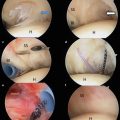
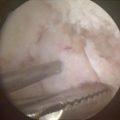
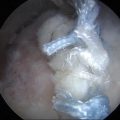
Fig. 23.1
(a–c) MRI and arthroscopic images demonstrating trochlear and radial head lesions (arrows) (a, c: Reprinted from Jans LBO et al. MR imaging findings and MR criteria for instability in osteochondritis dissecans of the elbow in children. Eur J Radiol. 2012;81:1306–10, with permission from Elsevier)
OCD has also been implicated in developmental instability of the elbow, the majority of which is posterolateral instability [15]. Lateral ligament injuries resulting in instability can cause posterior impaction of the capitellum that may be confused with OCD. It is also important not to confuse the normal pseudodefect on the posterior inferior capitellum with an actual lesion. This pseudodefect represents the normal transition of the capitellar cartilage with the nonarticular lateral epicondyle (Fig. 23.2) [3].
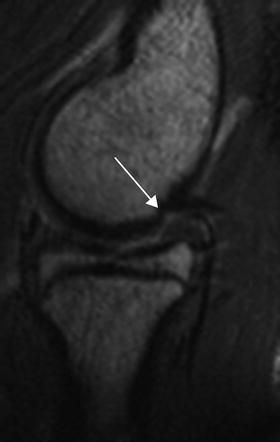

Fig. 23.2
Pseudodefect of posterior inferior capitellum (reprinted from Jans LBO et al. MR imaging findings and MR criteria for instability in osteochondritis dissecans of the elbow in children. Eur J Radiol. 2012;81:1306–10, with permission from Elsevier)
Diagnosis (Clinical/Imaging)
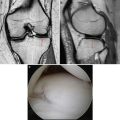
Although OCD lesions can be seen on plain radiography, they can be missed in up to 50 % of cases. To help increase the ability to detect OCD on plain radiographs, a view with the distal humerus in 45° of flexion and 30° of external rotation has been described [10, 16, 17]. Early lesions on radiographs can appear as a slight flattening and sclerosis and progress to radiolucent bone overlying the sclerotic area at approximately 4 months [18]. See Figs. 23.3a–d and 23.4a, b.
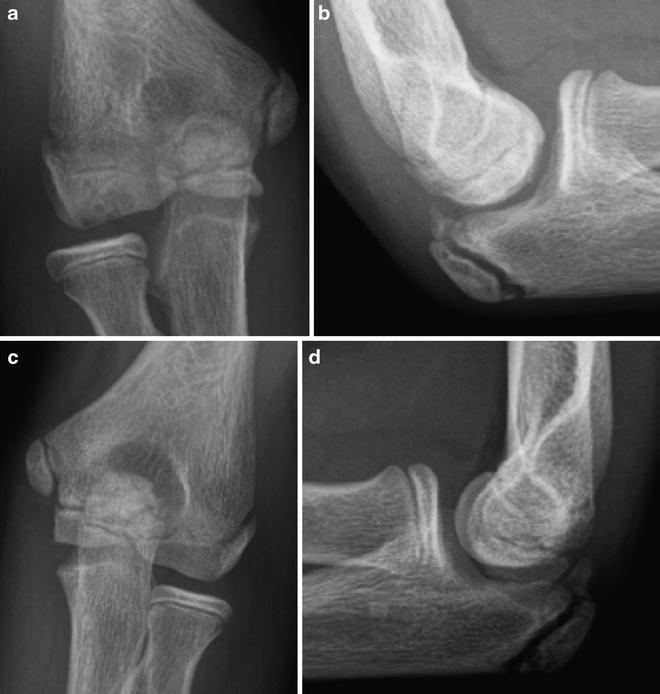
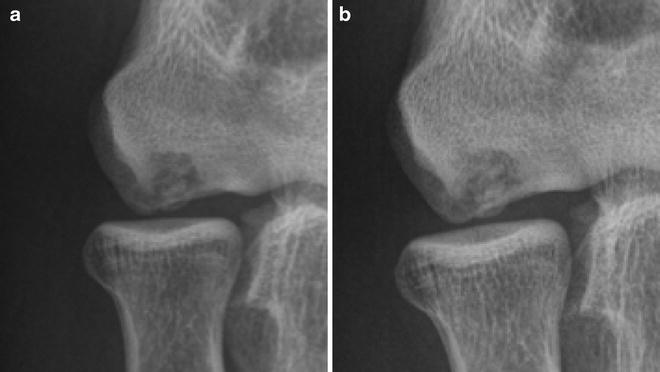

