- •
CVD is highly prevalent in patients with CKD. Both conditions seem to worsen each other’s prognosis and complicate most interventions.
- •
The most common cause of death in patients with CKD or on dialysis is CVD.
- •
Patients with CKD often present with nonclassical ischemic heart disease symptoms and require added attention for referral and interpretation of appropriate CAD testing.
- •
Exercise testing in patients with CKD is limited because of a frequent inability to achieve the target heart rate and because the presence of left ventricular hypertrophy (LVH) and associated repolarization changes decrease the specificity of the test.
- •
Radionuclide MPI is an important management tool in this high-risk population.
- •
Like in the general population, abnormal radionuclide MPI identifies high-risk patients.
- •
CKD patients with visually normal myocardial perfusion scans remain at increased risk for CV mortality/morbidity. In patients with visually normal scans, a preserved coronary flow reserve as assessed by PET MPI identifies a low-risk subgroup.
- •
Radionuclide MPI, especially PET MPI, is an effective tool for CAD screening and risk assessment in patients undergoing evaluation for kidney transplant. A normal coronary flow reserve effectively excludes high-risk obstructive CAD and identifies low-risk patients.
Introduction
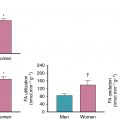
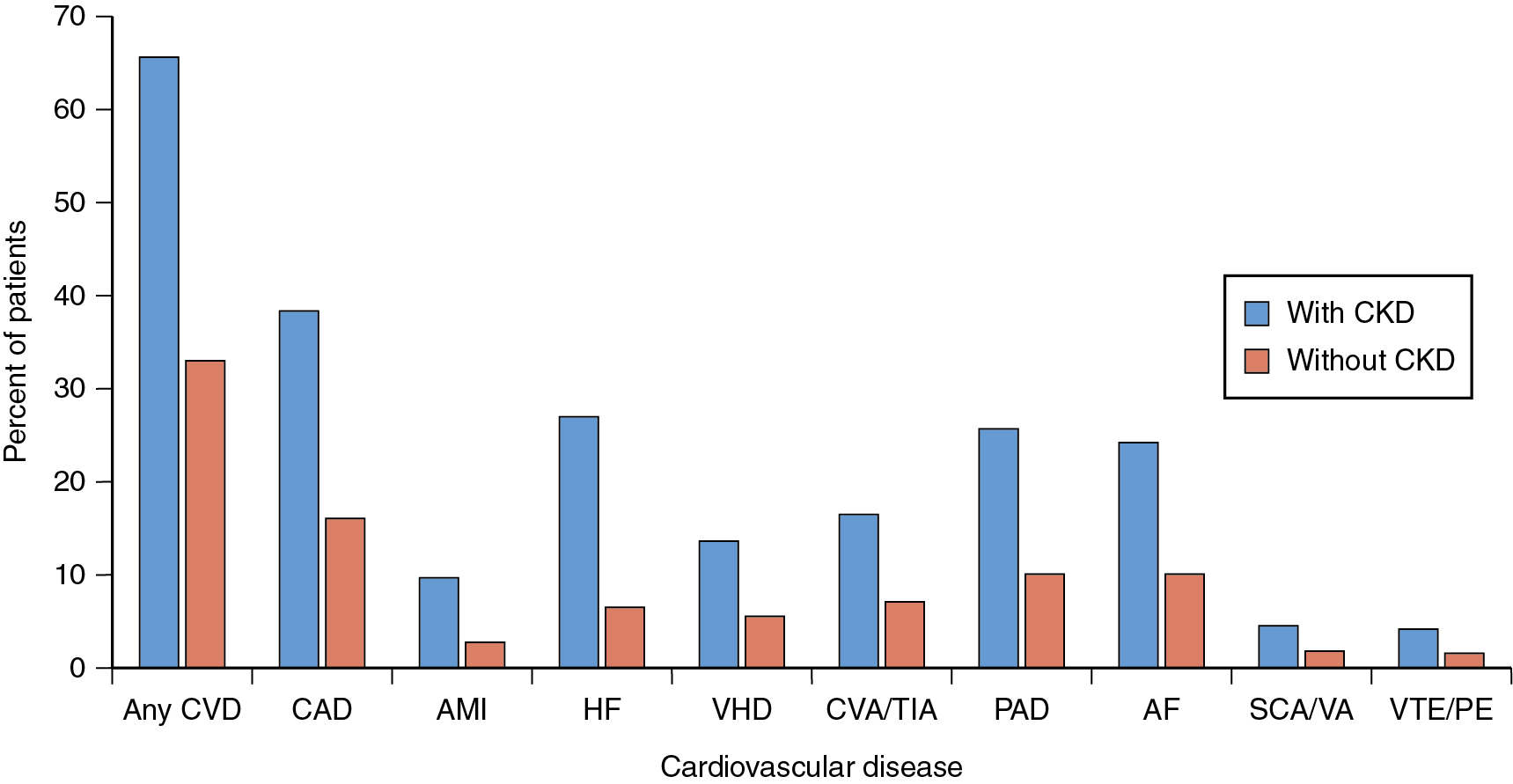
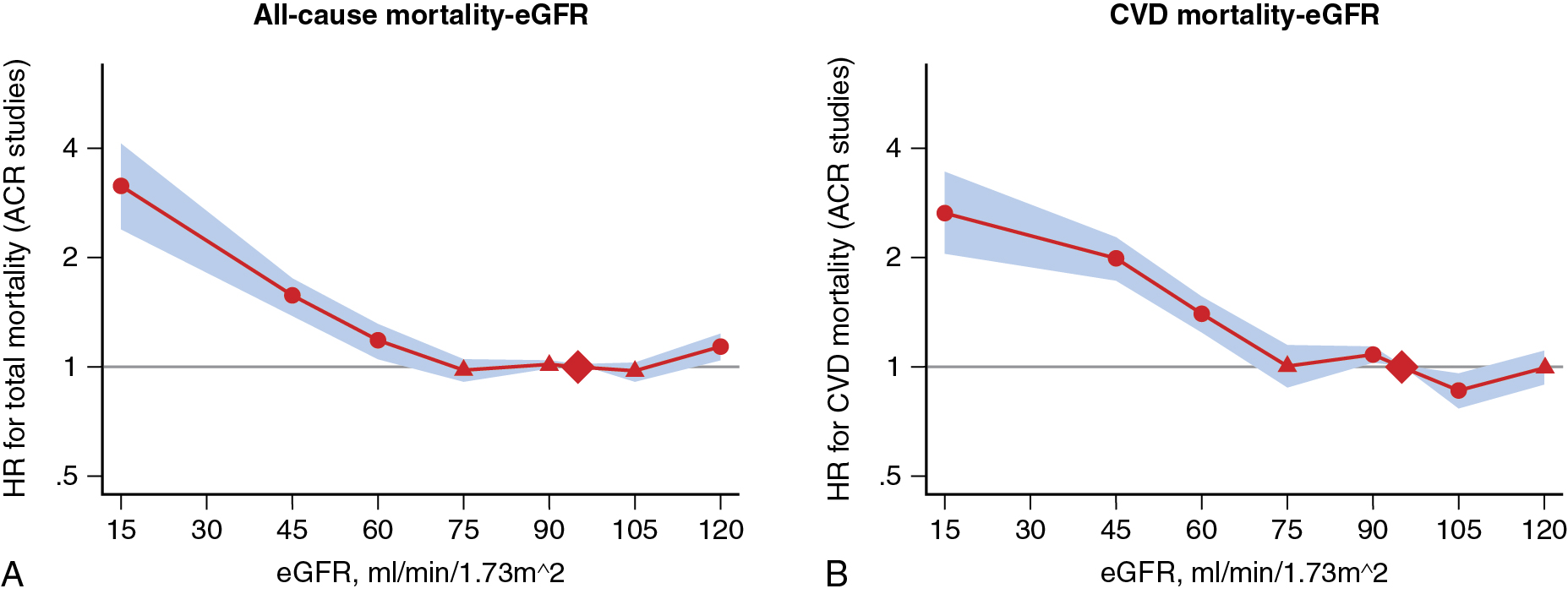
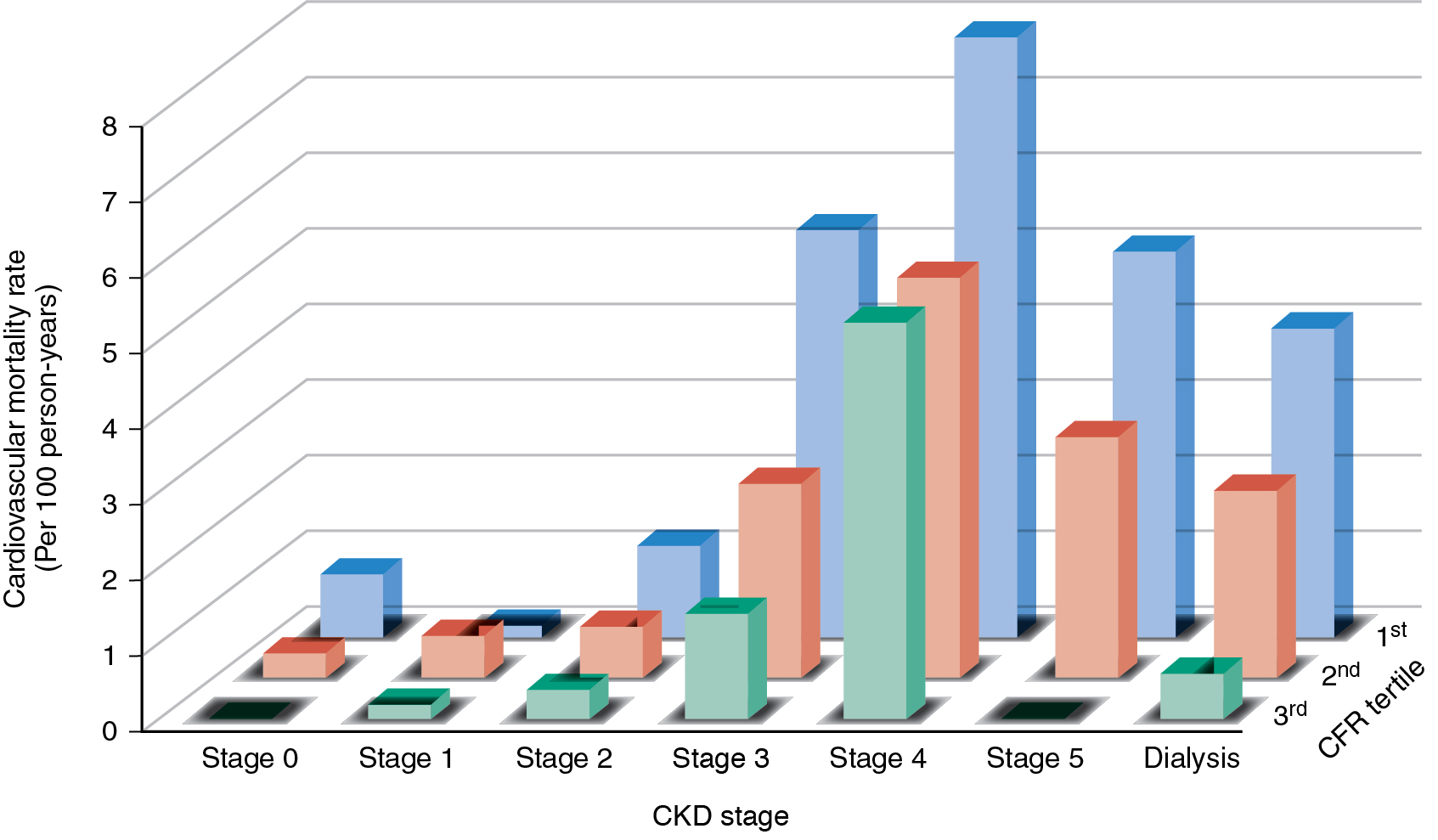
Chronic kidney disease (CKD) is highly prevalent in the general population, with current estimates at 15% (37 million people) of the U.S. population. Its presence remains associated with a significantly increased risk for cardiovascular (CV) morbidity and mortality, and it often complicates management of CV disease (CVD). Among individuals aged 66 years or older with CKD, the prevalence of CVD is nearly double that of those without CKD (64.5% vs. 32.4%). Specifically, among subjects with CKD, coronary artery disease (CAD) is present in nearly 40%, heart failure in about 30%, and peripheral arterial disease in 25% ( Fig. 15.1 ). In addition, CVD remains the leading cause of death in subjects with CKD, with the risk of all-cause mortality and CV mortality inversely proportional to estimated glomerular filtration rate (eGFR) ( Fig. 15.2 ).
It is important to note that CAD and attendant myocardial ischemia in patients with CKD or end-stage renal disease (ESRD) often lead to atypical presentations. Accordingly, a high degree of suspicion should be maintained in these patients with a high prevalence of CAD, especially with less typical symptoms such as dyspnea or fatigue. Patients can also be completely asymptomatic yet remain at high cardiovascular risk, especially those considered for kidney transplantation. Consequently, diagnosis and risk stratification of known or suspected CAD in patients with CKD and ESRD is important, especially in asymptomatic patients being considered for kidney transplant. In this chapter, we will review the strengths and weaknesses of noninvasive approaches to the evaluation of CAD in this special population with a focus on radionuclide imaging. Technical considerations regarding the radiotracers and protocols, identification and prevention of artifacts, and quantitative assessment of radionuclide images have been reviewed in Chapters 3 , 4 , and 5 . We will also discuss alternative approaches where appropriate.
Patient-centered clinical applications of radionuclide and multimodality imaging in patients with CKD
Case vignette 1: Screening for CAD
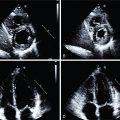
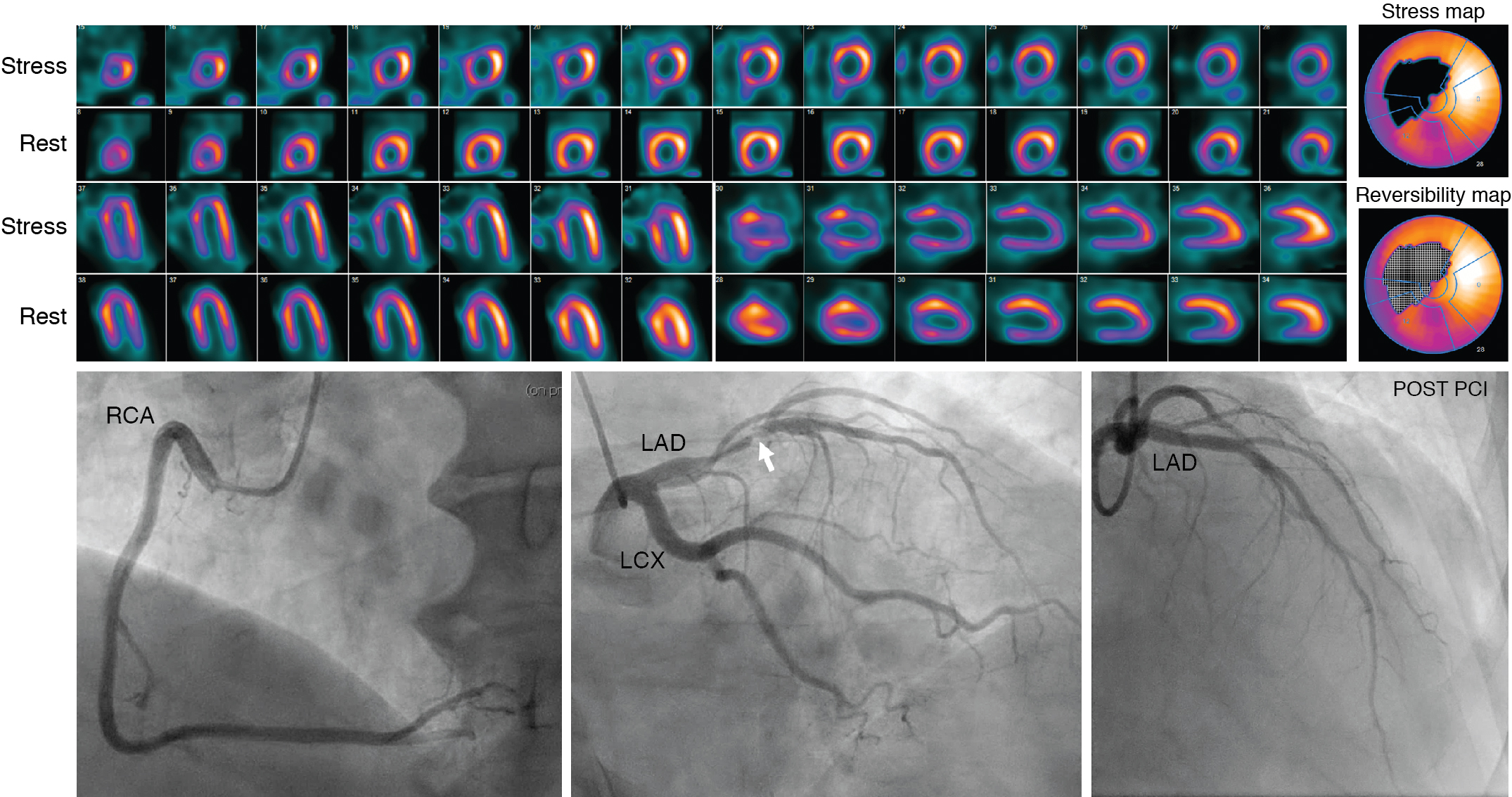
A 71-year-old male with a history of hypertension, obesity, type 2 diabetes, hypercholesterolemia, and former smoking presented with several weeks of exertional chest pressure and dyspnea. His clinical examination was remarkable for a resting blood pressure of 147/75 mm Hg, pulse of 78 beats per minute, body mass index of 28 kg/m 2 , and no evidence of volume overload. His baseline electrocardiogram (ECG) showed sinus rhythm with no significant ST-segment abnormalities. His blood work was remarkable for a creatinine of 1.6 mg/dL (eGFR of 43 mL/min/1.73m 2 ). Given his risk factors and symptoms, the patient underwent a vasodilator stress single photon emission computed tomography (SPECT) myocardial perfusion imaging (MPI) study. Gated imaging showed normal left ventricle (LV) size with an LV ejection fraction (LVEF) of 59% at rest and 67% post stress. The MPIs demonstrated a large and severe perfusion defect throughout the anterior and anteroseptal walls, the apical LV segments, and the LV apex, showing complete reversibility ( Fig. 15.3 ). Invasive coronary angiography demonstrated a 90% lesion in the proximal LAD (see Fig. 15.3 ). He underwent successful percutaneous coronary intervention (PCI) with placement of a drug-eluted stent to his LAD without complications. Follow-up creatinine was stable.
CAD is highly prevalent in patients with CKD, and it increases significantly with declining eGFR below 60 mL/min/1.73 m 2 . Screening for CAD in symptomatic patients with CKD is important because it is associated with a markedly increased risk for myocardial infarction (MI), heart failure, and death. In patients with CKD, similarly to those without CKD, imaging stress tests help identify and quantify ischemic burden, assess prognosis, and guide management. Exercise tolerance testing (ETT) is a commonly used test to assess functional capacity and help diagnose CAD. Nevertheless, patients with CKD often have exercise limitations from associated comorbidities, including anemia, deconditioning, and peripheral arterial disease. In addition, baseline ECG abnormalities, notably LV hypertrophy from chronic hypertension and secondary repolarization abnormalities, further limit the sensitivity and specificity of ETT findings. A meta-analysis by Wang et al. noted limited sensitivity (0.36, 95% confidence interval [CI] 0.21 to 0.54) with good specificity (0.91, 95% CI 0.83 to 0.96) but overall reduced diagnostic accuracy. Given all of the aforementioned reasons, it is not surprising to see that ETT without imaging in the CKD population remains at around 5% of all stress testing modalities used in this population.
Because of the exercise limitations previously outlined, pharmacologic stress testing is often used in patients with CKD, especially vasodilator stress radionuclide MPI. Regadenoson, a selective adenosine receptor agonist, is one of the most commonly used vasodilator stress agents. Because of its renal excretion, it has undergone specific testing in subjects with CKD or ESRD and been shown to be safe and effective. In a Cochrane meta-analysis of patients with ESRD, radionuclide MPI showed moderate accuracy with a pooled sensitivity and specificity of 0.74 and 0.70 for angiographically obstructive CAD, respectively. Dobutamine stress echocardiography (DSE) is an alternative stress imaging approach in patients with CKD. In a Cochrane meta-analysis of 13 studies, totaling 745 participants, sensitivity of stress echocardiography ranged from 44% to 96%, and specificity from 60% to 100%, with a pooled sensitivity of 0.79 (95% CI 0.67 to 0.88) and specificity of 0.89 (95% CI 0.81 to 0.94) for angiographically obstructive CAD. The use of CCTA is limited in patients with CKD because of the risk of contrast-induced worsening of renal function. Although the incidence of acute kidney injury was relatively frequent after CT angiography (up to 12%), in a study of predialysis patients (mean eGFR 12.7 mL/min/1.73 m 2 ), renal function returned to its baseline and there was no case of urgent dialysis required. Similarly, concerns associated with the risk of nephrogenic systemic fibrosis from gadolinium-based contrast agents limit the use of cardiac magnetic resonance (CMR) imaging in patients with advanced kidney disease, especially those with eGFR less than 30 mL/minute/1.73 m 2 , acute kidney injury, or on dialysis. Other CMR techniques based on T1 mapping or blood oxygen level–dependent imaging have shown promising results at detecting CAD and ischemia in the general population; however, these remain largely untested in patients with CKD or ESRD.
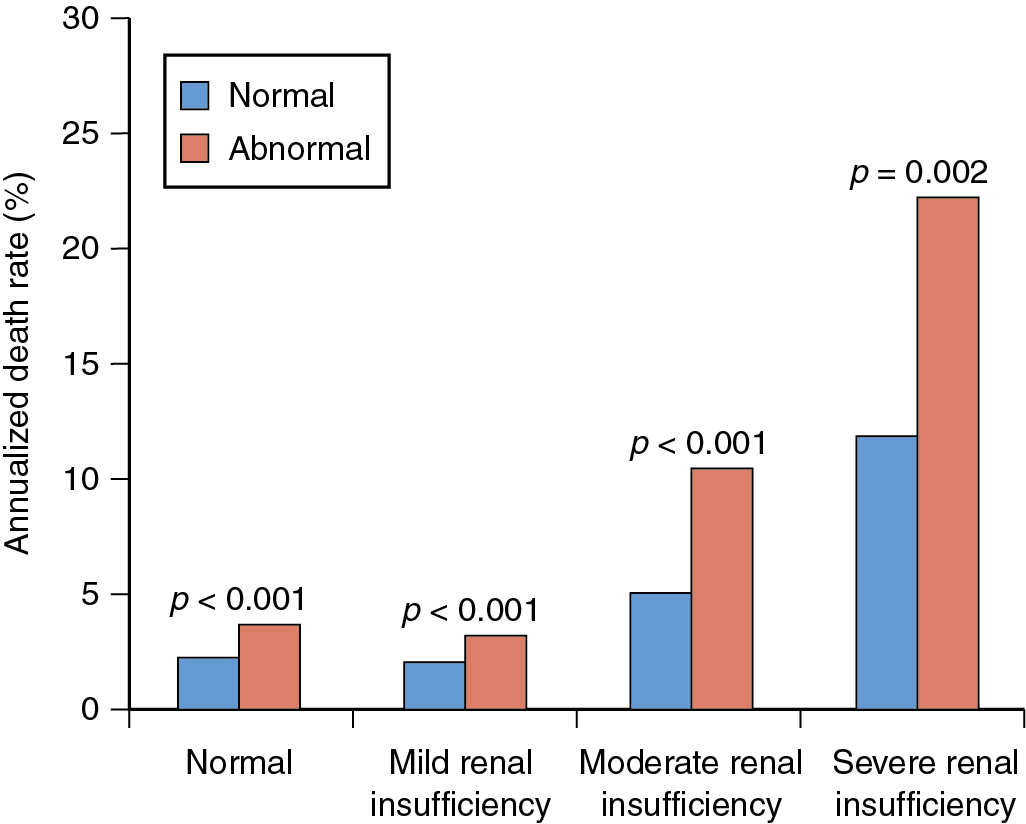
As discussed in Chapter 17 , radionuclide imaging provides robust risk stratification across a wide range of patient groups, including those with CKD. In patients with moderate or severe CKD, the presence of an abnormal scan was associated with a nearly twofold increase in the risk of death ( Fig. 15.4 ). Nevertheless, a normal myocardial perfusion scan in patients with severe CKD did not identify subjects at low risk because the annualized mortality was greater than 10% in patients with normal MPI. It is paramount to keep in mind this heightened risk even in the absence of overt myocardial perfusion defects on SPECT imaging. One possible explanation for this observation is that the semiquantitative assessment of myocardial perfusion with SPECT MPI lacks the sensitivity to uncover other manifestations of ischemic heart disease, namely, diffuse atherosclerosis and coronary microvascular dysfunction, which are prevalent in patients with CKD and associated with enhanced clinical risk. Consequently, given the superior sensitivity/specificity of positron emission tomography (PET)/computed tomography (CT) MPI compared with SPECT imaging, it is reasonable to consider a PET/CT MPI study to evaluate patients with CKD. Indeed, the addition of quantitative myocardial blood flow (MBF) information via PET offers an opportunity to interrogate the entire spectrum of ischemic heart disease, including the presence of obstructive CAD, diffuse atherosclerosis, and coronary microvascular dysfunction. In so doing, it provides improved risk stratification and risk reclassification in the general population and in patients with CKD, , including those on dialysis , ( Fig. 15.5 ). The presence of preserved coronary flow reserve identifies patients at low risk, averaging less than 1% of annualized cardiac mortality across the spectrum of renal function.
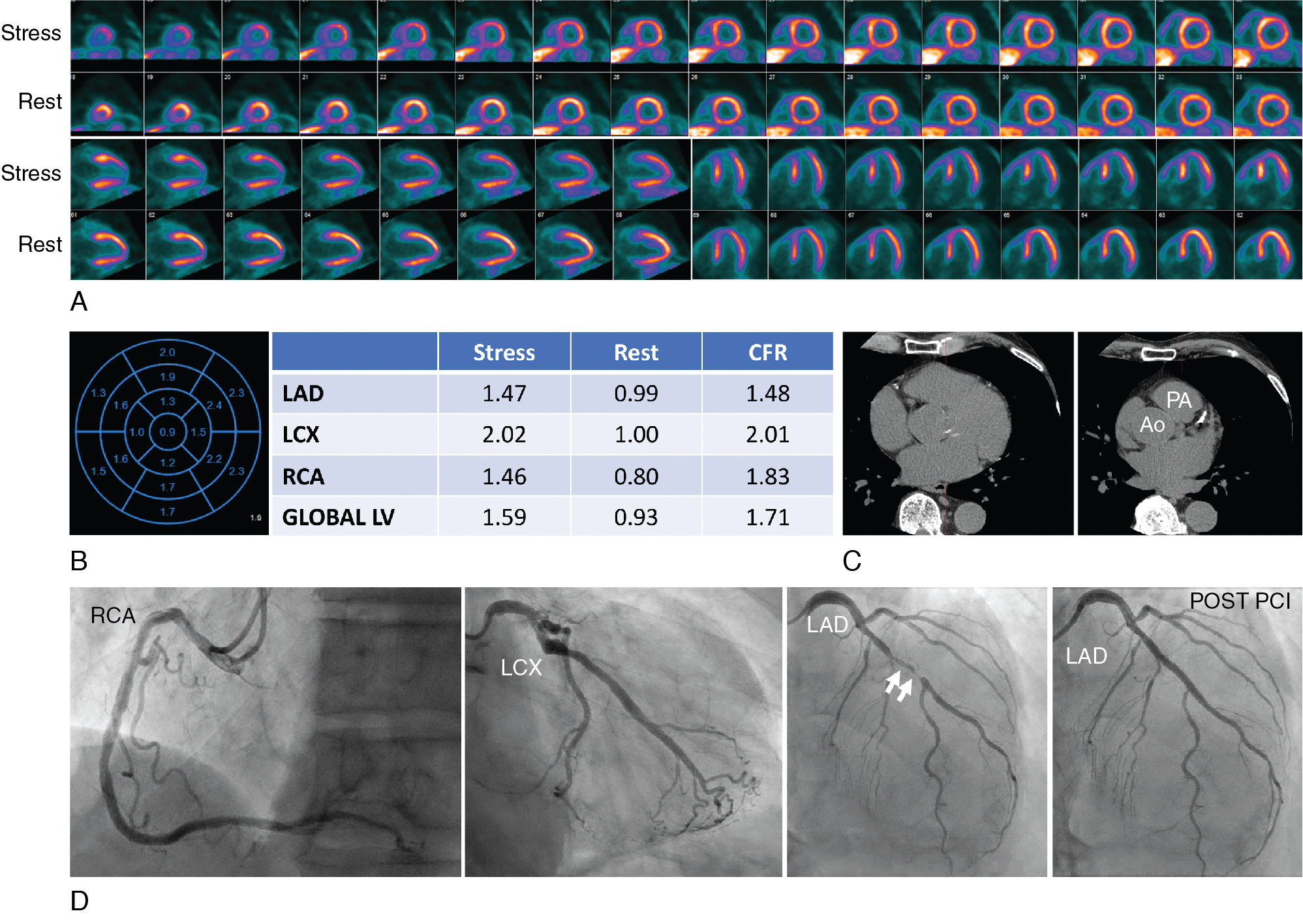
Case vignette 2: Evaluation of a patient with CKD and atypical chest pain
A 79-year-old male with hypertension, hypercholesterolemia, and CKD (creatinine of 2.0 mg/dL, eGFR of 31 mL/min/1.73 m 2 ) presented with atypical chest pain. He has had a long history of gastroesophageal reflux disease, which 7 years prior to this presentation led to a coronary angiogram that showed mild nonobstructive CAD. He denied dyspnea, palpitations, lightheadedness, or syncope. His rest ECG was notable for a left bundle branch block. A resting echocardiogram showed borderline concentric LV hypertrophy with normal cavity size, mild apical hypokinesis, and an LVEF of 52%. He underwent a rest and vasodilator stress 13 N-ammonia PET/CT myocardial perfusion study ( Fig. 15.6 ). The PET images demonstrated mild LV dilatation and normal right ventricle (RV) size and tracer uptake. The stress images showed a large and severe perfusion defect involving the mid anterior and anteroseptal walls, the apical LV segments, and the LV apex, showing complete reversibility. The ECG-gated images demonstrated severe hypokinesis of the anteroapical wall and LV apex with a LVEF of 48% at rest that increased to 56% during stress. The transmission CT scan showed dense calcification in the proximal LAD and RCA. The stress MBF was severely reduced in the LAD territory with a marked gradient from base to apex and an estimated PET fractional flow reserve (FFR) of 0.45 and a coronary flow reserve (CFR) of 1.48. The left circumflex (LCx) territory showed normal stress flow and flow reserve. The RCA territory showed moderately reduced stress flow without base to apical gradient and minimally reduced flow reserve. Overall, the PET findings were consistent with severe flow-limiting CAD in the mid LAD territory and nonobstructive CAD in the RCA territory. Follow-up invasive coronary angiography demonstrated a long 90% stenosis in the mid LAD and mild nonobstructive CAD in the RCA (see Fig. 15.6 ). The patient underwent successful PCI with stent placement in the LAD (see Fig. 15.6 ).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree