e44-2).
Fig. e44-2).
Technique
Vascular Malformations
Anatomy and Approach
Management of Low-Flow Vascular Malformations