Fig. 23.3
(a–d) A 14-year-old baseball player with 2-month history of right elbow pain demonstrating cystic appearance and limited area of sclerosis. There is flattening of the capitellum most noticeable on the lateral view (a, b). Comparison views of the left elbow (c, d)

Fig. 23.4
(a, b) A 14-year-old quarterback treated nonoperatively. Initial presentation radiograph of lesion in capitellum (a). Radiograph 4 months (b) after initiation of treatment demonstrating progressive bony filling of capitellar defect
New bone continues to form over the flattened area and may have the appearance of a nondisplaced fragment which will begin to form and unite with the underlying bone at approximately 4 months for patients with an open growth plate and may take up to 8 months for those with a closed growth plate. This can continue until past a year. Complete healing can be expected at skeletal maturity [19, 20].
Takahara et al. have described a radiographic classification. Grade I lesions (Fig. 23.5a, b) show a translucent cystic shadow in the lateral or middle capitellum with or without localized flattening. Grade II lesions have a clear zone or split line between the lesion and the adjacent subchondral bone which represents a nondisplaced fragment (Fig. 23.6). Grade III lesions are a displaced or detached fragment, often associated with the presence of loose bodies (Fig. 23.7) [21]. Two subgroups were added that divided grade II into IIA, which is a fragment not demarcated by sclerosis, and IIB which have a sclerotic rim [22]. These classifications have been used in studies to determine lesion stability as described below.
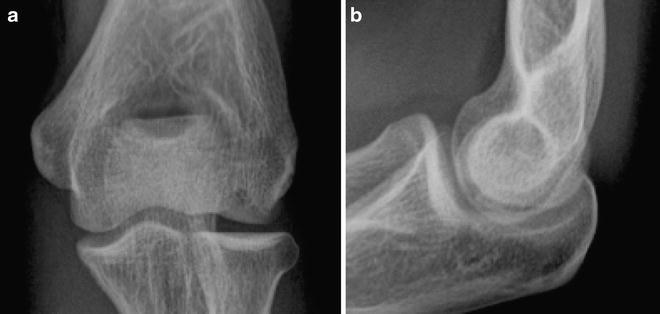
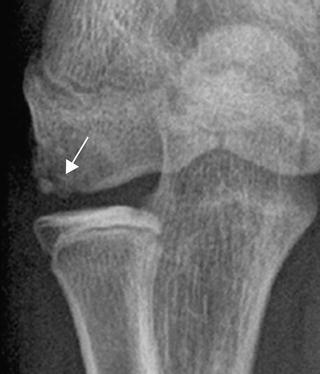

Fig. 23.5
(a, b) Grade I lesion with translucent cystic appearance on AP view (a) and flattening of capitellum noted on the lateral view (b)

Fig. 23.6
Grade II lesion with cystic lesion of capitellum and linear lucency dividing the bone and nondisplaced fragment along the distal-lateral capitellum

Fig. 23.7
Grade III lesion with loose fragments visible in the anterior compartment
MRI can also provide information regarding the size, location, and stability of the lesion. MRI of the elbow is optimally performed with the arm at the patient’s side, supinated, and in full extension. This position maximizes the patient’s comfort and image quality. Early lesions may demonstrate a low signal on T1 with more subtle abnormalities seen on T2 especially at the periphery [18]. See Figs. 23.8a, b and 23.9a, b.
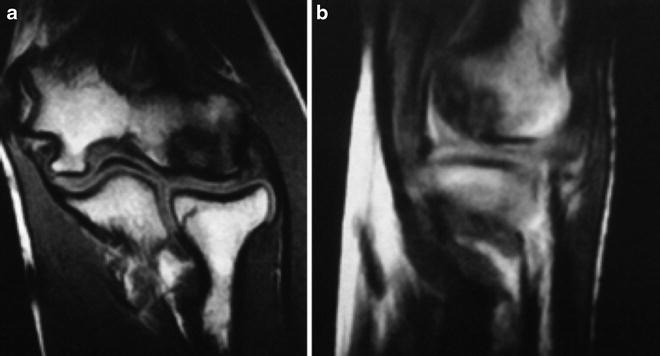
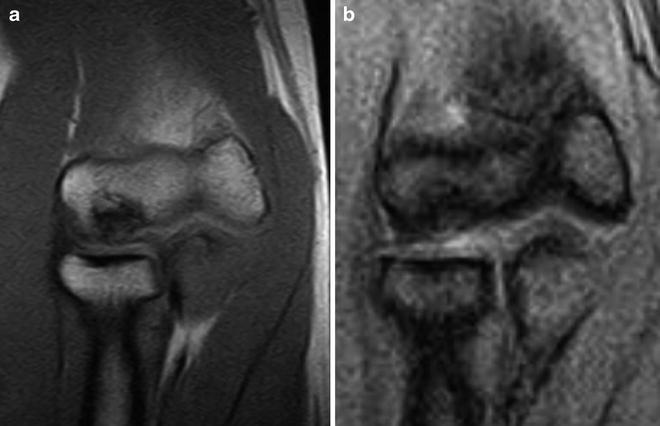

Fig. 23.8
Coronal (a) and sagittal (b) T1 MRI demonstrating low signal intensity in a stable capitellar lesion

Fig. 23.9
Another example of a T1 MRI of a stable lesion (a) and the corresponding T2 (b). The periphery is indistinguishable from the surrounding marrow
Fast spin echo sequences can be used to emphasize cartilage, and fat suppression sequences will better show subchondral edema and small fluid/cystic collections [23]. T1 sequences appear similar when comparing stable and unstable lesions with variable low, intermediate, and heterogeneous low and intermediate signal intensity (Fig. 23.10a–c). However, on T2, there can be a high signal along the periphery of unstable lesions likely representing the interposition of synovial fluid (Fig. 23.11) [18, 24].
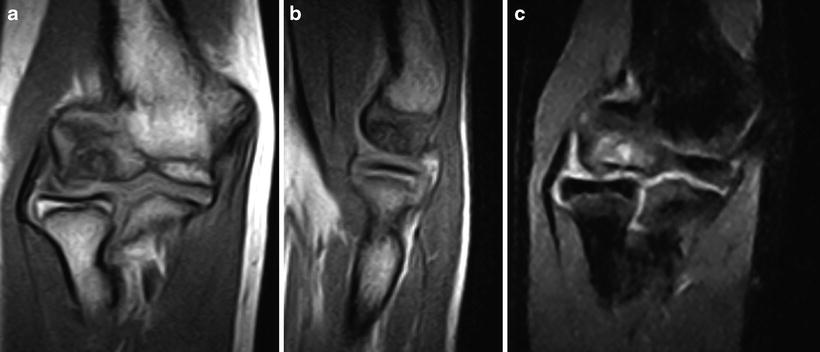
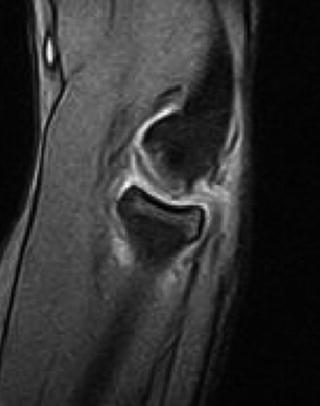

Fig. 23.10
T1 MRI with capitellar lesion with low peripheral signal and heterogeneous intermediate intensity in the central portion (a, b). T2 coronal image of the same lesion showing features consistent with an unstable lesion such as high-signal cystic and linear areas (c)

Fig. 23.11
T2 MRI demonstrating high-intensity signal at interface between the lesion and subchondral bone
Histologically, diffuse enhancement on MRI at the subchondral junction may also indicate granulation tissue interposition and an unstable fragment [25]. There can be a lack of signal along the periphery of stable lesions which makes the lesion difficult to distinguish from the adjacent marrow. In addition, MRI may reveal surface flaps, early delamination, displacement of the lesion, and presence of loose bodies. Underlying cysts may also be seen (Fig. 23.12) [21, 23, 24].
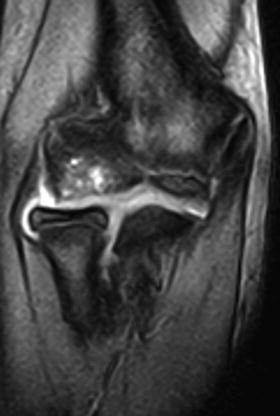

Fig. 23.12
Example of cystic area beneath lesion in the capitellum
Two systems by De Smet et al. [24, 26] and Dipaola et al. (Table 23.1) [27, 28] have utilized MRI findings to classify the stability of OCD lesions. However, both of these often cited studies surveyed lesions of the femoral condyles and talar dome. MRI signs as described by De Smet are summarized as follows:
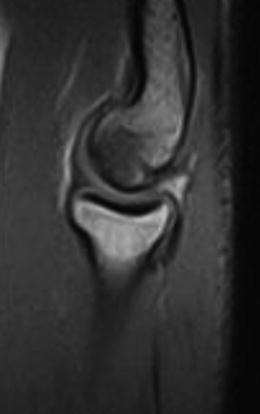
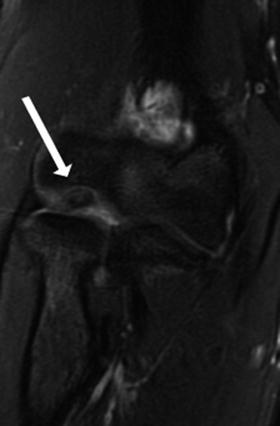
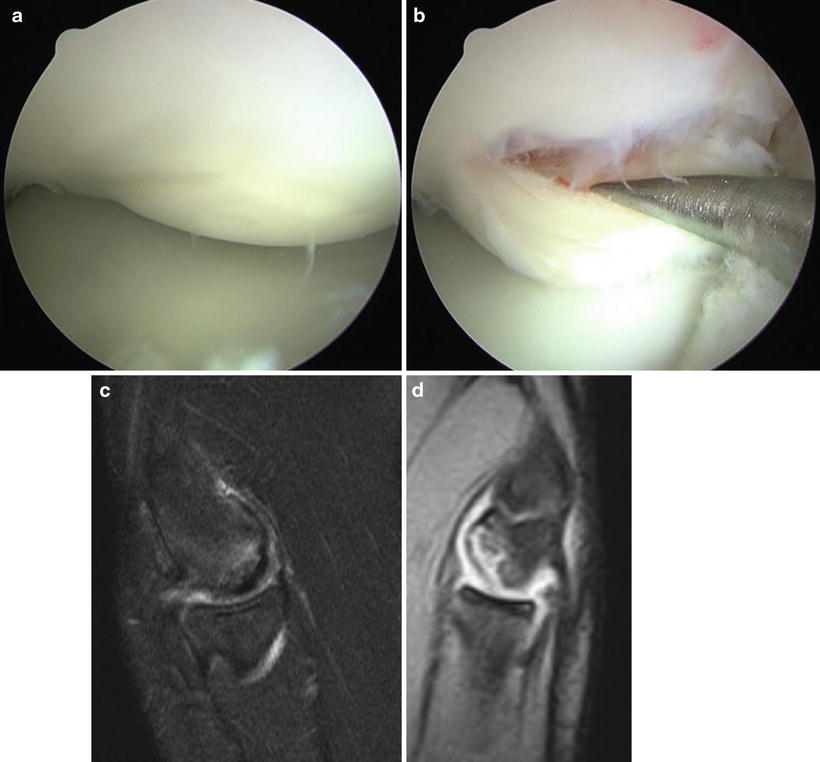
Table 23.1
OCD staging system
Stage | Arthroscopic findings | MRI |
|---|---|---|
I | Irregularity and softening of articular cartilage. No definable fragment | Thickening of articular cartilage and low-signal changes. See Fig. 23.13 |
II | Articular cartilage breached, definable fragment, not displaceable | Articular cartilage breached, low-signal rim behind fragment indicating fibrous attachment. See Fig. 23.14 |
III | Articular cartilage breached, definable fragment, displaceable but attached by some overlying articular cartilage | Articular cartilage breached, high-signal changes behind fragment indicating synovial fluid between fragment and underlying subchondral bone. See Fig. 23.15a–d |
IV | Loose body |
A thin, ill-defined or well-defined line of high signal intensity at the interface between the osteochondral lesion and underlying bone
A discrete, round area of homogeneous high signal intensity beneath the lesion such as a cyst
A focal defect in the articular surface of the lesion
A high-signal-intensity line penetrating the articular cartilage and subchondral bone into the lesion

Fig. 23.13
Example of Dipaola stage I demonstrating low-signal changes in the capitellum

Fig. 23.14
Example of Dipaola stage II lesion. White arrow pointing to low-signal rim suggesting fibrous union of fragment

Fig. 23.15
Intraoperative images of Dipaola stage III lesion still attached to some overlying cartilage but still displaceable (a, b). MRI examples demonstrating a (c) thin and ill-defined line and (d) a more well-defined line dividing the lesion and underlying subchondral bone
In their study, they reviewed MRI reports and intraoperative assessments in an average patient population age of 25.7 years. They were able to correctly identify 97 % of unstable lesions and 100 % of stable lesions using their classification system. The most frequent sign of instability was a high-signal line at the junction between the lesion and subchondral bone seen in 72 % of cases. This line was not seen in any of the patients with stable lesions. The presence of at least one sign indicates fragment instability [26].
Dipaola et al. performed a double-blinded, prospective study of 12 patients with an average age of 26.5 years using a 0.35T MRI. They devised their own system, and the arthroscopic stages are not those of the ICRS classification. This staging system originally used arthroscopic, MRI, and radiographic findings. However, more recent studies have only included the arthroscopic and MRI components [29]. MRI was able to correctly identify all stage I, III, and IV lesions. One patient with stage II lesion was incorrectly identified as a stage III lesion. Stages III and IV are considered unstable [27]. See Table 23.1 [27, 28].
The ICRS classification system has more recently been created for arthroscopic evaluation of OCD lesions which is summarized in Table 23.2 [30].
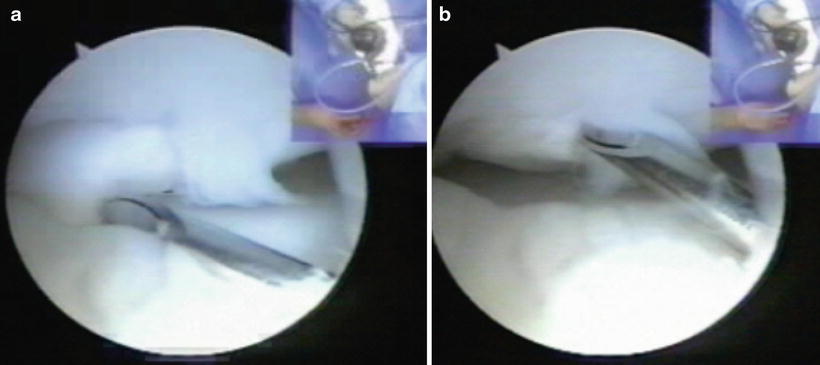
Table 23.2
International Cartilage Repair Society classification for OCD
ICRS OCD stage | Findings |
|---|---|
I | Stable in continuity, softened area covered by intact cartilage |
II | Partial discontinuity, stable on probing |
III | Complete discontinuity, “dead in situ,” not displaced |
IV | Displaced fragment, loose within the bed or empty defect. See Fig. 23.16a, b |
Subgroup B | >10 mm depth |

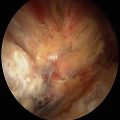
Fig. 23.16
Example of ICRS stage IV lesion. The spinal needle is manipulating a loose fragment within its bed in the capitellum (a, b)
Even though MRI has been the standard for evaluating articular cartilage, it can have limitations. Theodoropoulous et al. looked at 31 patients with an average age of 38.7 years (range 15–63) with elbow articular cartilage defects that did not include OCD lesions diagnosed during arthroscopy and compared the findings with those of the MRI. The accuracy was 45 %, 65 %, 20 %, and 30 % for the radius, capitellum, ulna, and trochlea, respectively. This study included ICRS grade II defects (not the ICRS OCD classification, lesion >50 % depth of cartilage) and used a 1.5T MRI and fast spin echo T2 sequences to highlight articular cartilage defects [31].
MRI also may have limitations predicting OCD stability especially in a younger, overhead athlete population. Using intraoperative assessment as the gold standard, Iwasaki et al. looked at 27 overhead athletes (average age of 14 years, range 11–30 years) and compared lesion stability between MRI using the classification systems of De Smet and Dipaola and the ICRS OCD classification system. The majority used a 1.5T MRI. For the De Smet system, sensitivity was 89 % and specificity was 44 % diagnosing fragment instability. The positive predictive value (PPV) and negative predictive value (NPV) were 76 % and 67 %, respectively. False negatives and false positives were 11 % and 56 %, respectively. For the Dipaola system, sensitivity was 83 % and specificity 44 %. PPV and NPV were 75 % and 57 %, respectively. False negative and false positives were 17 % and 56 %, respectively. Furthermore, all MRI predicted stable lesions but did not accurately predict the intraoperative/arthroscopic stage. It was able to predict 57 % of stage III and 64 % of stage IV unstable lesions. The authors stated that many patients with predicted stable lesions are unlikely to have a stable lesion once evaluated intraoperatively, especially in teenage patients who repetitively stress the radiocapitellar joint such as overhead athletes. Thus, these patients still have histopathological instability despite findings on MRI. MRI was better overall for predicting unstable lesions that were in fact unstable during arthroscopic evaluation [29].
Satake et al. [32] retrospectively reviewed 78 patients to determine signs of lesion instability. The average age between operatively and nonoperatively treated patients was 13.3 and 11.2 years. ICRS grading was used during intraoperative evaluation. Along with determining signs seen on MRI, the authors also used plain radiographs and CT scan. Their findings are summarized below.
Correspondence with intraoperative instability:
1.
Minami grade III on plain radiographs
2.
Closure of the epiphyseal line on the capitellum and lateral epicondyle
3.
Articular irregularity on MRI
4.
High signal intensity on T2 MRI at interface of lesion
5.
Displaced fragment on CT scan
No correspondence with intraoperative instability:
1.
Intralesional segmentation on CT
2.
Signal line through articular surface on MRI
3.
Articular defect (not irregularity) on MRI
Sensitivity >80 % to detect unstable lesion:
1.
Epiphyseal closure of capitellum on radiographs
2.
Segmentation on CT
Specificity >80 %:
1.
Minami grade III
2.
Epiphyseal closure of lateral epicondyle on radiograph
3.
Articular irregularity
4.
T2 high signal at interface
5.
Displaced fragment on CT (may miss cartilaginous-only lesion)
The authors concluded that radiographic grade III lesions, closure of the epiphyseal line of the capitellum and lateral epicondyle, MRI findings of irregular articular contours, and high signal intensity at the lesion interface as well as a displaced fragment on CT were indicative of fragment instability.
Jans et al. retrospectively reviewed 25 patients with an average age of 14 years and found that using instability criteria for the knee described above by De Smet et al. (high T2 rim signal, surrounding/sublesional cysts, high-T2-signal fracture line, and a fluid-filled osteochondral defect). When all four criteria were combined, MRI was 100 % sensitive for unstable OCD of the elbow. Enhancement of the OCD fragment post-gadolinium contrast suggests that the lesion is viable [14].
Based on the studies described above, MRI findings suggestive of lesion instability are summarized below [17, 24, 26, 29, 32]:
Get Clinical Tree app for offline access

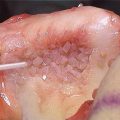
Articular cartilage breached and a thin, ill- or well-defined line of high signal intensity at the interface between the osteochondral lesion and underlying bone. See Fig. 23.17a–e.
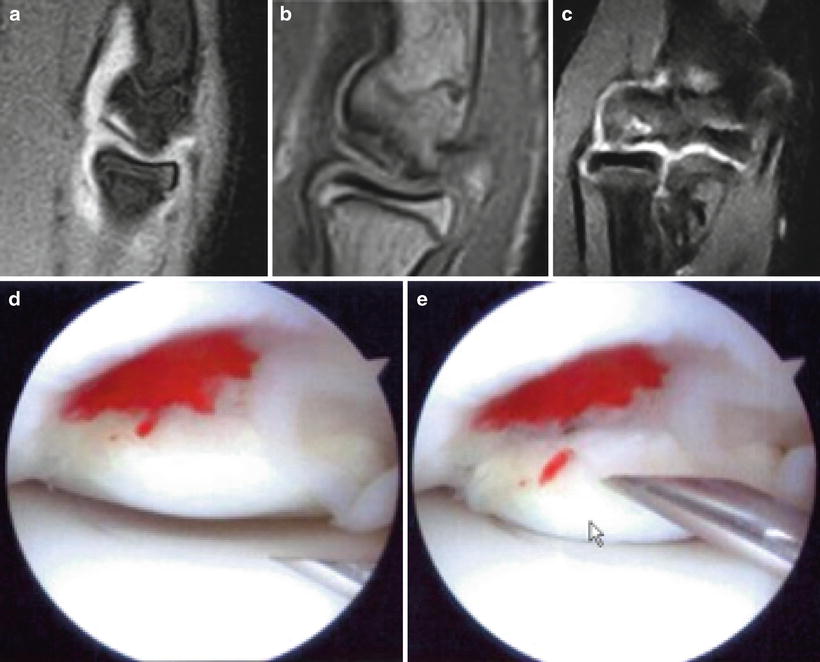
Fig. 23.17
MRI images illustrating a thin line demarcating the lesion from subchondral bone (a–c). Arthroscopic images demonstrating a similar lesion (d, e)
A discrete, round area of homogeneous high signal intensity beneath the lesion (cystic structure). See Fig. 23.18.
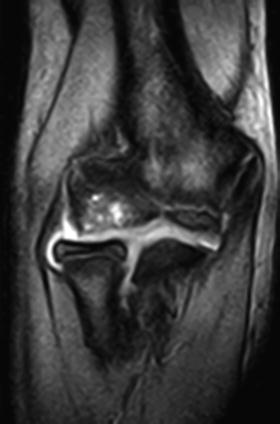
Fig. 23.18
Capitellar lesion with multiple cystic areas surrounding the lesion
A focal defect in the articular surface of the lesion, multiple articular breaks, or high-signal-intensity line penetrating the articular cartilage and subchondral bone into the lesion. This is based on the criteria by De Smet et al. [24, 26]. This finding is not suggestive of lesion instability according to Satake et al. [32].
Displaced or loose fragment. See Fig. 23.19a–e.
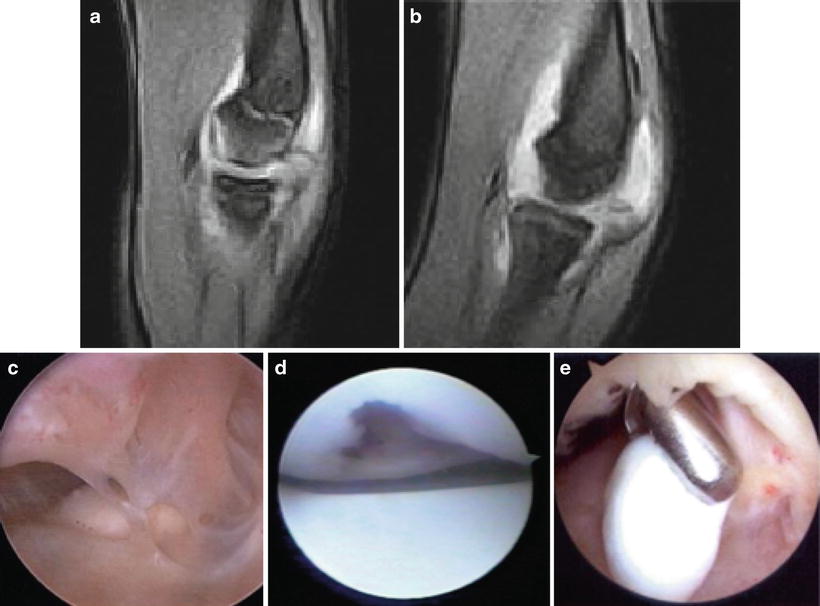
Fig. 23.19
Sagittal T2 MRI with high-signal line beneath the lesion in the capitellum (a) and repeat MRI of the same patient approximately 6 months later with empty bed and loose body in posterior compartment (b). Arthroscopic image of adhesions and loose body (c). Other image examples of empty bed and loose-body removal (d, e)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree