T-stage
Breslow thickness
Excision margin
Level and grade of evidence
pTis
In situ melanoma
5 mm
III, B
pT1
<1.0 mm
1 cm
Ib, A
pT2
1.0–2.0 mm
1–2 cm
Ib, A
pT3
2.0–4.0 mm
1–2 cm
Ib, A
pT4
>4.0 mm
2 cm
Ib, B
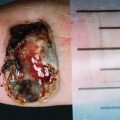
Lesions excised with a margin less than the recommended criteria should be re-excised to achieve these margins. A flap repair or skin graft may be necessary when tissue flexibility is limited to achieving an adequate margin. Referral to a specialist centre should be considered for all cases with melanomas greater than 1 mm thick.
3.2 Sentinel Lymph Node Biopsy
Sentinel lymph node biopsy (SLNB) should be considered in primary melanomas greater than 1.0 mm thick as it provides staging and prognostic information [4, 5, 9, 10]. Patients who have a positive SLNB have tradionally been offered completion lymphadenectomy or referred to a specialist centre to discuss further treatment options [9]. Results from the MSLT-1 study did not demonstrate a significant difference between the 10-year melanoma-specific survival in the SLNB and the observation arms (81.4% versus 78.3%, p = 0.18) [11]. Therefore, there is uncertainty about whether routine lymphadenectomy improves outcome, and this is currently being assessed in the MSLT-2 study [9].
3.3 Management of Locoregional Recurrent Melanoma
Surgery is the treatment of choice for single local or regional metastases [12]. In-transit and local metastases can be managed with localised treatment modalities [11, 13]. Isolated limb infusion (ILI) or perfusion (ILP) with melphalan alone or combined with actinomycin-D can be considered for recurrence on a limb with multiple or rapidly progressive deposits unsuitable for local treatments [14]. ILI is less invasive than ILP but may be less effective [15, 16]. Suspected local regional nodal recurrence should be confirmed by fine needle biopsy. Lymph node dissection can be undertaken for clinically involved nodes with no previous dissection [9, 17].
3.4 Radiotherapy
Adjuvant radiotherapy can be considered in selected cases with narrow or positive margins or where re-excision of the primary disease is unsuitable [18]. The use of adjuvant radiotherapy following nodal resection with adverse pathological features can be considered in carefully selected cases but remains controversial. It is associated with reduced lymph node field recurrence but has no impact on relapse-free survival or overall survival. Its benefits must be weighed up against the risk of regional toxicity, lymphedema and reduced quality of life [19]. Concurrent treatment with interferon alpha significantly increases toxicity and should be avoided [20]. Palliative radiotherapy to symptomatic metastatic or unresectable nodal, satellite or in-transit disease may offer some benefit. Stereotactic radiosurgery and/or whole brain radiation therapy either as adjuvant or the primary treatment can be considered in patients with cerebral metastases.
3.5 Adjuvant Systemic Therapy of Melanoma
Patients with resected stage 1–3 melanoma should be followed up by clinical surveillance [21]. Sentinel lymph node biopsy should be considered for disease stratification and is often used to determine eligibility for adjuvant clinical trials. Adjuvant therapy with high-dose interferon alpha can be discussed with patients with stage III, high-risk disease, carefully outlining the risks and benefits associated with this therapy before proceeding [22].
3.6 Management of Metastatic Melanoma
Advanced melanoma should be profiled for the presence of a driving mutation. BRAF V600 mutation is detected in approximately 40% of melanomas [23]. Patients with acral or mucosal melanoma that do not contain a BRAF mutation should be assessed for a KIT mutation.
Treatment with either a targeted therapy or immunotherapy should be considered as first-line therapy depending on the mutational status of the tumour (see Fig. 3.1 and Table 3.2) [24, 25]. Immunotherapy can be used in the first line in both BRAF-mutant and wild-type patients. Cytotoxic chemotherapy can be considered for palliative disease control but has never demonstrated to improve survival [26].


Fig. 3.1
Systemic treatment pathway for advanced melanoma
Table 3.2
Common side effects with systemic therapy
Drug | Common associated toxicities |
|---|---|
Vemurafenib | Skin-related toxicity, keratoacanthomas, low-grade squamous cell carcinomas, photosensitivity, elevated liver enzyme, arthralgia |
Dabrafenib | Skin-related toxicity, keratoacanthomas, low-grade squamous cell carcinomas, fever, fatigue, arthralgia, headaches |
Combined dabrafenib and trametinib | Fever, chills, fatigue, headaches |
Ipilimumab | Immune-related toxicities including skin toxicity, colitis, endocrinopathies, hepatitis, neuropathy |
Trametinib | Rash, diarrhoea, peripheral oedema |
Dacarbazine | Modest immunosuppression, mild fatigue and nausea |
Temozolomide | Modest immunosuppression, mild fatigue and nausea |
Carboplatin/paclitaxel | Immunosuppression, peripheral neuropathy, alopecia, fatigue and nausea |
3.6.1 Targeted Systemic Therapy with MAPK Pathway Inhibition
Approximately 40% of metastatic melanomas have a V600 BRAF mutation that activates the MAPK pathway (see Fig. 3.2). Treatment with a BRAF inhibitor can produce rapid tumour regression with significantly improved progression-free survival, overall survival and intracranial activity in BRAF- mutated disease [27]. The median time to relapse on monotherapy is around 6 months [28–30]. Combination with a downstream MEK inhibitor improves response, delays resistance and reduces skin-related toxicities but increases the frequency of pyrexia [31–33].


Fig. 3.2
MAPK pathway
3.6.2 Immunotherapy
Immunotherapy should be considered in patients whose melanoma is BRAF wild type. Ipilimumab, an anti-CTLA-4 antibody, potentiates T cell function. It has demonstrated improved overall survival in metastatic melanoma with a median survival of 10–11 months and long-term durable responses in 19% at 4-year follow-up [24, 25]. The programmed death-1 (PD-1) protein is another key immune checkpoint inhibitor expressed by activated T cells that mediate immunosuppression. PD-1 functions primarily in the peripheral tissues where T cells may encounter ligands (PD-L1 and PD-L2) expressed by tumour cells and/or stromal cells (see Figs. 3.3 and 3.4). There are three anti-PD1 monoclonal antibodies that have demonstrated activity in advanced melanoma in clinical trials, pembrolizumab, nivolumab and atezolizumab. Anti-PD1 therapy has reported objective response rates in the range of 26–31% [35, 36]. Anti-PD1 monoclonal antibodies are now approved for the treatment of advanced melanoma, both as single agents and in combination with ipilimumab. Responses to immunotherapy are typically slower than with targeted therapies and transient apparent worsening of disease on imaging before disease stabilisation or tumour regression may be seen. A new immune-related criterion has been defined to evaluate immunotherapy responses [37].



Fig. 3.3
Immunotherapy mechanisms of action for CTLA-4 and PD1 blockade

Fig. 3.4




Therapeutic immunotherapy checkpoint targets for T cell stimulation. Adapted from Mellman et al. (2011) [34]
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree