Pelvicalyceal System and Ureter
Over the past several years, excretory urography (IVP) has been essentially replaced by computed tomography urography (CTU) for evaluation of the urinary tract. With the refinement of the techniques for CTU and increasing resolution of multidetector CT machines, it appears that virtually all lesions of the calyces, renal pelves, and ureters that could be seen on IVP can now be seen on CTU. Images from CTU, IVP, and antegrade/retrograde pyelography are used to illustrate the pertinent findings.
▪ PHYSIOLOGY
The intrarenal collecting system and ureter serve as conduits for transport of urine from the kidney to the bladder. The ureter transports urine from the renal pelvis to the bladder through a series of peristaltic contraction waves. Each contraction is preceded by a wave of depolarization of the smooth muscle membranes. These depolarization waves are generated from a site in the renal pelvis (the pacemaker) and spread from this point throughout the collecting system and down the ureter to the bladder. The depolarization causes coordinated contraction of the smooth muscle. The frequency of the depolarization and contraction waves is proportional to the rate of urine output from the kidney and is probably mediated by the degree of distention of the renal pelvis: the faster urine is made, the more rapidly the wall stretches, and the more frequently the pacemaker fires. In the normal ureter, each wave completely coapts the inner surfaces of the wall, so that the bolus of urine inferior to it is propelled ahead of the contraction wave.
The timing of the contraction waves in one ureter is independent of the other, and the ureter is usually empty between boluses. As the rate of urine formation increases, there is an increase in both the frequency and the volume of each peristaltic contraction. During maximal diuresis, the ureter may fill with urine so quickly that individual contraction waves do not form and the ureter fills with urine from the pelvis to the bladder as a continuous column.
With normal rates of urine flow, the renal pelvic pressure stays low and coaptation of the ureteral walls prevents urine from flowing retrograde back into the renal pelvis. The ureterovesical junction (UVJ) acts as a one-way valve and keeps urine from flowing from the bladder into the ureter, even when the ureteral musculature is relaxed. The ability of ureteral peristalsis and a
competent ureterovesical valve to keep pressure in the renal pelvis low is crucial: elevated renal pelvic pressure reduces renal medullary blood flow, which normally takes place through the relatively low-pressure vasa recta, and thus compromises renal function.
competent ureterovesical valve to keep pressure in the renal pelvis low is crucial: elevated renal pelvic pressure reduces renal medullary blood flow, which normally takes place through the relatively low-pressure vasa recta, and thus compromises renal function.
▪ PAPILLARY NECROSIS
Papillary necrosis is a clinical pathologic entity found in association with several diseases that affect the kidney. Each disease is thought to be associated with renal medullary ischemia, which causes necrosis and sloughing of papillary tissue. This sloughing results in characteristic radiologic findings.
Papillary necrosis varies greatly in severity and rate of progression. It may be part of a condition that produces severe acute renal abnormalities with massive sloughing of most papillary tissue accompanied by hematuria and colic or may follow an indolent course, with only the mildest of nonprogressing radiographic abnormalities and no clinical symptoms.
The possible etiologies of papillary necrosis may be recalled using the mnemonic nonsteroidal anti-inflammatory drug (NSAID): NSAIDs, sickle cell hemoglobinopathies, analgesic abuse, infection (such as tuberculosis and pyelonephritis), and diabetes mellitus. The most common etiologies are analgesic use, diabetes mellitus, and sickle cell anemia.
CAUSES OF PAPILLARY NECROSIS
N, NSAID (Nonsteroidal anti-inflammatory drugs)
S, Sickle cell disease
A, Analgesic nephropathy
I, Infection, especially tuberculosis
D, Diabetes mellitus
Analgesic Nephropathy
Analgesic nephropathy was first described in 1953 in Sweden, where several patients with interstitial nephritis were found to have consumed large amounts of analgesics, which usually consisted of combinations of aspirin and phenacetin. This combination is nephrotoxic, especially to the renal medulla and papillae, and particularly when consumed in large doses for a long time. Although over-the-counter combinations of aspirin and phenacetin have been removed from the market, they have been replaced by combinations of aspirin and acetaminophen. Both in combination and alone, these drugs may, in large doses, also produce renal disease. Similar analgesic nephropathy is caused by NSAIDs. Patients with renal disease resulting from analgesic abuse also have an increased incidence of transitional cell carcinoma (TCC), especially of the upper tracts.
Sickle Cell Disease
Sickle cell disease is the result of the homozygous hemoglobin S gene, which is present in approximately 10% of African Americans and in approximately 30% of black Africans. The gene alters the solubility of hemoglobin, which results in the classic sickle cell shape of erythrocytes; these cells are less malleable than normal and tend to occlude capillaries, producing regions of ischemia and infarction in the renal medullae and papillae. Up to 50% of patients with sickle cell disease develop renal papillary necrosis. Papillary necrosis may also occur in patients with sickle cell trait.
Infection
The association of pyelonephritis and papillary necrosis sometimes is confusing. In some cases, the main factor may be a comorbidity such as diabetes. However, when the kidneys are infected by tuberculosis, the bacilli initially lodge in the cortex and move down the nephrons to the medulla, where they cause focal caseating granulomata, which then slough portions of tissue and produce papillary necrosis.
Diabetes Mellitus
This disease produces various renal abnormalities. Ischemia caused by small-vessel disease can cause papillary necrosis, but the abnormalities are not limited to this region. Diabetic nephrosclerosis affects the entire kidney and ultimately may lead to severe renal failure.
Radiology
The plain film is commonly normal; rarely, a sloughed but retained papilla may develop a ring of calcification on its surface and become visible. If the whole papilla is necrotic, the calcifications may be 5 to 6 mm in diameter.
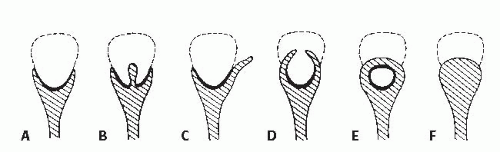
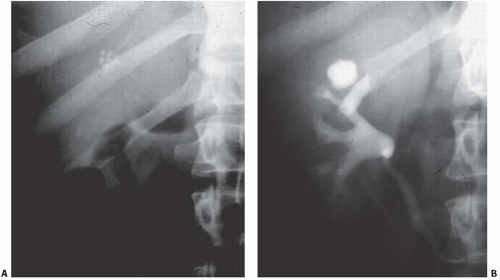
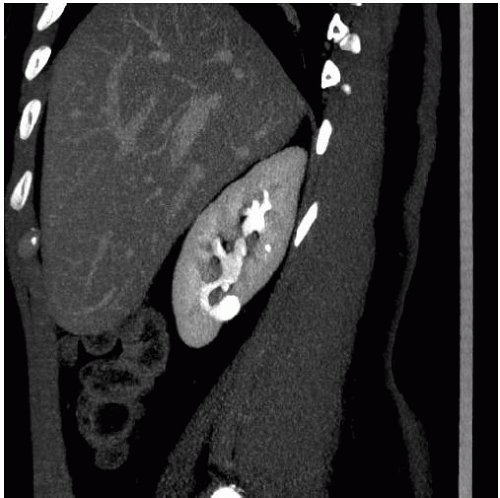
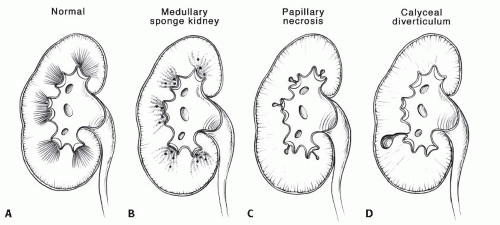
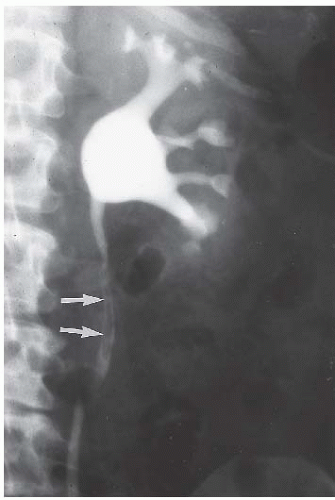
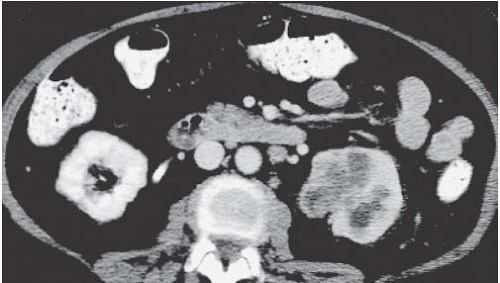
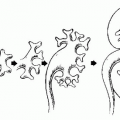
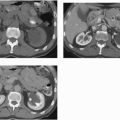
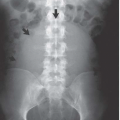
The classic radiologic features of renal papillary necrosis are best shown by excretory or retrograde pyelography but are also seen on high-resolution CTU, especially on coronal reformatted images. There are several patterns of calyceal changes (Fig. 14.1). Small collections of contrast medium may be seen in the papillary region (Figs. 14.2 and 14.3) and may be round, elongated, or irregular in shape. They may extend from the calyceal fornices or directly into the tip of the papillae (Fig. 14.4). The collections usually extend outside the interpapillary line. Central erosion of the papilla is known as the medullary type of papillary necrosis. Alternatively,
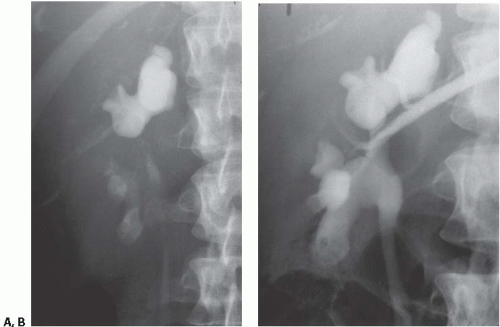
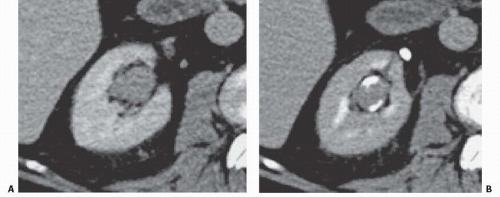
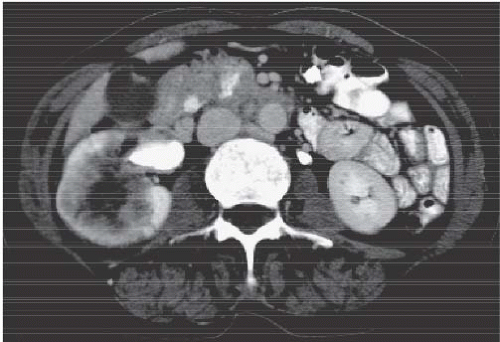
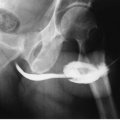
the entire papilla may become necrotic and slough; this is known as the papillary type of papillary necrosis. If the sloughed tissue has disappeared from the calyx and has been passed down the ureter, the calyx may be blunted with no remnant of the papilla at all. Alternatively, the entire papilla or large pieces of it may be retained and may form circular or irregular filling defects (Fig. 14.5); these defects may be calcified on their peripheries (Fig. 14.6).
the entire papilla may become necrotic and slough; this is known as the papillary type of papillary necrosis. If the sloughed tissue has disappeared from the calyx and has been passed down the ureter, the calyx may be blunted with no remnant of the papilla at all. Alternatively, the entire papilla or large pieces of it may be retained and may form circular or irregular filling defects (Fig. 14.5); these defects may be calcified on their peripheries (Fig. 14.6).
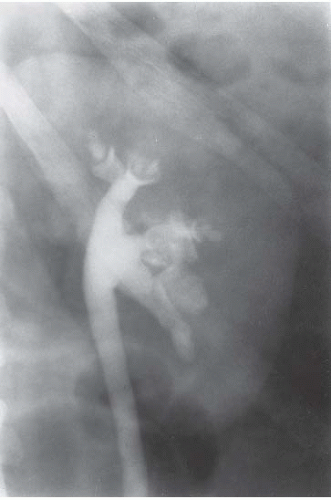 FIGURE 14.2. Papillary necrosis. Collections of contrast medium of varying sizes are present in the papillary regions adjacent to normal calyces. |
▪ MEDULLARY SPONGE KIDNEY (BENIGN TUBULAR ECTASIA)
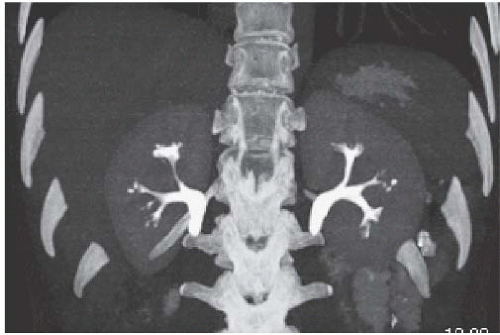
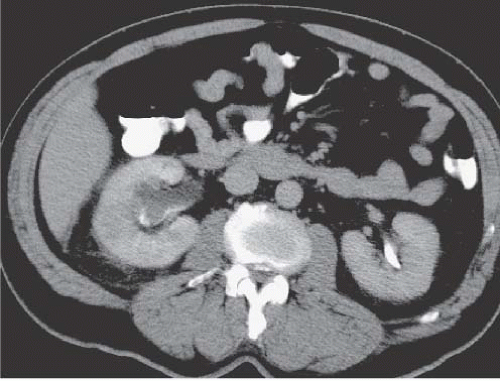
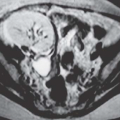
Medullary sponge kidney (MSK) is a condition characterized by dilatation of the renal collecting ducts (of Bellini) in the papillae. The dilatation may be cylindrical or saccular, and on contrast studies such as urography and CTU, they are seen as parallel streaks of contrast in the papillae. This finding is called benign tubular ectasia and can involve only one, a few, or all of the calyces. In some cases, there is sufficient stasis of urine in the tubules to result in the formation of small linear stones. This is termed MSK and is one cause of medullary nephrocalcinosis (Fig. 14.7).
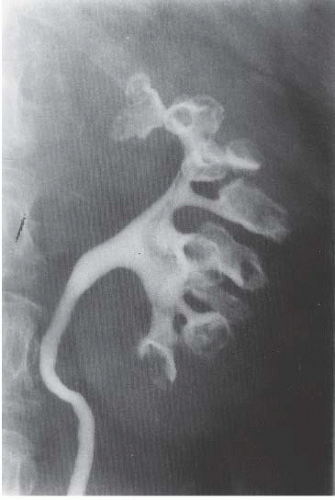 FIGURE 14.4. Papillary necrosis. Retrograde pyelogram shows rounded filling defects in many calyces representing sloughed, but retained, papillae. |
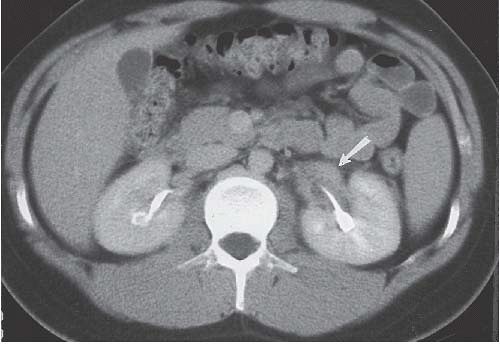
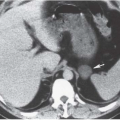
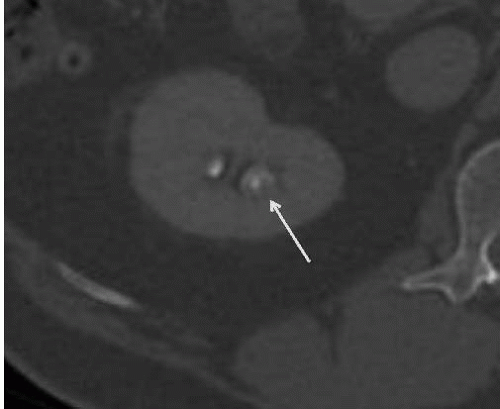 FIGURE 14.5. Papillary necrosis. The contrast-filled papillary tip (arrow) is seen on this axial CT image. |
Patients with benign tubular ectasia are asymptomatic. Patients with MSK may have microhematuria, and on occasion stones may erode into the collecting system and produce episodes of renal colic.
▪ CALYCEAL DIVERTICULUM
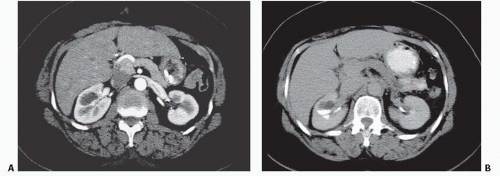
Calyceal diverticulum is a cystic cavity connected to the intrarenal collecting system by a narrow neck. It can arise from any part of the collecting system from the renal pelvis to a fornix but is most commonly seen extending from the fornix of an upper or lower pole calyx. These lesions can range in size from a few millimeters to several centimeters in diameter and typically lie outside the interpapillary line. They may contain calcified stones, which may be single, but characteristically appear as a cluster of small stones. Calyceal diverticula, with or without stones, are usually asymptomatic but may cause microhematuria. The stones that form within them may rarely pass and cause ureteral colic, but are usually confined to the diverticulum by the narrow neck.
On imaging studies, a calyceal diverticulum classically appears as a spherical collection of contrast medium adjacent to a papilla but outside the area where calyces typically lie. A cluster of small stones outside the normal topography of the calyces is usually an indicator of a calyceal diverticulum (Fig. 14.8). If the diverticulum is large and arises from the pelvis, it is more likely to cause infundibular or calyceal compression and displacement. The fluid-filled cavity comprising a calyceal diverticulum, together with any stones it contains, may be seen on CT or ultrasound. The diverticulum will typically opacify after the collecting system due to retrograde filling (Figs. 14.9 and 14.10).
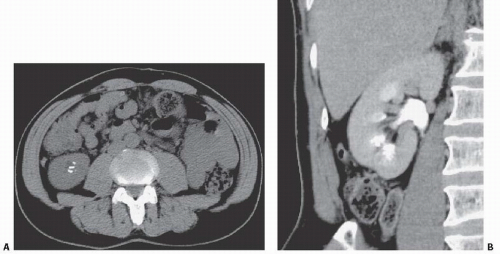 FIGURE 14.7. MSK. A: Noncontrast CT shows elongated medullary stones in the right lower kidney (no stones were seen elsewhere). B: CT urogram shows coarse striations around right lower pole calyces. |
Although MSK, papillary necrosis, and calyceal diverticula all have contrast outside the calyx, the size and location of the contrast determine the underlying etiology (Fig. 14.11).
▪ RENAL SINUS
Renal Sinus Fat
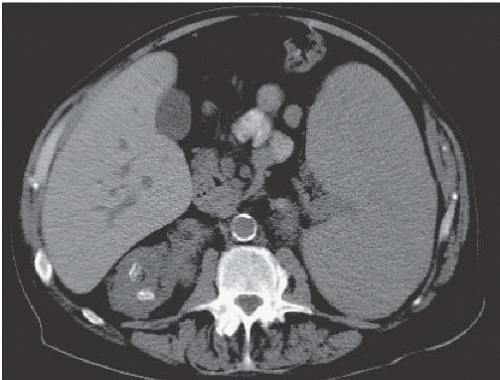
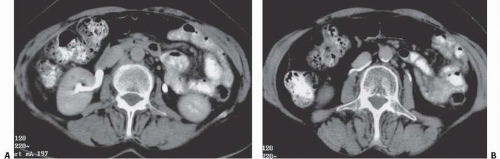
Fat is normally found in the renal sinus in varying amounts surrounding the pelvicalyceal and vascular structures. This fat is so sparse in infancy and childhood as to be invisible by clinical imaging. But with advancing age, this fat increases and becomes detectable. In anatomically normal patients, this change probably reflects the loss of renal parenchymal volume that occurs with age. In patients who are obese, who have excess truncal fat from exogenous steroids or Cushing syndrome, or whose renal parenchymal volume has been reduced by disease, the amount of sinus fat may be considerable and is known as sinus lipomatosis. The extreme of this condition is termed replacement fibrolipomatosis. In these patients, there is reactive deposition of dense fibrofatty tissue in the renal sinus in response to severe loss of renal parenchyma. This parenchymal wasting is often due to chronic stone disease. In both entities, the infundibulae may look very attenuated, or thinned, or even slightly bowed, but infundibular obstruction is not a feature of this condition.
As with fat elsewhere, renal sinus fat is echogenic, resulting in the central echo complex in normal kidneys. Enlargement of the central renal echo complex is seen in sinus lipomatosis or replacement fibrolipomatosis, and the parenchyma may be thinned. CT directly reveals the sinus fat (Fig. 14.12), which is usually of the same density as other retroperitoneal fat.
Renal Sinus Hemorrhage
Hemorrhage into the renal sinus or collecting system may occur in various clinical circumstances including benign or malignant renal tumors, arteritis, aneurysms, arteriovenous malformations, trauma, coagulation disorders, or other bleeding diatheses. In the central portion of the kidney, bleeding may occur into the renal sinus or the wall or lumen of the collecting system.
The radiologic appearance depends on the compartment into which bleeding has occurred. Hemorrhage into the collecting system may produce intraluminal clots that appear as filling defects. These filling defects are usually well defined, conform to the configuration of the collecting system, and are associated with gross hematuria. Because the kidney produces urokinase, blood clots often dissolve, although they may reform if bleeding persists. Blood clots have moderate echogenicity, less than that of renal sinus fat. They do not produce the acoustic shadowing seen with stones. CT demonstrates blood clot as an opacity slightly denser than adjacent soft tissues (Fig. 14.13). They can be clearly distinguished from stones, because even uric acid stones, which are not seen on plain films, have attenuations of 300 to 500 Hounsfield units (HU) and are clearly seen on CT. Blood clots may also pass down the ureter and may cause ureteral obstruction.
Hemorrhage into the wall of the intrarenal collecting system and/or ureter is seen most frequently in patients with a bleeding disorder or those receiving anticoagulant medication. These patients often present with hematuria. CT reveals a high attenuation thickening of the renal pelvic or ureteral wall due to the suburothelial hemorrhage (Fig. 14.14).
The radiographic appearance of bleeding into the renal sinus includes narrowing of the major infundibula, renal pelvis, or proximal ureter. CT can clearly identify the source of mass effect in the renal pelvis (Fig. 14.15). The density of the hematoma depends on the age of the lesion. Acute thrombus is denser than unenhanced renal parenchyma. Over time, the clot may liquefy to a water density or may be resorbed.
Intraluminal, intramural, and renal sinus hemorrhage may occur independently, in combination, or accompanied by renal parenchymal or perinephric bleeding. The radiographic evaluation should be directed not only at diagnosing this hemorrhage but also at identifying the underlying etiology. In some cases, extensive bleeding precludes an adequate CT examination, and a follow-up study may be necessary to exclude a small neoplasm or other subtle condition.
 FIGURE 14.12. Replacement fibrolipomatosis. End-stage kidneys with fibrofatty tissue filling the renal sinuses, which are enlarged due to renal parenchymal atrophy. |
 FIGURE 14.14. Blod clot in collecting system. Filling defects in the lower pole collecting system A: and ureter B: are due to blood clots. |
▪ VASCULAR IMPRESSIONS/CROSSINGS
The renal artery and vein and their major branches are in close association with the collecting system and can cause bandlike impressions on the infundibulae and renal pelvis. Vessel abnormalities protruding into the renal sinus, such as aneurysms (Fig. 14.16) or arteriovenous malformations, may also indent the collecting system. Tortuosity or aneurysms of the iliac vessels or aorta may deflect the ureters. Patients with severe renal artery stenosis may form collaterals that produce vascular notching on the ureter; retroperitoneal branches from the aorta supply the ureteral artery, which enlarges and becomes tortuous before supplying flow that enters the renal artery distal to the stenosis. Venous collaterals associated with inferior vena cava or renal vein obstruction may also produce an irregular notching of the ureter. At several well-defined points between the calyces and the bladder, vessels crossing the collecting system may actually result in obstruction to the normal flow of urine; these are discussed in the following paragraphs.
Fraley Syndrome
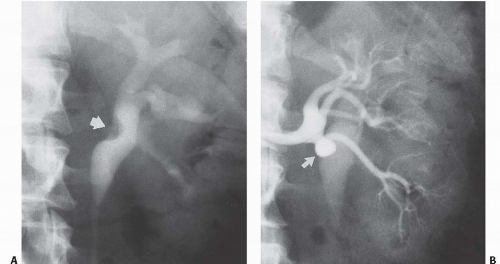
Nonobstructive bandlike indentations of the infundibulae or renal pelvis are the typical findings when a renal artery or vein lie immediately adjacent to the collecting system. This is most frequently seen involving the upper pole infundibulum or anterior surface of the renal pelvis. On rare occasions, the upper pole infundibulum may be scissored between two vessels resulting in hydrocalyx and pain. Alternatively, the pressure from a single crossing vessel can also result in the finding of an isolated hydrocalyx (Fig. 14.17). Either is known as Fraley syndrome.
Crossing Vessels in Ureteropelvic Junction Obstruction
Ureteropelvic junction (UPJ) obstruction is typically caused by a deficiency or derangement of the smooth muscle at the UPJ, preventing transmission of a normal peristaltic wave. However, crossing vessels, typically accessory renal arteries and veins, may cause extrinsic compression and varying degrees of obstruction of the UPJ and are found in association with up to 65% of UPJ obstructions. These vessels may exacerbate an intrinsic obstruction and can complicate endoscopic treatment of the UPJ obstruction by excessive bleeding or reduced treatment success rates. It is therefore necessary that accurate preoperative imaging be performed to allow for appropriate treatment planning, as a crossing vessel is an indication for open or laparoscopic repair for many surgeons. During antegrade or retrograde pyeloureterography, a vascular impression may be seen at the UPJ in these patients (Fig. 14.18). CT angiography can usually identify the presence of a crossing vessel.
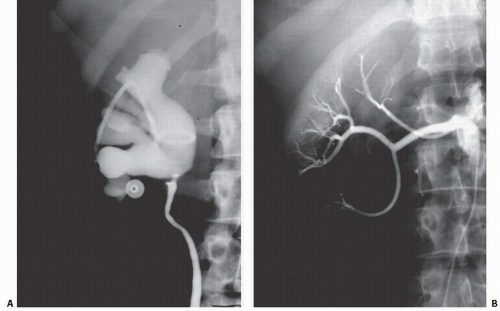
Retrocaval (Circumcaval) Ureter
There are two varieties of this condition, and both occur only on the right side. In the less common situation, the proximal ureter courses medially behind the IVC and then swings around it before descending into the pelvis. This type is seldom obstructive and is known as the 7 configuration referring to the course of the ureter medially and then down. In the more obstructive type, the proximal ureter dilates and becomes tortuous before traversing behind the IVC; this is often referred to as resulting in the Reverse
J configuration of the dilated proximal ureter. CT clearly delineates the retrocaval course of the ureter in both configurations (Fig. 14.19).
J configuration of the dilated proximal ureter. CT clearly delineates the retrocaval course of the ureter in both configurations (Fig. 14.19).
Ovarian Vein Syndrome
The right ovarian vein crosses the ureter at approximately the level of L-3, but it seldom causes a distinct vascular impression. Cases of mild ureteral dilation and hydronephrosis proximal to the level where the ovarian vein is presumed to cross the ureter have been reported, sometimes in association with pyelonephritis. These been attributed to thrombosis of the vein. The syndrome typically occurs in young women who have had one or more pregnancies and is known as the ovarian vein syndrome (Fig. 14.20).
▪ BENIGN COLLECTING SYSTEM AND URETERAL MASSES
Papillomas
Papillomas are benign tumors of the transitional epithelium that may appear in the collecting system, ureter, or bladder; they are rare in the upper tract. Some pathologists do not make a clear distinction between papillomas and low-grade transitional-cell carcinomas. Papillomas appear as polypoid filling defects that may have a recognizable stalk. Inverted papillomas are extremely unusual lesions that have a central core composed of transitional epithelium rather than of connective tissue and are covered with the normal layer of transitional cell epithelium. They are small and intrinsically
benign, but a few patients have been described in whom urothelial malignancy has been found near the inverted papilloma or elsewhere in the urinary tract.
benign, but a few patients have been described in whom urothelial malignancy has been found near the inverted papilloma or elsewhere in the urinary tract.
Connective Tissue Tumors
Connective tissue tumors of the collecting system are rare; both malignant and benign tumors originating from muscular, vascular, fibrous, and neural tissues have been described. They present as masses arising from the calyceal, renal pelvic, or ureteral walls. As a group, their surfaces may be less irregular than those of epithelial malignancies, but their specific diagnoses cannot be established radiologically.
Fibroepithelial Polyps
The most common of the nonepithelial tumors of the ureter is the benign fibrous polyp, also known as a fibroepithelial polyp. The lesion consists of a core of fibrous and vascular tissue covered with normal transitional epithelium. The lesion is usually solitary, but multiple lesions have been reported. These polyps may bleed slightly or cause obstruction and flank pain. They are not premalignant.
Imaging may reveal the lesion to be mobile, owing to its long stalk. The polyps vary in size from a few millimeters to many centimeters in length. Most fibroepithelial polyps have a smooth cylindrical appearance (Fig. 14.21), but multitentacled or frondlike appearances have been reported. Unlike primary malignant tumors, these polyps are most commonly found in the proximal one-third of the ureter and typically occur in patients 20 to 40 years of age. They should be distinguished from vermiform (wormlike) clots in the ureteral lumen.
▪ MALIGNANT TUMORS
Primary Urothelial Tumors
Fewer than 10% of primary renal malignancies develop in the intrarenal collecting system, and tumors of the ureter are three to four times less comon than those occurring in the renal pelvis. TCCs of the bladder are much more common than tumors of the upper urinary tract due to the reservoir function of the bladder; this results in longer contact between carcinogens in the urine and the urothelium. Most urothelial malignancies are TCCs; squamous cell carcinomas are unusual, and adenocarcinomas are rarer still. The histologic types are not distinguishable radiologically.
TCCs have a tendency to be multiple, so that both synchronous and metachronous lesions may be seen in a given patient; 70% of patients have an antecedent or subsequent urethelial lesion elsewhere. Up to 40% of patients with TCCs of the collecting system will have carcinomas of the bladder diagnosed within a short time. Regardless of location, 40% of papillary tumors, and most nonpapillary lesions, are invasive lesions.
If TCCs of the upper tract are treated with local resection, the remaining collecting system and ureter are particularly prone to develop another tumor. For this reason, complete nephroureterectomy, including resection of a generous cuff of bladder around the ipsilateral ureteral orifice, is the standard therapy for upper tract lesions that are resectable at the time of diagnosis. Predisposing factors that seem to place patients at risk for developing TCC include analgesic abuse, exposure to aniline dyes or petroleum derivatives, administration of cyclophosphamide, and a long history of heavy smoking. Long-standing upper tract stone disease may be a risk factor for squamous cell carcinoma; whether leukoplakia or upper urinary tract cholesteatomas increase the risk is debatable.
Any upper urinary tract tumor may present with gross or microscopic hematuria. The patients may have pain, especially if the tumors have caused hydronephrosis, and voided urine may contain exfoliated malignant cells that are detectable on cytologic examination.
Upper urinary tract tumors can be staged by a scheme similar to that used for bladder carcinomas. Stage 0 tumors are limited to the transitional epithelium, stage A lesions have invaded the lamina propria, and stage B tumors have invaded into, but not completely through, the muscularis. The prognosis for these less invasive tumors is good; 50% survivals of 5 to 7 years are typical. Stage C tumors have
invaded the adjacent fat, renal parenchyma, or other retroperitoneal tissues; and stage D tumors have metastasized (nodal and lung metastases are common). The prognosis for these more invasive lesions is usually poor, and many patients die of carcinomatosis within 1 year. Overall, patients with ureteral tumors have a worse prognosis than those with tumors of the renal pelvis at the same stage or grade.
invaded the adjacent fat, renal parenchyma, or other retroperitoneal tissues; and stage D tumors have metastasized (nodal and lung metastases are common). The prognosis for these more invasive lesions is usually poor, and many patients die of carcinomatosis within 1 year. Overall, patients with ureteral tumors have a worse prognosis than those with tumors of the renal pelvis at the same stage or grade.
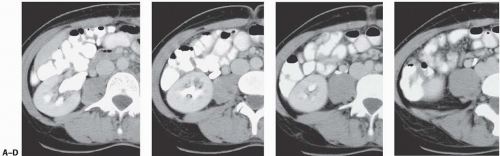
Small carcinomas of the collecting system may be detected by CT or retrograde urography (Fig. 14.22). Tumors may appear as intraluminal filling defects, which may be polypoid or flat; their surface is usually fronded or irregular (Fig. 14.23). Reconstructed images may be useful in depicting these urothelial lesions (Fig. 14.24). If the tumor has completely obstructed an infundibulum, the associated calyx may fail to opacify (the so-called amputated calyx) or evidence of local hydronephrosis may be visible (Fig. 14.25). If the renal pelvis or UPJ is obstructed, varying degrees of failure to function and hydronephrosis involving the entire kidney may be encountered.
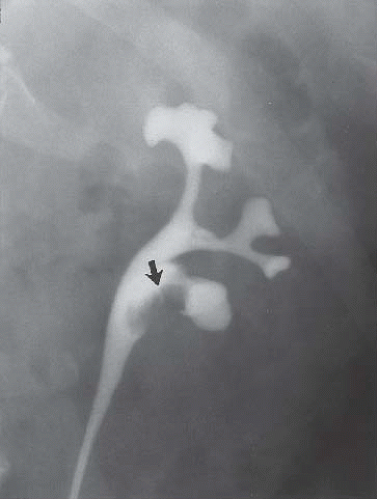 FIGURE 14.22. TCC of the renal pelvis. Retrograde pyelography reveals a solitary renal pelvic filling defect (arrow). |
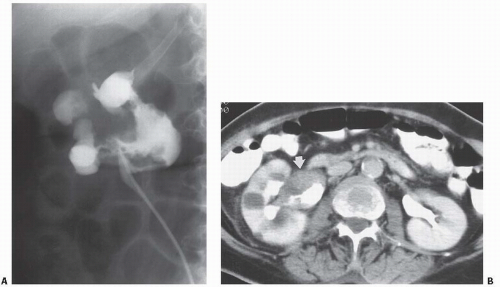 FIGURE 14.23. TCC. A: Retrograde pyelography demonstrates a large, irregular filling defect in the renal pelvis. B: The soft-tissue mass (arrow) is shown on this enhanced CT examination. |
The differential diagnosis of a filling defect in the upper urinary tract includes tumor, blood clot, stone, sloughed papilla, fungus ball, involvement of the urothelium by infection or inflammation, and invasion of the collecting system by a renal parenchymal tumor. A blood clot can be virtually excluded if there is no gross hematuria. Conversely, the diagnosis of blood clot is supported if the CT density is consistent with clot or if subsequent examinations show the filling defect to lyse and disappear. Stones may be identified on ultrasound by their echogenic surface and acoustic shadowing. On CT, even uric acid stones exhibit attenuation in the range of 300 to 500 HU, which is much higher than either tumors or blood clots. A sloughed papilla sometimes may look like a polypoid tumor, but is usually associated with a blunted calyx at the site from which the papilla detached. A fungus ball (mycetoma) may be diagnosed by finding hyphae in the urine. Rarely, cholesteatomas may mimic tumors. A normal papilla may protrude into the renal pelvis without an intervening infundibulum; contrast in the papilla’s fornix may produce a dense ring around the lucency and provide a clue to the diagnosis (Fig. 14.26). Benign strictures may mimic obstructing tumors; these are usually caused by tuberculosis and may be accompanied by other signs of renal tuberculosis and sometimes by pyuria and positive urine cultures.
INTRALUMINAL FILLING DEFECTS
Tumor (benign or malignant)
Blood clot
Sloughed papillae
Myecetoma (fungus ball)
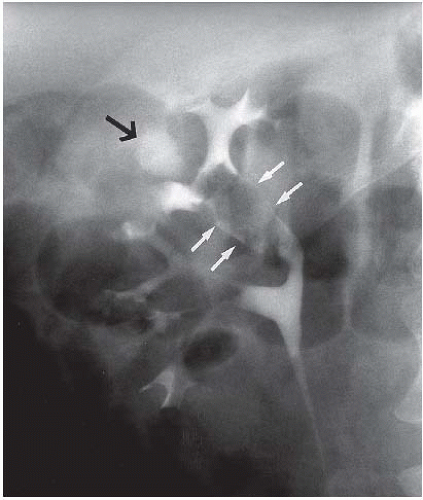 FIGURE 14.24. TCC. The tumor (white arrows) is obstructing the infundibulum and causing hydronephrosis of one calyx (black arrow). |
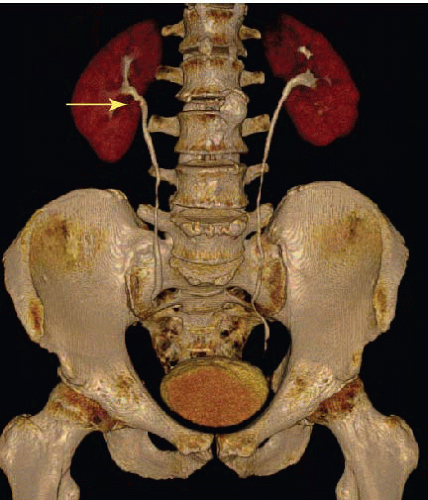 FIGURE 14.25. Urothelial carcinoma. The filling defect in the proximal right ureter (arrow) on these reconstructed images was found to be a TCC. |
Ultrasound is not as sensitive as urography for detecting small tumors and has little value for demonstrating ureteral lesions. Ultrasound reveals TCC to be a relatively hypoechoic region (Fig. 14.27) within the renal sinus, which normally is quite echogenic. Ultrasound may also demonstrate any hydronephrosis that the tumor has produced.
CTU or standard excretory phase CT, on the other hand, reveals the mass in either the renal pelvis or the ureter, and also will depict any associated hydronephrosis. The mass is usually of soft-tissue density and may enhance slightly with intravenous contrast administration. The sensitivity of CTU in detecting urothelial lesions of the upper urinary tract is greater than 90% when axial, coronal reformatted, and curved-planar reformatted images are used. Large tumors in the renal pelvis may contain low-density necrotic regions that do not enhance. If the tumor appears as a relatively discrete intraluminal mass with clean fat surrounding either the renal pelvis or ureter, it is likely to be of relatively low (O, A, or B) stage (Fig. 14.28). If the edges of the mass extend into the renal sinus fat (Fig. 14.29) or the renal parenchyma or if the periureteral tissues are infiltrated in the region of a collecting system tumor, it is likely to be stage C; local lymphadenopathy usually indicates stage D disease (Fig. 14.30).
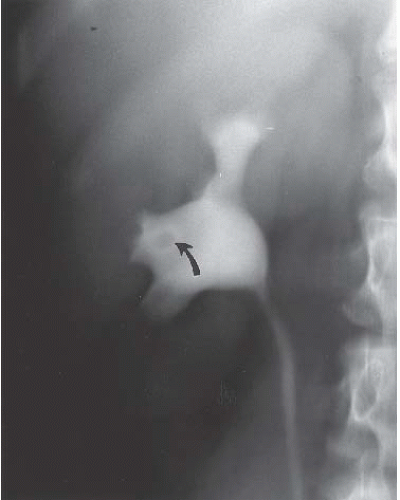 FIGURE 14.26. Normal papilla causing a lucent filling defect in the renal pelvis. Note the dense ring (arrow) caused by contrast in the papilla’s fornix. |
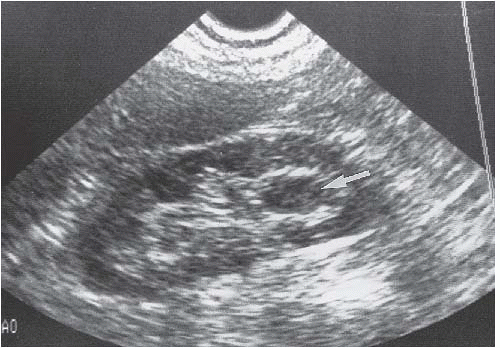 FIGURE 14.27. TCC. Ultrasound examination showing hypoechoic discrete mass (arrow) in the lower pole calyceal region. |
Renal TCC can usually be differentiated from renal cell carcinoma. The urothelial tumor may invade into and involve a large part of the kidney. However, the reniform shape of the kidney (bean) is usually preserved, whereas renal cell carcinoma typically presents as a round mass that often distorts the renal contour (ball). Occasionally, it may be difficult to differentiate the two, especially if a TCC invades the renal parenchyma focally (Fig. 14.31), or if renal cell carcinoma grows into the collecting system.
Ureteral tumors present as a single filling defect or as multiple intraluminal filling defects on CT. When the kidney is obstructed and the ureter fails to opacify, retrograde pyelography may be necessary. Solitary polypoid lesions frequently demonstrate slight dilation of the ureter around and distal to the tumor, as well as above it (Fig. 14.32); this sign helps to differentiate the lesion from benign filling defects such as calculi or blood clots, because these do not grow and progressively stretch the ureteral walls. The localized expansion of the ureter at the inferior margin of the lesion produces an appearance known as the champagne glass or goblet sign on retrograde ureterography (Fig. 14.32C). Intramural lesions may infiltrate the ureteral wall, so that the ureter becomes fixed; the lesion will demonstrate an irregular narrowing of the ureteral lumen with abrupt, nontapering superior and inferior margins. The lesion may be eccentric, or it may encircle the ureter, producing an apple-core appearance.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree