Vascular malformations are commonly encountered in the pediatric population. This article reviews the imaging appearances of simple and syndromic vascular malformations in infants and children that radiologists should know and provides imaging guidelines based on an evidence-based approach. Malformations are discussed within the framework of the International Society for the Study of Vascular Anomalies classification system.
Key points
- •
Ultrasound and MR imaging are the mainstay for imaging diagnosis of vascular malformations in infants and children.
- •
Vascular malformations should be characterized as low flow or high flow to direct management.
- •
Radiologic reports should employ terminology of the International Society for the Study of Vascular Anomalies classification to effectively communicate findings with multidisciplinary vascular anomaly subspecialists.
Introduction
Vascular anomalies encompass a wide-range of entities differing in clinical presentation, natural history, imaging findings, and management. Vascular malformations, a subset of vascular anomalies, are congenital and grow proportional to the patient. Vascular malformations are diagnosed most commonly in the first 2 decades of life and occur in approximately 0.5% of the population. ,
Mulliken and Glowacki devised a cell-oriented classification of vascular anomalies in 1982, delineating entities based on pathophysiology, namely endothelial cell characteristics. , Vascular tumors, such as hemangiomas, are true neoplasms that exhibit endothelial proliferation and hyperplasia. On the other hand, vascular malformations do not exhibit endothelial proliferation; rather, they the result from errors in vascular morphogenesis. Vascular malformations are categorized by predominant vessel type, including capillary, venous, lymphatic, arterial, and combinations thereof. The most widely accepted classification of vascular anomalies was created by the multispecialty International Society for the Study of Vascular Anomalies (ISSVA) in 1996 and was last revised in 2018. The ISSVA classification provides a standardized nomenclature, which takes into account clinical presentation, lesion histology, natural history, imaging findings, and management. Radiologists should utilize accurate ISSVA terminology when evaluating vascular malformations, because the use of improper or nonuniform terminology may lead to diagnostic delay and preclude appropriate clinical management.
Clinical history and physical examination often can lead to a correct diagnosis for a vascular malformation, although imaging remains essential for characterizing clinically indeterminate lesions and establishing the extent of disease as well as planning and monitoring treatment. , Radiologists may recommend additional imaging in cases of suspected syndromic association or multiorgan involvement. The purpose of this article is to review evidence-based imaging guidelines and recommendations for the evaluation of pediatric vascular malformations.
Evidence-based imaging algorithm
There is a relative paucity of rigorous scientific evidence concerning imaging of vascular malformations. This may in part be due to limited adherence to ISSVA terminology in clinical practice, which hasbeen shown to hamper management. In a study of more than 5000 referrals to a dedicated center of excellence for vascular lesions, 65% of referrals were vascular malformations and 35% were tumors, yet up to 50% of these referrals carried with incorrect referral diagnosis. Multidisciplinary vascular anomaly clinics have been shown to overcome to these hurdles and improve treatment success. Vascular anomaly centers of excellence have been shown crucial for accurate evaluation and treatment of these conditions. , Additionally, value-based care achieved through multidisciplinary vascular anomaly clinics has been shown to be financially advantageous to patients and the health care system at large.
Ultrasound (US) and MR imaging are the mainstay modalities in vascular malformation imaging work-up and often are complementary in establishing a diagnosis and treatment plan. Distinction between high-flow and low-flow lesions is essential to guide management. , In general, low-flow lesions that require treatment can be treated percutaneously with sclerotherapy, whereas high-flow lesions often require intravascular embolization. Dynamic US images and maneuvers and key MR imaging features, discussed later, can help distinguish types of low-flow lesions. , ,
Practical imaging approach to pediatric vascular malformations
Ultrasound
US should be the initial examination performed in the work-up of a suspected vascular malformation, because it often can make a correct diagnosis, especially in superficial and focal lesions ( Fig. 1 ). , , Superficial lesions may be entirely characterized with US, whereas deeper or more extensive lesions may be more difficult to assess. UA is widely available, is inexpensive, is noninvasive, and does not require sedation, which is an important consideration in pediatric patients.

Grayscale
Presence of a solid hypervascular mass is not consistent with the US appearance of vascular malformations and therefore an important distinction for the initial evaluation. The presence of a solid mass virtually excludes an arteriovenous malformation (AVM) and should prompt suspicion of a neoplasm. Cystic or heterogenous lesions are common appearances for lymphatic malformations (LMs) and venous malformations (VMs). VMs and LMs both can have anechoic spaces with intervening septations. If thrombosed or after intralesional hemorrhage, a malformation may demonstrate features, such as fluid-fluid levels or masslike solid appearance.
Spectral Doppler
Spectral Doppler US is an essential component of the initial imaging evaluation and is obtained relatively easily, especially in superficial lesions. Doppler is needed to distinguish high-flow and low-flow lesions, a critical part of the US interpretation. High-flow lesions, such as AVMs, demonstrate arterial waveforms, although septations within a low-flow malformation may contain vessels that demonstrate arterial or venous waveforms and should not be confused with AVMs. Low-flow lesions include VMs and LMs, discussed later.
Dynamic maneuvers
Compressibility, a feature suggestive of VMs and to a lesser extent LMs, can be assessed and documented on cine images easily. If a VM undergoes thrombosis, it may not compress, however. Maneuvers, such as tourniquet application for lesions located in the extremities and Trendelenburg for head and neck lesion, can demonstrate lesional engorgement suggestive of VM.
MR Imaging
MR imaging plays a pivotal role in the imaging evaluation of vascular malformations owing to excellent contrast resolution and ability to depict a lesion’s relationship to adjacent anatomic structures. MR imaging, better than US, allows depiction of the relationship of the lesion with adjacent critical structures, in particular nerves, which is important when intervention is contemplated. The full extent of a lesion, including focal, multifocal, or diffuse, can be assessed accurately with MR imaging. In a study comparing the utility of MR imaging versus US in head and neck vascular malformations, delineation of lesion extent was superior with MR imaging (54% of cases) compared with US (44% of cases). A disadvantage of MR imaging compared with US is the need for sedation in younger or uncooperative patients, because MR imaging examinations are lengthy and may require intravenous insertion for contrast administration.
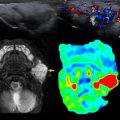
On routine MR imaging sequences, high-flow lesion, such as AVMs, may have tortuous signal flow voids indicative of fast flow. Intravenous contrast with dynamic, multiphase magnetic resonance angiogram (MRA) can subcategorize vascular malformations into low-flow and high-flow lesions. , , The feeding arteries and draining veins of high-flow lesions often are well depicted on time-resolved maximum intensity projection (MIP) reconstructions. Low-flow lesions, such as VMs, and LMs, typically are hypointense on T1-weighted images and hyperintense (fluid signal) on T2-weighted images and may contain internal septations.
Radiography
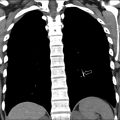
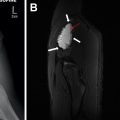
Radiographs often are performed in the initial evaluation of extremity pain or mass. Although soft tissue lesions often are occult on radiographs, important information sometimes can be gleaned regarding the nature of a vascular malformation. Phleboliths appear as small rounded calcifications and are characteristic of VMs. Studies have shown, however, that phleboliths are seen on radiographs in only 15% to 25% of cases ( Fig. 2 ), although can be seen in 80% of cases with high-resolution US Phleboliths that are present argue against LMs or high-flow lesions, such as AVMs. Radiographs also may give insight into the presence of intraosseous involvement. Intraosseous involvement occurs in up to 18% of AVMs and is important to recognize because it can lead to orthopedic complications. Radiographic findings of AVM intraosseous extension include lucent serpiginous lesions involving the bony cortex and medullary space. Radiographs also may be useful in the evaluation of limb length discrepancy and hemihypertrophy, which may occur in the setting of complex vascular malformation syndromes.

Computed Tomography
Computed tomography (CT) often is avoided in pediatric patients due to associated radiation burden. For example, in the evaluation of suspected VMs and AVMs, multiple phases are required to assess the lesion accurately, which increase the overall radiation dose. The relatively lower-contrast resolution of CT compared with MR imaging makes CT less helpful in the characterization of vascular malformations. CT can be used when MR imaging is not available, in cases less amenable to MR imaging evaluation, such as those with lung involvement, and in pediatric patients requiring cross-sectional imaging who are not able to undergo general anesthesia for MR imaging. If used, however, CT requires the administration of intravenous contrast. CT with its 3-dimensional capabilities can depict the degree of intraosseous involvement and deformity critical in the preoperative planning, particularly with AVMs. On noncontrast CT, VMs usually are low attenuation and enhance gradually after intravenous contrast. Depending on the timing and presence of contrast, VM appearance on postcontrast imaging may range from hypodense to hyperdense. Phleboliths easily are detected on CT due to the presence of calcium.
Interventional Radiology
Invasive imaging, including arteriography and venography, is used primarily in the treatment of vascular malformations, although may be required to fully characterize AVMs and other high-flow lesions.
Spectrum of pediatric vascular malformations
With appropriate adherence to ISSVA terminology in daily radiology practice, the most commonly encountered vascular malformations fall under the simple category, in particular LMs and VMs, with AVMs and arteriovenous fistulas (AVFs) seen less frequently. The first step when it comes to imaging is to distinguish low-flow and high-flow vascular malformations, because the treatment of these lesions is vastly different. LMs and VMs are low-flow lesions, and AVMs/AVFs are high-flow lesions. Vascular malformations associated with other anomalies are less common but often produce characteristic imaging appearances. Capillary malformations (CMs) often are diagnosed clinically, and MR imaging usually is not necessary. Combined malformations include varying combinations of LM, VM, CM, and AVM.
Low-flow Vascular Malformations
Venous malformations
VMs are the most common vascular malformations (67% overall), encountered in the head/neck (40% of cases), extremities (40%), and trunk (20%). VMs have 2 distinct morphologic forms that are reflected on imaging, including spongiform and dysplastic types. Dysplastic types frequently have a collection of ectatic veins, often spanning multiple tissue planes and compartments. Intramuscular VMs can have a delayed presentation, leading to higher morbidity; given these disparate appearances, diagnosis of VM not always is straightforward.
US typically shows a hypoechoic lesion, sometimes with internal anechoic channels. Up to 78% of VMs demonstrate presence of venous waveforms on Doppler US, with up to 16% showing no substantial flow detected. Arterial flow may be present within VM septations or normal vessels crossing through the VM and should not be confused with the arterial flow of an AVM. Phleboliths have been reported to be seen in 80% of cases with high-resolution US, previously reported in 16% to 22% on older US studies. ,
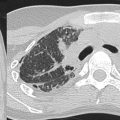
Applying compression is essential to distinguish from other malformations or tumors, because VMs without intralesional thrombosis demonstrate compressibility ( Fig. 3 ). With Valsalva or Trendelenburg maneuvers or the application of a tourniquet, VMs often engorge or fill on color Doppler ( Fig. 4 ).