Tumor size
SRS (%)
SRT (%)
Total (%)
Decreased
22 (34.9)
24 (33.3)
46 (34.1)
No change
36 (57.1)
46 (63.8)
82 (60.7)
Increased
5 (7.9)
2 (2.7)
7 (5.2)
Total
63 (100)
72 (100)
135 (100)
Tumor control
58 (92)
70 (97.2)
128 (94.8)
Table 20.2
Clinical follow-up results
Neurologic findings | SRS (%) | SRT (%) | Total (%) |
|---|---|---|---|
Improved | 22 (35) | 21 (32.3) | 43 (33.5) |
Unchanged | 36 (57.1) | 42 (64.6) | 78 (60.9) |
Worsened | 5 (7.9) | 2 (3.1) | 7 (5.4) |
Number of cases | 63 (100) | 65 (100) | 128 (100) |
Table 20.3
Tumor growth control after stereotactic radiation of atypical meningiomas
Tumor size | SRS (%) | SRT (%) | Total (%) |
|---|---|---|---|
Decreased | 1 (6.2) | 1 (20) | 2 (9.5) |
No change | 4 (25) | 2 (40) | 6 (28.5) |
Increased | 11 (68.7) | 2 (40) | 13 (62) |
Total | 16 (100) | 5 (100) | 21 (100) |
Tumor control | 5 (31.2) | 3 (60) | 8 (38.1) |
The analysis of the long-term follow-up (mean, 72.5 months) group of 32 patients revealed tumor growth control rate of 92.3 % and 100 % for the SRS- and SRT-treated patients, respectively. Worsening of previous neurologic deficit was identified in two (7.9 %) patients treated with SRS. No complications were found in the SRT patients [42].
De Salles et al. [43] studied the role of SRS and SRT in the management of skull base meningiomas and proposed a grade system (see the section “Patient Algorithm” later). They studied 40 patients (34 SRS; 6 SRT) treated during an 8-year period. Tumor control rates were 90 % for grade I, 86 % for grade II, 86 % for grade III, 42 % for grade IV, and no control for grade V (Table 20.4). The control rate correlated well with the tumor grade (p < 0.036). Additionally, the complication rate was 7.5 % (three cases) occurring only in SRS treatments. Since this report, grades III, IV, and V are treated with SRT at our institution.
Table 20.4
Tumor control rates for cavernous sinus meningiomas according to the UCLA grading system
Grade | Meningioma radiologic aspect | Tumor control (%) |
|---|---|---|
I | Confined to the cavernous sinus | 90 |
II | Involvement of the petroclival region without brain-stem compression | 86 |
III | Extension to and compression of the optic nerve, chiasm, or tract | 86 |
IV | Involvement of the petroclival region with compression of the brain stem | 42 |
V | Extensive involvement of both cavernous sinus | 0 |
The experience of treatment of cavernous sinus meningiomas with SRT was recently reviewed by Selch et al. [44]. They studied 45 patients treated with a median prescribed dose of 50.4 Gy delivered to a median tumor volume of 14.5 cm3. The 3-year progression-free survival rate was 97.4 % and the preexisting neurologic complaints improved in 20 % of patients. One patient had an ipsilateral cerebrovascular accident 6 months after SRT, although this complication is not likely related to the procedure.
Stereotactic Radiosurgery
SRS has become a well-accepted modality for the treatment of a number of neurologic indications, ranging from primary and metastatic malignancies to benign tumors, arteriovenous malformations (AVMs), pain, and other neurologic conditions. Unlike conventional fractionated radiotherapy, SRS does not exploit differences in the repair capacities of normal and neoplastic cells after repeated exposure to sublethal doses of irradiation. SRS achieves a therapeutic ratio by virtue of the steep dose gradient between the isocenter and surrounding normal parenchyma, inevitably produced by the convergence of multiple small radiotherapy beams. The biologic effect is irreparable cellular damage and vascular occlusion within the high dose volume.
Meningiomas have several characteristics that make them excellent tumors for radiosurgery [45]. First, they usually grow slowly, which allows time for the effects of irradiation to materialize. Second, they are easily imaged with both computed tomography (CT) and magnetic resonance imaging (MRI), thereby facilitating accurate stereotactic targeting and appropriate dose planning. Finally, a high dose with rapid fall-off can be directed to the imaging-defined tumor margin with confidence because meningiomas are usually well demarcated and rarely invade surrounding brain tissues (Fig. 20.1). The application of stereotactic radiation using all available techniques, including linear accelerator (linac) [41, 43, 44, 46–48]. Gamma Knife [49–54], and proton beam [55], has been reported in the literature.


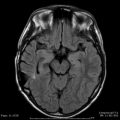
Fig. 20.4
(a) MRI scan showing an incidental finding of a small falx meningioma in a 77-year-old woman with high operative risk. The lesion had a slight increase in size during 1 year of follow-up and was treated with 14 Gy, 90 % isodose line. (b) Stable size of the tumor 40 months after treatment
The effectiveness of SRS and the risks of complication depend on the size of the tumor. Lesions larger than 3 cm in diameter must be treated with significantly lower doses of radiation to avoid excessive risk of complications. Such small radiation doses may make radiosurgery ineffective. Tumor location also affects the treatment decision, depending on the proximity to eloquent structures (especially the optic apparatus). Single-dose SRS is indicated for meningiomas smaller than 3 cm or 20 cm3 in volume and with a minimal distance from the optic apparatus of between 2 and 4 mm [54, 56, 57]. Furthermore, tumors causing significant brain-stem compression may not be suitable for single-dose radiosurgery because a slight swelling during the course of tumor necrosis induced by radiosurgery may not be tolerable. Many alternatives were attempted in order to overcome such limitations, usually resulting in treatment failure. This included two-staged procedures [49, 58] or a decrease in the ideal peripheral dose [59].
A lack of homogeneity in the radiosurgery dose has been a controversial issue in SRS treatment of brain tumors. Dose homogeneity has been sacrificed to obtain conformal tumor planning, thereby avoiding nearby normal structures. However, those structures encased by the tumor, such as cranial nerves, the carotid artery, or even the optic apparatus, may be jeopardized with inhomogeneous radiation treatment. Multiple isocenters restrict the prescription to low isodose volumes with a very high gradient of dose between the periphery and interior of the tumor. According to Nedzi et al. [60], dose inhomogeneity ≥5 Gy was the most significant factor associated with morbidity after SRS.
Within the past few years, however, the development of miniature multileaf collimators has altered the way in which radiosurgery is performed with linac systems. This technique allows the beam to conform to the target closely and exclude normal structures. In addition to high target conformity, the “single isocenter shaped beam approach” results in a homogeneous dose within the target volume, as well as a highly efficient planning and treatment process.
SRS has already proved to be effective in the treatment of meningiomas. Kondziolka et al. [52] evaluated the long-term outcome of 85 patients (90 % of them skull base located) who underwent Gamma Knife radiosurgery. Tumor control was obtained in 93 % of patients. Tumor resection was necessary in five (5.8 %) patients because of delayed tumor growth or persistent symptoms. New or worsened neurologic deficits occurred in five (5.8 %) patients during the follow-up.
Thirty-four percent to 71 % of meningiomas treated by SRS are expected to decrease in size, and 37–57 % are expected to stabilize [41, 51, 61, 62]. DiBiase et al. [63] studied the factors predicting local tumor control after Gamma Knife radiosurgery in 162 patients harboring meningiomas. The median prescribed dose was 14 Gy to the 50 % isodose line and the median tumor volume was 4.5 cm3. The 5-year disease-free survival was 86.2 %. The factors correlated with a worse prognosis in their study were male patients, conformity index less than 1.4, and tumor volume greater than 10 cm3. However, patients who were treated with less conformal plans that covered the dural tail had better outcomes (Fig. 20.2).


Fig. 20.5
(a) MRI scan from a 74-year-old German woman with an incidental found meningioma in the tip of the left temporal fossa. She was followed with serial MRIs and growth was documented. The patient refused surgery and accepted radiosurgery. (b) Treatment plan showing inclusion of the dural tail within the prescription isodose line (90 %, 14 Gy, shaped beam)
Hakim et al. [46] published the results of 127 patients with 155 meningiomas treated by SRS with linac, including atypical and malignant meningiomas. The median marginal dose was 15 Gy and the median volume was 4.1 cm3. Freedom from progression was observed in 84.3 % of patients at a median time of 23 months. The actuarial tumor control for the patients with benign meningiomas was 89.3 % at 5 years. Permanent complications were observed in 4.7 % of patients.
SRS for patients with small- to medium-sized meningiomas was recently reported to provide tumor control equivalent to Simpson grade 1 resection. Pollock et al. [64] compared two groups of patients with lesions smaller than 35 mm. The 62 patients who underwent SRS with a mean margin dose of 17.7 Gy had no statistically significant difference in the 3- and 7-year actuarial PFS when compared with 57 patients who underwent complete surgical resection, including their dural attachments and bone abnormalities (Simpson 1) with no radiation. Furthermore, SRS provided a higher PFS rate compared with that in patients in whom resections were Simpson grade 2 (p < 0.05) and grade 3–4 (p < 0.001). The complication rate was 10 % for SRS and 22 % for the surgical group (p = 0.06).
Patients treated by radiosurgery for meningioma must be followed carefully. Flickinger et al. [65] studied a group of 219 meningiomas without pathologic verification. The actuarial rate of identifying a diagnosis other than meningioma was 2.3 % at 5 and 10 years. Nakatomi et al. [66] reported one patient treated by SRS for a supposed cavernous sinus meningioma that required surgical resection 17 months after because of recurrence. The postoperative diagnosis was primary cavernous sinus malignant lymphoma.
Kim et al. [67] recently analyzed the role of Gamma Knife radiosurgery in 23 patients with 26 superficially located meningiomas. The median tumor volume was 4.7 cm3, and the mean margin dose was 16 Gy at the 50 % isodose line. The local control rate was 95 % with stable MRI imaging in 65 % of the cases. Symptomatic and transient peritumoral high signal on T2-weighted magnetic resonance (MR) images was observed in 43 % of patients. This complication was associated with a high integral dose and a large tumor volume. Moreover, tumor shrinkage was more prominent in the patients with symptomatic high signal (p = 0.03).
MRI showed peritumorous imaging changes in 24 % of patients treated by SRS in the series published by Chang et al. [61]. Their overall rate of symptomatic edema was 9.3 %. Furthermore, these imaging findings were related to tumor dose in univariate analysis, and with tumor location in the multivariate analysis. Radiation-induced imaging changes were more often seen in convexity, parasagittal, and falx meningiomas than in those of the skull base.
Skull Base Meningiomas–SRS
Meningiomas in the skull base account for the largest group of incompletely resected lesions usually due to cavernous sinus involvement, brain-stem adhesion, and infiltration of adjacent structures. Complete excision in these situations is unlikely without causing morbidity [68–70].
Meningiomas are the most frequent benign tumors treated by radiosurgery, and the majority of them are located on the skull base (Fig. 20.3). Liscak et al. [71] studied 176 patients with meningiomas located on the skull base. They obtained local control in 98 % of patients and a persistent morbidity rate of 4.5 %. The rate of radiation-induced edema was 11 %. Significantly lower edema occurrence was observed when the dosage to the tumor margin was lower than or equal to 14 Gy.


Fig. 20.6
(a) MRI scan showing a cavernous sinus and petro-clinoid ligament meningioma in a 71-year-old woman with pain in V2 and V3 region. (b) Treatment plan with multiple isocenters, 16 Gy, 50 % isodose line. (c) MRI scan showing stable image after 9 years of follow-up. The patient is requiring only sporadic medication for facial pain
Chuang et al. [72] studied the linac-based SRS for 43 unresectable or partially resected meningiomas in the base of the skull. The 7-year local control in the SRS-alone group and surgical excision with SRS group was 100 % and 84.4 %, respectively (p = 0.21).
Using a mean margin dose of 13 Gy with a Gamma Knife unit for treatment of 32 petroclival meningiomas, Roche et al. [73] achieved local control in all patients. Although no new cranial deficit was observed, two patients developed hemiparesis due to pontine infarction.
Spiegelmann et al. [48] studied 42 patients with meningiomas involving the cavernous sinus treated with linac. A mean dose of 14 Gy was delivered to the tumor margin and the median tumor volume was 8.2 cm3. Tumor control was observed in 97.5 % of the patients. Cranial nerve complications included new trigeminal neuropathy in 4.7 % and a new visual defect in 2.8 %.
Despite the dosimetric advantages of SRS, permanent cranial nerve deficit is more common in the treatment of cavernous sinus meningiomas. Tishler et al. [74] studied 62 patients (42 with meningiomas) inside or near the cavernous sinus treated by SRS. New cranial neuropathies were found in 19 % of patients. Additionally, all patients who developed optic system neuropathy (6.4 %) received radiation doses higher than 8 Gy to optic apparatus.
Leber et al. [75] correlated the doses delivered in the treatment of cavernous sinus lesions by SRS with incidence of optic neuropathy. The actuarial rate was zero for patients who received a dose of less than 10 Gy, 26.7 % for patients receiving 10 to less than 15 Gy, and 77.8 % for those who received doses of 15 Gy or more (p < 0.0001).
Stafford et al. [76] reported 218 parasellar SRS procedures with 73 % of patients receiving more than 8 Gy to a short segment of the optic apparatus. The overall incidence of radiation neuropathy was 1.9 %; the risk of neuropathy was 1.1 % for patients receiving 12 Gy or less.
Morbidity after SRS is strongly associated with tumor volume and location [15, 77]. For these reasons, application of SRS to cavernous sinus meningiomas has generally been restricted to tumors <3 cm in greatest dimension and located several millimeters from the optic apparatus [56, 77–79]. These are the grade I and grade II of the UCLA classification [43]. Tumors grade III, IV, and V are candidates for SRT.
See Table 20.5 for doses used in stereotactic radiosurgery for intracranial meningiomas.
Table 20.5
Doses utilized in radiosurgery for intracranial meningiomas
Author, Year | Peripheral Dose | Variation (Gy) | Technique |
|---|---|---|---|
Hakim, 1998 [46] | 15 | 9–20 | LINAC |
Liscak, 1999 [59] | 12 | 10–14 | GK |
Kondziolka, 1999 [51] | 16 | 9–25 | GK |
Shafron, 1999 [47] | 12.7 | 10–20 | LINAC |
Stafford, 2001 [54] | 16 | 12–36 | LINAC |
Villavicencio, 2001 [114] | 15 | 12–18.5 | LINAC |
Friedman, 2005 [115] | 16 | 10–20 | LINAC |
Hasegawa, 2007 [116] | 13 | 7.5–17 | GK |
Pollock, 2012 [117] | 16 | N/A | GK |
Stereotactic Radiotherapy
SRT is a treatment option that may be undertaken when the risk of SRS is unacceptably high; for instance, in cases in which tumors involve the optic pathways, the brain stem, or lesions with larger volume near eloquent structures.
This technique combines the physical dose localization advantage of SRS with the radiobiologic benefits of dose fractionation [80]. Unlike SRS, the treatment planning fiducial system and patient immobilization device can be applied noninvasively [81]. The accuracy of patient repositioning during SRT has been documented [82–84]. A total fractionated dose of ionizing radiation, known from conventional radiation therapy experience to be effective for meningioma yet tolerated by the central nervous system, can be delivered by the SRT technique.
Lo et al. [85] studied a group of 18 patients treated by SRT for meningiomas with a large volume or adjacent to eloquent structures. The median dose was 54 Gy and the median volume was 8.8 cm3. The 3-year local control rate was 92.3 %; one complication was reported (5.5 %). These findings were not statistically significant, different when compared with another group of 35 patients who underwent SRS with a median dose of 14 Gy delivered to a median volume of 6.8 cm3.
The application of SRT was recently reviewed by Zabel et al. [86] in 317 patients with benign and atypical meningiomas in all intracranial sites. The median total dose was 57.6 Gy delivered to a median volume of 33.6 cm3. The overall local control rate was 93.1 %. As expected, the local tumor failure was significantly greater in patients with WHO grade 2 meningiomas than in patients with WHO grade 1 or unconfirmed histology (p < 0.002). Moreover, patients with a tumor volume larger than 60 cm3 had a higher recurrence rate (15.5 %) when compared with patients with tumor volumes smaller than 60 cm3 (p < 0.001). They observed worsening of preexisting symptoms and new clinical symptoms in 8.2 % and 2.5 % of patients, respectively.
Skull Base Meningiomas–SRT
Skull base meningioma patients for whom microsurgery and/or SRS are contraindicated seem to be the ideal application of SRT (Fig. 20.4). Alheit et al. [82] studied a group of 24 patients (21 of them harboring skull base meningiomas) treated by SRT with doses ranging from 50 to 55 Gy delivered to a median volume of 21 cm3. The progression-free survival rate was 100 %, and two (8 %) patients experienced early side effects, including one VII nerve palsy and one Addisonian state.


Fig. 20.7
(a) MRI scans of a 52-year-old woman experiencing double vision and lack of balance showing a large meningioma involving the left cavernous sinus, left petroclival region extending into the prepontine and premedullary cisterns. The patient refused surgery and was treated by SRT. (b) Treatment plan using shaped beams with 50.4 Gy in 28 fractions, 90 % isodose line, volume 39.22 cm3. (c) MRI scans showing decrease in tumor size 43 months after treatment
Debus et al. [87] published a large series of 189 patients treated by SRT for skull base meningiomas with a mean radiation dose of 56.8 Gy. The median target volume was 52.5 cm3. The tumor control rates for WHO grade 1 and WHO grade 2 tumors were 98.3 % and 77.7 %, respectively. Preexisting cranial nerve symptoms resolved completely in 28 % of the patients. Only 7 % of patients showed an impairment of preexisting neurologic symptoms, and 1.6 % experienced new clinically significant deficits. These excellent results corroborate the UCLA experience with skull base meningiomas treated with SRT [44]. See Table 20.6 for doses used in stereotactic radiosurgery for intracranial meningiomas.
Table 20.6
Doses utilized in stereotactic radiotherapy for intracranial meningiomas
Author, year | Dose/variation (Gy) | Fractions (Gy) |
|---|---|---|
Andrews, 2002 [93] | 51 (50–54) | 1.8 |
Selch, 2004 [44] | 50.4 (N/A) | 1.8 |
Henzel, 2006 [118] | 56 (50.4–60) | 1.8–2.0 |
Adler, 2006 [119] | 20.3 (N/A) | 2–5 |
Litré 2009 [120] | 45 (N/A) | 1.8 |
Combs, 2012 [121] | N/A (25–68) | 1.6–5 |
Intensity-Modulated Radiation Therapy
The intensity-modulated radiation therapy (IMRT) technique modulates the intensity of the photon beam across the treatment portal. It has shown the ability to conform the dose to complex-shaped target volumes. Baumert et al. [88] made comparisons between the treatment plans of SRT and IMRT for skull base lesions. The conformity index and the sparing of the organs at risk were better for the IMRT plan. The largest improvement was observed for multifocal and irregular tumors.
Pirzkall et al. published the results of IMRT treatment on 20 patients with benign skull base meningiomas. They used a median minimum dose of 40 Gy delivered to a median volume of 108 cm3. In a median follow-up of 36 months, the local tumor control rate was 100 %. The preexisting neurologic symptoms improved in 60 % and worsened in 5 % of patients. They also observed no radiation-induced peritumoral edema or new onset of neurologic deficits.
Interestingly, the application of intensity-modulated stereotactic radiosurgery (IMSRS) for treatment of small skull base lesions was compared with dynamic conformal arc technique (DCA). Superior homogeneity and coverage for DCA was observed. The IMSRS also increased the time for planning, dose delivery, and integral dose to the brain [89].
The intensity-modulated stereotactic radiation therapies (IMSRT or IMSRS) have limited application when treating intracranial meningiomas; specific cases where the eyes or the brain stem are surrounded by the lesion may benefit from this technique (Case Study 20.2).
Special Applications of Stereotactic Radiation
Optic Sheath
Optic nerve sheath meningiomas are uncommon, slow-growing tumors that can be categorized as primary and secondary according to their origin. Although conservative management can be considered, the most common treatment is resection, which may lead to blindness due to damage to the pial vascular supply of the optic nerve [90]. Radiation therapy has been used in recent decades to treat these tumors [91, 92].
SRT has been used as one alternative to avoid radiation-induced optic neuropathy. Andrews et al. [93] treated 30 patients with 33 optic nerve sheath meningiomas with SRT. Local tumor control was observed in all patients, and they observed 92 % of preserved vision among the optic nerves with vision before treatment. Improvement in visual acuity and/or visual field occurred in 42 % of nerves. Visual loss was observed in 2 (6.6 %) patients and 1 (3.3 %) developed optic neuritis responsive to steroids.
Becker et al. studied the role of SRT in 39 patients with either primary or secondary optic nerve sheath meningiomas using a median dose of 54 Gy. The tumor local control was 100 % after a median follow-up of 35 months. The rates of visual field stabilization, improvement, and worsening were 70 %, 25 %, and 4 %, respectively. Additionally, new endocrinologic deficits after treatment were found in 14 % of patients with primary tumors and in 8 % of patients with secondary tumors. The visual improvement observed in patients treated by SRT for optic sheath meningiomas was recently reported to occur within 1–3 months after treatment in 81 % of patients who experienced visual improvement [94]. Homogeneity of the dose distribution becomes important when planning SRT for these tumors, as frequently the optic nerve is surrounded by the tumor. This is possible with shaped beams.
Atypical and Malignant Meningiomas
The role of conventional radiation therapy and brachytherapy in the management of atypical and malignant meningiomas is well established [33, 36–39]. Although many series are focused on stereotactic radiation for treatment of meningiomas, relatively few published studies have defined its application in atypical and malignant histologic features (Fig. 20.5). At our institution, SRS and SRT are used to salvage recurrences after surgery and regional radiation.


Fig. 20.11
(a) MRI scan showing a left frontoparietal dural based recurrent atypical meningioma. (b) Treatment plan with shaped beans (with a left to right inversion), 47.6 Gy, 85 % isodose line in 28 fractions, volume 20.68 cm3. (c) MRI scan showing the tumor increasing in size after 20 months of follow-up. The pathology report revealed malignant progression to WHO grade III meningioma
Ojemann et al. [95], using a Gamma Knife unit, treated 22 patients with primary or recurrent malignant meningiomas. The overall 5-year survival and progression-free survival rates were 40 % and 26 %, respectively. Age (<50 years) and tumor volume (<8 cm3) were significant predictors of better prognosis. Complications were observed in 5 (23 %) patients who developed radiation necrosis.
In the meningioma series published by Stafford et al. [54], 22 patients harboring either atypical (n = 13) or malignant (n = 9) meningiomas were treated by Gamma Knife SRS. They obtained 5-year survival rates of 76 % and 0 % for atypical and malignant meningiomas, respectively. The 5-year local control rates were 68 % and 0 % for atypical and malignant tumors, respectively. Univariate analyses of tumor histologic features demonstrated no correlation between complications arising after radiosurgery.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree