, Valery Kornienko2 and Igor Pronin2
(1)
N.N. Blockhin Russian Cancer Research Center, Moscow, Russia
(2)
N.N. Burdenko National Scientific and Practical Center for Neurosurgery, Moscow, Russia
According to Hardy and Harvey (1989), the analysis of a small group of ovarian carcinoma cases detected metastases in the brain in 11.6% of cases. Bruzzone et al. (1993) identified metastatic brain lesions only in 2.2% of cases among 413 patients with ovarian carcinoma treated with platinum-based therapy from 1981 to 1989. Mayer et al. (1978) found brain metastases in 0.9% of 567 autopsies of patients who died from ovarian cancer. In another large study by Pectasides et al. (2006), during 22 years in the analysis group (n = 1450 patients with ovarian cancer), only 17 (1.17%) developed brain metastases.
In our material, the proportion of ovarian cancer metastases in the brain was 1.8%, and there was a tendency to its increase in recent years.
Most ovarian cancer metastases in the brain manifest as single lesions. Lesions have a solid-cystic structure with a predominance of the cystic component. Almost always an area with a perifocal edema is visualized. The accumulation of the contrast agent by the solid part of the metastasis is expressed; the solid component of metastases is well visualized on MRI. When lesions are multiple, metastases may have different forms and can be detected by the presence of necrosis. Hemorrhages in the structure of ovarian cancer metastases are rare.
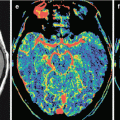
On SWI (SWAN) MRI, ovarian cancer metastases have a small number of hypointense inclusions. On the spectrum, there is a Lip-Lac complex typical of all metastases. On ASL images, the blood flow activity is directly proportional to the volume of the solid tumor, while the tumor is not visualized when its cystic component is prevalent.
The solid part of ovarian cancer metastases showed the lowest CBV values (5.34 ± 1.45 ml/100 g). Metastatic ovarian cancer is also characterized by the lowest CBF (49.51 ± 19.6 ml/100 g/min) values and mean MTT values (10.58 ± 6.27 s). Based on CT perfusion findings that identified perfusion indices specific to this type of metastases, the primary source of metastases can be assumed.
Despite the complexity of the diagnosis related to the close location of the pelvic organs and the presence of concomitant non-oncological diseases (tubo-ovarian abscesses, cysts, endometriosis), PET allows quite accurately the identification of the initial site of the primary tumor and its spread.
Grigsby et al. (2001), based on the data of the comparative analysis of the results from a survey in women with cervical and ovarian carcinoma (101 cases) by CT and PET, found that even single-organ PET was three times more effective than CT in detecting metastatic lesions of the pelvic lymph nodes (67 and 20%, respectively). Also, PET additionally identified metastatic involvement of supraclavicular lymph nodes in 8% of patients in this series.
The use of PET with 18F-FDG for the primary diagnosis of metastatic brain lesions in ovarian cancer is impractical, both due to the predominantly cystic structure of such metastases and due to relatively low RP accumulation by the solid part of the tumor that merges with the image of the intact brain substance, since it physiologically accumulates the radiopharmaceutical.
23.1 Clinical Cases
See Figs. 23.1, 23.2, 23.3, 23.4, 23.5, 23.6, 23.7, 23.8, and 23.9.
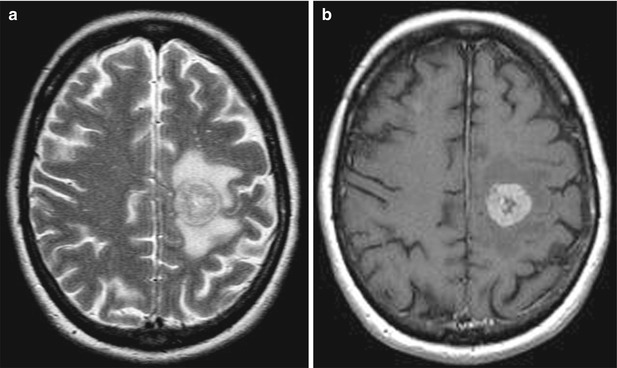

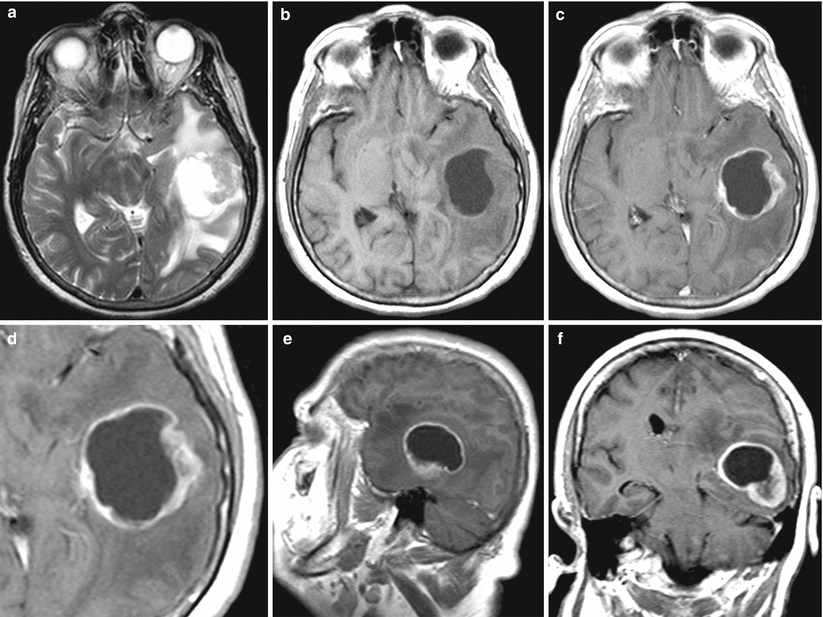
Fig. 23.1
An ovarian cancer metastasis in the brain. On MRI in the left frontal region, there is a rounded tumor lesion with an increased signal and signs of a perifocal edema on T2 images (a), intensely accumulating the contrast agent (b)—ring enhancement