Abstract
Omphalocele is a result of a full-thickness midline defect of variable size that allows herniation of fetal bowel or liver or both into an amnioperitoneal sac at the level of the umbilicus. The reported incidence of omphalocele, when stillbirths and abortions are considered, ranges from 2.5 : 10,000 to 4 : 10,000. Omphalocele is more common in women at the extremes of reproductive age. Approximately 55% to 80% of cases are associated with additional fetal anomalies, and 13% to 43% of cases of omphalocele are associated with fetal karyotype abnormalities. Omphalocele results when the physiologic herniation of the embryonic gut into the umbilical cord fails to return to the abdominal cavity. Approximately 70% of omphaloceles are associated with elevations in maternal serum alpha-fetoprotein (MSAFP). The classic appearance of omphalocele on US is an anterior abdominal mass at the site of cord insertion with a covering membrane. The umbilical cord is inserted into the apex of the sac, and the umbilical vein may be seen traversing through the center. Smaller defects, not containing fetal liver, have a higher incidence of karyotype abnormalities compared with larger, liver-containing defects. Differential diagnosis includes gastroschisis, limb–body wall complex, bladder exstrophy, cloacal exstrophy, ectopia cordis, pentalogy of Cantrell, umbilical cord cysts, and urachal abnormalities. Delivery can be vaginal and should occur at a center able to care for the neonate. Postnatal therapy includes primary closure, use of a Silastic spring-loaded silo, or staged closure. Overall survival rate directly correlates with the presence of additional structural and chromosomal abnormalities.
Keywords
omphalocele, ventral wall defect, ultrasound findings, prenatal diagnosis, obstetrical management
Introduction
Omphalocele is classified as a ventral wall defect in which there is midline herniation of abdominal viscera into the base of the umbilical cord. Before the 1950s, gastroschisis was considered a variant of omphalocele. It is now known that each entity has a separate etiology, unique risk factors, and distinct outcomes. As a result of widespread use of prenatal screening with maternal serum alpha-fetoprotein (MSAFP) and second-trimester ultrasound (US) examination, omphalocele is commonly diagnosed during pregnancy. Fetuses with a diagnosis of omphalocele are at a significantly increased risk for having additional structural or chromosomal abnormalities, or associated conditions such as Beckwith-Wiedemann syndrome ( Chapter 109 ). Because of these associations, there is an elevated risk of subsequent fetal loss in affected pregnancies. Early and accurate identification of omphalocele is the critical first step in providing appropriate prenatal counseling for families affected by this congenital anomaly. Detection of additional abnormalities, appropriate genetic counseling, and involvement of appropriate pediatric specialties are critical components of the optimal antepartum management of omphalocele.
Disease
Definition
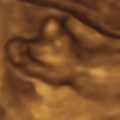
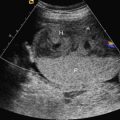
Omphalocele, also known as exomphalos, is a midline abdominal wall defect of variable size in which bowel or liver or both herniate into the base of the umbilical cord. Although possible, it is less common for other abdominopelvic organs to be present in an omphalocele ( Figs. 21.1 , 21.2 ). The defect is characterized by the absence of fascia, abdominal muscles, and skin. The herniated abdominal contents are covered by a membrane consisting of peritoneum on the inner surface, Wharton jelly in the center, and amnion on the outer surface. The umbilical vessels insert into the membrane, distal from the body wall, and the umbilical vein can be seen coursing through the omphalocele sac ( Figs. 21.3 , 21.4 ).




Identification of the covering membrane and abnormal location of the umbilical cord insertion are two of the key anatomic features of omphalocele. Correct detection allows for differentiation from gastroschisis. Although rare, as documented by case reports, it is possible for the amnioperitoneal covering to rupture in utero. If rupture occurs, differentiation from gastroschisis becomes difficult, especially in the setting of advanced gestational age. The presence of extracorporeal liver may help identify a ruptured omphalocele accurately because liver is rarely herniated in isolated gastroschisis.
Prevalence and Epidemiology
The reported incidence of omphalocele ranges from 1.0 : 10,000 to 2.5 : 10,000 live births. The incidence ranges from 2.5 : 10,000 to 4.0 : 10,000 when stillbirths and abortions are considered. In contrast to gastroschisis, the incidence of omphalocele does not seem to be increasing. A retrospective review of cases of prenatally diagnosed omphalocele in the state of Western Australia from 1998–2007 identified 107 cases of omphalocele in live births, stillbirths, and terminations of pregnancy from 256,500 registered births. The average rate of omphalocele was 4 : 10,000 births (range 2.3–7.8 : 10,000 births). In the 1995 EUROCAT survey of 3 million births from 1980 to 1990, 732 cases of omphalocele were registered. The average total prevalence rate was 2.53 : 10,000 births (range 1.11–4.79 : 10,000 births). Significant heterogeneity was observed in rates of omphalocele in Europe. One possible explanation for the observed regional differences may be the underregistration of some types of births, notably terminations. The 2001 survey from the EUROSCAN study group found a prevalence of 1.98 : 10,000 births. There was a considerable amount of heterogeneity between the rates in different geographic regions, ranging from 0.15 : 10,000 to 6.09 : 10,000 births. The National Birth Defects Prevention Network in the United States examined 2308 cases of omphalocele over 11 years (1995–2005) and found that the prevalence of omphalocele remained constant over this time span.
The association between omphalocele and chromosomal abnormalities is well characterized, with published rates of chromosomal abnormalities in omphalocele ranging from 13% to 42%. The wide range may be explained by the high rate of intrauterine lethality for many chromosomal abnormalities, resulting in the observed decrease in prevalence with advancing gestational age. This effect is described in a US screening study of 15,726 women, which estimated the prevalence of chromosomal defects in 153 fetuses with omphalocele to decrease from 39.4% at 12 weeks’ gestation to 27.5% at 20 weeks’ gestation and 14.4% at live birth. The most commonly associated aneuploidy is trisomy 18. Approximately 9% to 15% of fetuses with omphalocele have a recognized syndrome, and many fetuses with omphalocele have additional structural abnormalities, with incidence ranging from 54% to 78% ( Table 21.1 ).
| Study | Year Published | Associated Anomalies, % ( n / N ) | Karyotype Abnormality, % ( n / N ) (% Trisomy 18) | Recognized Syndrome, % ( n / N ) |
|---|---|---|---|---|
| EUROCAT | 1995 | 54 (397/732) | 12 (94/732) (64) | 9 (66/732) |
| EUROSCAN | 2001 | 56 (77/137) | 25 (34/137) (62) | 10 (14/137) |
| Western Australia | 2009 | 74 (66/89) | 43 (40/89) (78) | 5 (4/89) |
| US National Birth Defects Prevention Network | 2015 | 62 (1421/2308) | 17 (385/2308) (50) |
There are minimal racial and ethnic differences in the incidence of other anomalies with omphalocele. Of black infants with omphalocele, 38% had associated anomalies, whereas 41.7% of whites and 40.6% of Hispanics had associated anomalies. Survival was better among black infants (adjusted hazard ratio 0.52, 95% confidence interval [CI] 0.37–0.74).
Women at the extremes of reproductive age (younger than 20 and older than 35), multiple gestation, and male sex are associated with increased risk of omphalocele. Maternal obesity is also a risk factor for fetal omphalocele. In a population-based case-control study from the Atlanta Birth Defects Risk Factor Surveillance Study, obese women (body mass index ≥30) were more likely than women of average weight to have an infant with omphalocele (odds ratio 3.3, 95% CI 1.0–10.3). Additional maternal risk factors for the development of omphalocele identified in the National Birth Defects Prevention Study included alcohol consumption (adjusted odds ratio 1.53, 95% CI 1.04–2.25) and heavy smoking (adjusted odds ratio 4.26, 95% CI 1.58–11.52).
Etiology and Pathophysiology
Omphalocele is considered a sporadically occurring defect. However, familial recurrence of these birth defects is more common compared with gastroschisis. Ventral wall defects are a result of disturbances during embryonic development. The anterior abdominal or ventral wall develops between gestational weeks 5 and 12. This process involves the folding of the embryonic disk in a cranial-caudad direction and lateral direction. The resulting umbilical ring contains the allantois, umbilical vessels, extraembryonic coelom, vitelline duct, and associated vitelline vessels.
As the abdomen develops, there are different rates of development of the enclosed organs and the abdominal cavity itself. By gestational week 8, the embryonic liver begins to expand rapidly, and the elongating fetal midgut herniates into the extraembryonic coelom in the umbilical cord owing to the lack of sufficient intraabdominal space. This phenomenon is termed physiologic midgut herniation . The allantois, vitelline duct, and accompanying vessels are obliterated as they are incorporated into the umbilical cord. By gestational week 12, the midgut rotates, the extraembryonic coelom becomes progressively obliterated, and the embryonic bowel is directed back into the abdominal cavity. At the same time, the heart and the pericardium, which lie on the ventral surface of the embryo, are incorporated into the chest by the lateral and cephalic folds. Missteps during this critical period of embryonic development may result in anterior abdominal wall defects of varying severity or cardiac defects, or both.
In general, the earlier the embryologic abnormality occurs, the more complex the structural abnormality will be. Omphalocele is proposed to be a result of one or both of the following developmental errors: (1) failure of closure of the extraembryonic coelom with persistent herniation of abdominal contents or (2) defective fusion of the embryonic folds in the midline. The latter mechanism is also thought to be responsible for other ventral wall defects, including cloacal exstrophy and pentalogy of Cantrell.
As previously mentioned, omphalocele is strongly associated with other structural and chromosomal abnormalities. However, fetuses with omphalocele constitute a heterogeneous group. It is possible to have a karyotypically normal fetus with isolated omphalocele grow and develop into a healthy child with no additional complications. In many cases, omphalocele may be only a marker of a more serious developmental abnormality that can involve a chromosome aberration, syndrome, or additional anomalies.
The most commonly reported associated congenital abnormality is congenital heart disease, which accounts for 35% to 63% of identified malformations. Specific defects include ventricular septal defect, atrial septal defect, tetralogy of Fallot, dextrocardia, and ectopia cordis. Levorotation of the cardiac axis is commonly seen in omphalocele and its presence is not always correlated with congenital heart disease ( ![]() ). A case series in 2006 found 59% of fetuses with omphalocele to have an abnormal cardiac axis, but only 35% had a congenital heart defect ( Fig. 21.5 ). Isolated cardiac levorotation is likely because of the downward and outward displacement of abdominal contents. Additional associated abnormalities include central nervous system defects, craniofacial defects, esophageal atresia, diaphragmatic defect, limb abnormalities, genitourinary anomalies, and gastrointestinal defects ( Table 21.2 ).
). A case series in 2006 found 59% of fetuses with omphalocele to have an abnormal cardiac axis, but only 35% had a congenital heart defect ( Fig. 21.5 ). Isolated cardiac levorotation is likely because of the downward and outward displacement of abdominal contents. Additional associated abnormalities include central nervous system defects, craniofacial defects, esophageal atresia, diaphragmatic defect, limb abnormalities, genitourinary anomalies, and gastrointestinal defects ( Table 21.2 ).

| MUSCULOSKELETAL | Clubfoot, polydactyly, limb deficiency, vertebral anomalies, rib anomalies |
| URINARY | Renal agenesis, polycystic kidney, hydronephrosis, ureteral anomalies, vesicoureteral reflux, duplication of kidney and renal pelvis, agenesis of urethra or bladder, bladder exstrophy |
| CARDIOVASCULAR | Ventricular septal defect, atrial septal defect, tetralogy of Fallot, patent ductus arteriosus, tricuspid valve abnormality, ectopia cordis |
| GASTROINTESTINAL | Anal atresia, ileal atresia, malrotation |
| CENTRAL NERVOUS | Spina bifida, anencephaly, hydrocephaly |
| DIAPHRAGMATIC | Hernia |
| HEAD AND FACE | Cleft lip/palate, facial anomalies |
| GENITAL | Absence of uterus, hypospadias, ambiguous genitalia |
| RESPIRATORY | Laryngeal anomalies, pulmonary agenesis, cystic lung |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree