© Springer Science+Business Media Dordrecht and People’s Medical Publishing House 2015
Hongjun Li (ed.)Radiology of Infectious Diseases: Volume 110.1007/978-94-017-9882-2_2424. Nipah Virus Encephalitis
(1)
Department of Radiology, Beijing You’an Hospital, Capital Medical University, Beijing, China
Nipah virus encephalitis is a zoonotic infectious disease caused by Nipah virus (NiV), which is categorized into the genus of Henipavirus and the family of Paramyxoviridae. NiV is a new zoonotic virus that was firstly isolated from deceased patients in Nipah, a village in Perak, Malaysia, in the late 1990s. The virus has been mainly found in Malaysia, Bangladesh, and their surrounding areas in Southeast Asia. It has no pathogenicity to bats but is capable of infecting pigs. In addition, it is highly pathogenic to humans. NiV can cause severe encephalitis and respiratory diseases in animals and humans, with high incidence and mortality. The mortality rate in patients with NiV infection is up to 40–70 %, and therefore, the virus has been listed as one of the biosafety level 4 (P4), the most dangerous, pathogens.
24.1 Etiology
NiV is an RNA virus, which has highly homogeneous genome to Hendra virus (HeV) that was firstly found in Australia in 1994. The genomes of these two viruses have 2,000 more nucleotides than other viruses in the family of Paramyxoviridae. The protein antigenicity of these two viruses is also similar; both of which are classified in the genus of Henipavirus and the family of Paramyxoviridae. However, they are not identical. Among the viruses in the family of Paramyxoviridae, both NiV and HeV are the closest to measles virus. However, due to the long genome of both NiV and HeV and their respective unique sequence, both of their reservoir hosts are bats, and they can infect a variety of hosts. HiV and NeV have been separately classified into a new genus in the subfamily of Paramyxoviridae and the family of Paramyxoviridae, namely, the genus of Henipavirus.
NiV can grow in any of mammalian cell line to cause syncytial-like lesions but cannot grow in insect cell line. The virus can grow well in Vero, BHK, and PS cells but is unstable in external environment. It is sensitive to temperature, disinfectants, and detergents, which can be destroyed at a temperature of 56 °C for 30 min. In addition, it can be inactivated by commonly used disinfectants or detergents.
24.2 Epidemiology
24.2.1 General Introduction to Its Prevalence
During the period from 1998 to 1999, large-scale outbreak of NiV infection in pigs occurred in Malaysia, which resulted in 257 cases of human infection including 105 cases of death. Another two events of NiV infection occurred in Malaysia in June 2000. Meanwhile, due to the import of pigs from Malaysia, 11 cases of NiV infection in humans was reported, including 1 case of death. During the period from 2001 to 2005, five events of NiV infection in humans were reported consecutively, with a total of 69 death cases.
During the prevalence of NiV infection in Malaysia, NiV carried by fox bats may infect pigs with a tiny probability, which then proliferated in pigs in a large quantity and rapidly spread to other pigs via contacts. Humans were then infected via close contacts to these pigs. However, in the events occurring in Bangladesh, most of the NiV infected people had no history of close contacts to pigs, and pigs in these areas also had no sign of NiV infection. Therefore, pigs did not play an important role in the events occurring in Bangladesh. But the initial route of transmission for human infection remains unknown. In the events occurring in Malaysia, no evidence has been found to prove spreading of the virus between humans; while in the events occurring in Bangladesh, conclusive evidence has been found to prove spreading of the virus from person to person.
24.2.2 Source of Infection
Although NiV can infect a variety of livestock by fruit bats, pig is still the main source of infection. Recent reports have demonstrated that NiV can spread via contaminated food or spread from person to person.
24.2.3 Route of Transmission
NiV possibly spreads via body fluid or blood, such as contacts to body fluid from patients. The routes of transmission may include direct contact to urine, body fluid, or tracheal secretions from infected pigs, sharing needle, artificial fertilizing, and vertical transmission from mother to infant. The transmitting route of NiV from person to person is not the major route of its transmission. Mosquitoes and other insects are not the media for its spreading. Although other natural hosts such as hounds, cats, Percheron, wild pigs, and rats could be infected, the virus fails to spread widely due to rapid death of these infected animals. In the outbreaks of NiV infection by humans, pig plays an important role, which spreads the virus via contacts or airborne transmission. After NiV infection by pigs, the virus can multiply in their bodies in large quantities. Viremia may persist for a long period of time, during which the virus may be spread externally via respiratory tract, urine, feces, and saliva. In addition, starlings such as mynas and grackles seek food in the piggery, which often peck ticks on the back of pigs and fly within one piggery or among piggeries. Therefore, starlings may be another route of transmission for NiV.
24.2.4 Susceptible Population
Workers working in piggeries or slaughterhouses have much more chances to contact to diseased pigs due to their job. Therefore, this population is the high-risk population for NiV infection.
24.3 Pathogenesis and Pathological Changes
24.3.1 Pathogenesis
NiV interacts with the host cell surface receptor ephrin B2 via G protein to recognize and adhere to the surface of host cells. Ephrin B2 is ligand of the subgroup Eph B in receptor tyrosine kinases (RTKs). Eph and its ephrin ligands play an important role in the processes of CNS development, angiogenesis, and tumor formation. Ephrin B2 is mainly expressed in neuronal cells, smooth muscle cells, arterial endothelial cells, and capillaries, which is consistent to the tropism of NiV to endothelial cells and the nervous system. Ephrin B2 is conservative in mammals, which may be related to the fact that many mammals are susceptible to NiV. In addition, ephrin B3, one of the homogeneous proteins of ephrin B2, is also capable of independently facilitating NiV gaining its access into cells, which is possibly one of the NiV receptors. After the invasion of NiV into the endothelial cells, it replicates itself rapidly and affects the synthesis of interferon and its functions under effects of multiple proteins such as P, V, W, and C protein to evade the defense system of the host. Meanwhile, it fuses with endothelial cells under the effects of proteins F and G to form endothelial syncytium, with following rupture of the cell to release the virus. The emergence of endothelial syncytium is characteristically an NiV infection, with a detection rate of 25 % by pathological examination in clinically diagnosed cases. In addition, NiV can also infect neurons directly, where it grows and proliferates to cause neurological symptoms.
24.3.2 Pathological Changes
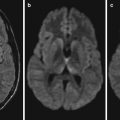
NiV infection can involve any system of human body. By autopsy, the brain is found to be the most seriously affected organ, with possible involvements of the gray matter, white matter, basal ganglia, cerebellum, brain stem, and spinal cord. Other organs may also be involved, including the lungs, heart, and kidneys. The basic pathological changes are multiple organs vasculitis and endothelial cell inflammation. Vasculitis commonly occurs in minor arteries, arterioles, capillaries, and venules, which is characterized by vascular wall necrosis, thrombosis, and infiltration of inflammatory cells like leukocytes and monocytes. The infection of CNS shows extensive and scattering small necrotic foci. By immunohistochemical assays, NiV antigen can be detected in CNS tissues, especially in vascular endothelial cells with vasculitis, endothelial syncytium, and parenchymal cells. A small amount of viral antigens can also be detected in other tissues. The occurrence of death in patients with NiV infection is commonly from extensive focal infarction of brain tissues caused by atrophy of large quantities of infected neurons.
24.4 Clinical Symptoms and Signs
NiV has an incubation period of 1–3 weeks in the human body. Clinically, the patients commonly experience fever and headache for 3–14 days. After that, symptoms of drowsiness and systemic myalgia occur. There are also obvious symptoms of encephalitis and/or meningitis such as neck rigidity, varying degrees of psychiatric symptoms, cerebellar dysfunction, weak or absent deep tendon reflexes, limb paralysis, clonic convulsion, language impairment, visual hallucinations, and auditory hallucination. In 24–48 h, the patients may further develop into coma. The characteristic symptoms of NiV infection are cervical and abdominal muscle spasms. The conditions have varying severity, with commonly neurological symptoms and rarely accompanying respiratory symptoms, hypertension, and tachycardia. Some patients may experience delayed onset of clinical symptoms, with delayed onset of neurological symptoms. In some patients, the neurological symptoms may even occur 4 years after the infection, and during this period, some cases may have no encephalitic symptom or even be asymptomatic. There is also rarely occurring relapse of acute encephalitis after its recovery, which is commonly found several months to 2 years after initial infection. In some cases, its relapse occurs even 4 years after the initial infection. The relapse may be related to persistent survival of the virus in the CNS tissues.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree