© Springer International Publishing AG 2017
Pietro Ruggieri, Andrea Angelini, Daniel Vanel and Piero Picci (eds.)Tumors of the Sacrum10.1007/978-3-319-51202-0_2626. Palliative Treatments for the Sacrum
Andreas F. Mavrogenis1 , Georgios N. Panagopoulos1, Andrea Angelini2 , Giuseppe Rossi3 , Alberto Bazzocchi3 and Pietro Ruggieri2
(1)
First Department of Orthopaedics, National and Kapodistrian University of Athens, Athens, Greece
(2)
Department of Orthopedics and Orthopedic Oncology, University of Padova, Via N. Giustiniani, 3, Padova, 35128, Italy
(3)
Diagnostic and Interventional Radiology, Istituto Ortopedico Rizzoli, Bologna, Italy
26.1 Introduction
A wide array of tumors can occur in the sacrum. Benign tumors include giant cell tumors (60%), aneurysmal bone cysts (4%), and osteoblastomas [1]. Malignant tumors of the sacrum include chordomas (50%), hematopoietic malignancies (lymphoma and multiple myeloma, 18%), Ewing’s sarcoma in children (8%), chondrosarcoma, and osteosarcoma [2]. However, the most common malignancy to occur in the sacrum is represented by metastatic tumors [3]. The most frequently implicated primary cancers include breast, lung, prostate, renal and thyroid, lymphoma, melanoma, and tumors of unknown origin follow, but are less common primary locations [4–9]. Spread is mainly by hematogenous dissemination, although direct extension in case of recurrent rectal tumors and drop metastases of intradural tumors have been also described [8, 9]. Sacral metastases are usually diagnosed in advanced stages, when they have already extended beyond bony margins and around sacral nerves and other surrounding structures [3, 10]. They generally grow insidiously causing ambiguous symptoms in the early stages, thus frequently resulting in a delayed diagnosis.
Patients may live with cancer for many years [11]. Their most common symptom is pain, which may be local, mechanical, or radicular in nature. This is typically followed by a progressive neurological deficit, eventually leading to bladder, bowel, and/or sexual dysfunction [12–14]. The treatment of sacral metastatic lesions is usually palliative, aiming primarily at pain control and preservation of neurological function [8, 9, 15–30]. The goal of palliative therapy is the prompt and cost-effective relief of symptoms, thus improving function and quality for the remainder of the patient’s life, with as little treatment-related morbidity as possible. Palliative care is not restricted to terminally ill patients and is an important aspect of overall cancer management.
There is a paucity of studies dealing with the management of sacral metastases, since most papers refer to primary sacral tumors [8, 9, 15–30]. There is also no consensus or precise strategy algorithm as to which method is the most appropriate, making management highly individualized on a patient-to-patient basis, depending to an extent on institutional preferences. The modalities commonly employed include radiotherapy and stereotactic surgery, surgical debulking, sacroplasty, embolization, various ablation techniques, and electrochemotherapy.
26.2 Radiotherapy
Radiotherapy is frequently chosen as a first-line initial therapy for radiosensitive sacral metastases, in patients without evidence of spinal instability or acute neurological deterioration, where pain reduction and neurological improvement are attainable [31, 32]. In fact, palliative radiotherapy as a first-line intervention for spinal metastasis has been the mainstay of management since the late 1960s, after several authors compared radiotherapy with laminectomy, finding no significant difference in patient outcome [33–35]. It must be taken into account that radiosensitivity varies among primary cancer types. In general, prostate and lymphoid tumors are radiosensitive; breast cancer is 70% sensitive and 30% resistant, whereas gastrointestinal tumors, renal cell carcinomas, and melanomas are radioresistant [3]. Radiation doses of 30–50 Gy are indicated for palliation with minimal associated risk. Dose fractioning is controversial. Cummings et al. [36] reported no differences in survival, duration of symptomatic response, and progression-free survival in a heterogeneous group of patients who underwent 50–60 Gy radiotherapy compared with conventional fractions to less than 50 Gy or by a hyperfractionated regimen of 44 Gy delivered in 1-Gy fractions, four times daily for 14 days. To achieve higher doses, specialists at the Harvard Cyclotron Laboratory used a combined treatment of X-ray and proton-beam radiation therapy [37].
Conventional radiotherapy for spinal metastasis is delivered through simple portals to doses of 30–40 Gy in 2- to 3-Gy fractions. For isolated sacral lesions, an opposed anterior posterior field is typically used, encompassing one or two vertebral segments above and below the lesion. Alternatively, a three-field arrangement with two lateral portals and a single posterior field is used to spare anterior pelvic structures. A single posterior field arrangement is generally avoided for sacral lesions, particularly if lower energy beams are used, because the treatment depth will vary along the sacral hollow [38].
26.3 Stereotactic Radiosurgery
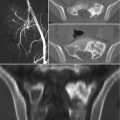
Stereotactic radiosurgery has been initially established as a safe and effective treatment modality for intracranial lesions. Precise tumor localization was achieved by fixation of stereotactic frames to the patient’s skull [1]. Spinal stereotactic radiosurgery (SRS) is an emerging form of radiotherapy, which allows more precise radiation delivery and high-dose hypofractionation. SRS makes it possible to administer a tumoricidal radiation dose even for radioresistant tumors, with minimal exposure of the surrounding normal tissues. Current commercial spinal SRS systems include the CyberKnife® (Accuray Incorporated, Sunnyvale, California, USA) and Novalis® (BrainLAB, Heimstetten, Germany) [3]. These systems consist of a lightweight linear accelerator mounted on a robotic arm, also incorporating an X-ray imaging feedback system. Spatial accuracy can be achieved in a frameless manner, with real-time imaging tracking. The patients are fitted with a custom body mold for immobilization, and fiducial markers (self-retaining tacks or self-tapping screws) are implanted via stab incisions in the sacrum in an ambulatory setting [38]. Unlike conventional radiation therapy in which a full dose is delivered to both the vertebral body and the spinal cord or cauda equina, the CyberKnife® can deliver a high-dose single fraction to the target tissue while sparing most of the adjacent neural elements, thus significantly reducing the possibility of radiation-induced myelopathy or injury to the nerve roots. This is the main advantage of stereotactic radiosurgery for treatment of many spinal and sacral tumors [39]. Recently, in an attempt to obviate the need for invasive fiducial marker insertion, a new tracking algorithm for fiducial-free tracking of spine and sacral lesions has been introduced for the CyberKnife® system [40].
26.4 Surgery
Metastatic disease to the sacrum is mostly treated nonoperatively with radiation, chemotherapy, embolization, or other palliative modalities. However, there are circumstances in which these therapies fail and surgical intervention is warranted. Whether surgery can be beneficial for these patients is not clearly defined [9]. Care must be taken in the decision making process, as sacral resections are challenging operations with a high incidence of potential complications, in a patient with an already limited life expectancy and compromised general health status [41]. On the other hand, resective surgery in carefully selected patients with sacral metastases may result in a palliative benefit. If pain derives from overt mechanical instability, as a result of structural failure of the lumbosacropelvic junction, it is unlikely for nonsurgical measures to have substantial benefit [9]. In such cases, stabilization with a modified Galveston technique can offer symptom control and improvement of ambulatory function [42].
Primary tumor type is also a variable that must be taken into account. Breast and prostate cancer are generally quite responsive to nonsurgical therapies, making surgery a rarely necessary therapeutic option, whereas lung, gastrointestinal, renal cell cancer, and melanoma tend to respond poorly to medical treatment [9]. Histology and overall tumor burden should be considered as well. For example, removal of secondary lesions of renal cell carcinoma tends to be associated with an increase in overall survival. A minimal disease burden (single-site metastasis) may signify that there is a greater change for surgery to achieve definitive local control [9].
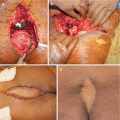
Once a decision for surgery is undertaken, its extent should take into account tumor location in the sacrum. The majority of sacral metastases involve the S1–S3, making en bloc resection too morbid a procedure to undertake for palliation. In such cases, aggressive intralesional resection is preferred. If the distal sacrum is involved, en bloc resection with preservation of the S1–S3 nerve roots may be feasible [15, 43, 44]. Wound healing is another potential issue of surgery. As soft tissue coverage in this territory may be challenging, plastic surgery feedback is frequently necessary [45].
26.5 Sacroplasty
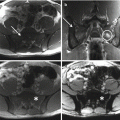
Sacroplasty is gaining favor in cases of metastatic disease without instability or neurologic compromise and represents a minimally invasive alternative to open procedures [46]. Mostly described in the degenerative/osteoporotic literature, sacroplasty has also been examined as a palliative option for metastasis-related insufficiency fractures [47–50]. The technique of sacroplasty is similar to vertebroplasty. Preoperative evaluation consists in obtaining a CT and/or MR imaging scan. The procedure may be done under local or general anesthesia. Image guidance is usually achieved by single-plain fluoroscopy. However, as exclusive fluoroscopy guidance might cause difficulty in visualizing the sacral foramina, many authors advocate for the use of a CT-guided approach [25]. This better defines spatial relationships within the treatment field and aids in preventing iatrogenic injury. A combination of the CT and fluoroscopic guidance may be the best alternative at present, allowing for both precise needle placement and real-time visualization of cement delivery [51]. Needle entry points are dictated by the fracture plane and conformation; cement is then injected under direct fluoroscopic vision to ensure maximal bony penetration and to prevent extravasation [49]. Sacral cement augmentation is not without risk. Potential complications of sacroplasty include hemorrhage, infection, durotomy with cerebrospinal fluid leak, direct injury of nerve roots or the lumbosacral plexus, ectopic cement injection (into the sacroiliac joint), migration, and embolization. Although cement migration is generally inconsequential, it might occasionally cause nerve root compression and radiculopathy, requiring decompression [49].
26.6 Selective Arterial Embolization
Embolization of sacral tumors is a useful, minimally invasive, palliative adjuvant therapy that may also aid in surgical management. Preoperative embolization of hypervascular metastatic lesions reduces intraoperative blood loss and improves the surgeon’s ability to subsequently perform surgical resection. Studies have shown that embolization may cause tumor growth arrest, pain alleviation, and shorter hospital stay [52–54]. The timing of preoperative embolization is also important. Typically, best results are achieved when surgery is performed within 24–48 h after embolization [55, 56]. Serial embolization can also be performed, if there is persistent pain and/or evidence of progressive disease on imaging. This has been shown to lead to tumor devascularization, tumor size reduction, calcification of margins, and consistent pain relief [57–59]. Serial embolization is typically performed in 4–6-week intervals until symptomatic improvement occurs or the tumor’s vascularity disappears. Unless there is a clear indication for general anesthesia, most procedures are performed under light conscious sedation. Arterial approach is determined based on lesion location and most often is the common femoral artery using a Seldinger technique. Commonly, a sheath is placed and standard 4 French or 5 French angiographic catheters are used for main arterial selection, and a 3 French microcatheter for subselectivity. A diagnostic angiogram is performed from a major vessel to delineate vascular supply to the tumor and to identify vessels of potential concern that may result in nontarget embolization. Typically, microspheres of 100–700 μm are used because of their ease of delivery, range of available sizes, and lower potential for recanalization. Sponge gel, once the embolic agent of choice, has now fallen in disuse, because of its temporary nature and propensity for recanalization. Coils are avoided as an embolic agent for tumor vessels, because re-treatment may be necessary and blocking access sites should be avoided. Completion of the procedure is determined by complete loss of tumor blush compared with the initial diagnostic angiogram [60]. Complication rate is generally low [61]. Risks of the procedure include nerve palsy, subcutaneous or muscle necrosis, post-embolization syndrome (fever, pain, malaise), ischemic pain (usually transitory), infection, and tumor bleeding [62].
26.7 Radiofrequency and Other Ablation Techniques
Over the past few decades, percutaneous ablation has emerged as an effective, minimally invasive, local treatment alternative to conventional methods, aiming to provide either palliation of painful bone lesions or local control of oligometastatic disease. Various image-guided ablation techniques have been applied to the treatment of bone metastases with varied levels of published evidence [63–67]. Thermal ablation methods include radiofrequency ablation (RFA), cryoablation, microwave ablation, laser ablation, and more recently MR imaging-guided focused ultrasound (MRgFUS) [63].
Radiofrequency ablation uses high-frequency alternating electrical current (200–1200 kHz) produced by the electrode. Many of the newer RFA devices utilize bipolar technology, eliminating the need for grounding pads, necessary in the past to avoid soft tissue burning. The alternating electrical current causes ionic agitation with subsequent frictional heat. The heat generated causes coagulative necrosis, with irreversible cell damage typically occurring between 60 and 100 °C [64, 65]. Some of the currently used RFA devices for bone include OsteoCool® RFA system (Baylis Medical, Burlington, Massachusetts), Dfine STAR® ablation system (Dfine, San Jose, California), and UniBlate® RFA system (Angiodynamics, Latham, NY). RFA cycles are typically 10–15 min long, depending on the size, shape, location, and intrinsic characteristics of the tumor. RFA also has a cauterizing effect that reduces the risk of bleeding. A limitation of RFA includes the inability to clearly visualize the treatment zone during ablation. The active tips of the RFA probes can be difficult to see on CT due to streak (beam-hardening) artifact. Frequently, multiple sequential, overlapping ablations are necessary to cover large lesions while carefully maintaining an adequate safety margin adjacent to critical structures. Post-procedural pain is also a concern [63].
Cryoablation techniques take advantage of the thermal properties of highly pressurized gases, typically argon or nitrous oxide. As the gas travels through the thermal probe to the tip, the gas expands at the applicator tip causing the temperature to rapidly drop. This is known as the Joule-Thomson effect, with temperatures of −80 °C to as low as −160 °C possible. A temperature between −20 and −40 °C is necessary and needs to persist 1 cm beyond the tumor periphery [66]. Some of the cryoprobes currently used for cryoablation are produced by Endocare® (Healthtronics/Endocare Incorporated, Irvine, California) and Galil Medical® (Galil Medical, Arden Hills, Minnesota). Cryoablation has the advantage of smaller ablation probes; it has the benefit of a clearly visible ablation zone (ice ball) during ablation, which can be seen on CT, MR imaging, or ultrasonography. It is also thought to have less post-procedural pain. More recently, newer probes offer post-ablation cauterization, in order to reduce the risk of post ablation bleeding.
MRgFUS is a noninvasive thermal ablation method that uses extracorporeal-focused ultrasound energy to heat and destroy tissues without the need for invasive placement of applicators. This technology takes advantage of the bone’s high acoustic absorption of ultrasound energy to palliate pain, presumably through destruction of periosteal innervation [67]. Evidence regarding MRgFUS of bone metastases is limited but promising. Potential complications include skin burns and injury to heat-sensitive structures, such as nerves and bowel.
26.8 Chemotherapy
Modern medical palliative chemotherapy regimens for painful bone metastases mainly include bisphosphonates and denosumab. The usefulness of these two classes of drugs derives from their ability to regulate osteoclast activity, each targeting a different component of the activation pathway. Bisphosphonates are an important advance in supportive care of patients with bone metastases. They inhibit normal and pathological osteoclast-mediated bone resorption by direct inhibition of osteoclast activity by cellular mechanisms that affect osteoclast attachment, differentiation, and survival. They also reduce osteoclastic activity indirectly, through effects on osteoblasts. In 1995, intravenous pamidronate was approved to treat patients with multiple myeloma or metastatic breast cancer, based on evidence from randomized controlled trials that pamidronate decreases the risk of skeletal complications [62]. In 2002, intravenous zoledronic acid was approved to treat patients with multiple myeloma and bone metastases from any solid tumor including prostate cancer [68]. Denosumab is a human monoclonal antibody for the treatment of osteoporosis, induced bone loss, bone metastases, rheumatoid arthritis, multiple myeloma, and giant cell tumor of bone. It is designed to target RANKL (RANK ligand), a protein that acts as the primary signal to promote bone loss. Denosumab was approved by US Food and Drug Administration (FDA) for use in postmenopausal women with risk of osteoporosis in June 2010 (Prolia®) and for the prevention of skeletal-related events in patients with bone metastases from solid tumors in November 2010 (Xgeva®) making it the first RANKL inhibitor to be approved by the FDA [62]. Direct comparisons of denosumab and bisphosphonates appear to favor denosumab [69]. Potential drawbacks of both drug families notably include the risk for osteonecrosis of the jaw and for the development of atypical femoral fractures [70, 71].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree