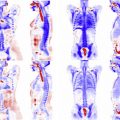
PET–magnetic resonance (MR) is a hybrid imaging modality that combines PET and MR. Evidence for this new modality is in the process of being developed, but both component modalities are well tested in the diagnosis and management of multiple myeloma. It allows advanced bimodality imaging of the whole body with an adaptable field of view and it can be used for monitoring plasma cell dyscrasias for progression to multiple myeloma, for assessing disease burden in patients with known multiple myeloma, for assessing response to therapy and relapse after remission, and for radiation therapy treatment planning.
Key points
- •
PET–magnetic resonance (MR) combines PET and MR and allows the integrated evaluation of multiple myeloma patients in 1 encounter.
- •
PET-MR can find areas of active disease in otherwise indolent-appearing plasma cell dyscrasias.
- •
PET-MR can assess both the current disease burden and changes in disease burden over time.
Introduction
Multiple myeloma is a rare cancer, with an estimated 30,330 new cases in 2016 and 12,650 deaths, representing 1.8% of all cancers in the United States. It is more common among men and people of African American descent, and portends a poor 5-year survival rate of 48%.
Multiple myeloma (MM) is the most advanced of the plasma cell dyscrasias, which are a set of white blood neoplasms characterized by monoclonal proliferation of plasma cells that typically lead to secretion of monoclonal myeloma proteins (M-proteins) in serum. M-proteins are composed of 2 heavy polypeptide chains of the same class (such as immunoglobulin [Ig] A or IgM) and 2 light polypeptide chains of the same type (such as kappa or lambda). Although they have been challenging to classify in a way that is biologically accurate and clinically useful, the current World Health Organization classification places them under mature B-cell neoplasms and subcategorizes them as monoclonal gammopathy of undetermined significance (MGUS), solitary plasmacytoma of bone (SPB), MM or plasma cell myeloma, extraosseous plasmacytoma, and monoclonal immunoglobulin depositional diseases.
MM is a disseminated malignancy of monoclonal plasma cells. Technically, to have MM, patients must have clonal bone marrow plasma cells of 10% or greater or an extramedullary plasmacytoma, plus at least 1 myeloma-defining event. These events are commonly referred to by the mnemonic CRAB, which stands for elevated serum calcium level (1 mg/dL above normal or over 11); renal clearance less than 40 mL/min or creatinine clearance greater than 2 mg/dL; anemia as manifested by hemoglobin level less than 100 g/L or greater than 20 g/L below the lower limit of normal; and bone lesions with 1 or more osteolytic lesions on bone survey, computed tomography (CT), or PET/CT. Not all increased monoclonal protein levels in serum or urine qualify as MM.
MM usually evolves from a previous MGUS or the so-called smoldering myeloma, both of which are asymptomatic. A patient with MGUS has increased monoclonal immunoglobulin level, but has serum monoclonal protein level less than 30 g/L, clonal bone marrow plasma cells less than 10%, urine monoclonal protein less than 500 mg/d, and no myeloma-defining events or amyloidosis. If IgG or IgA level is at least 30 g/L, urinary monoclonal protein is greater than 500 mg/d, and clonal bone marrow plasma cells are 10% to 60%, but there are no myeloma-defining events or amyloidosis, the patient instead has smoldering MM, an asymptomatic intermediate stage between MGUS and symptomatic MM that remains fairly stable over long periods of time. Such patients are usually followed.
Another forme fruste of MM is the solitary plasmacytoma of bone, which is a solitary bone lesion with no or minimal marrow involvement, otherwise normal MR or CT, and no other organ damage as previously described. SPB, which constitutes about 5% of all cases of plasma cell myeloma, is the proliferation of monoclonal plasma cells without the involvement of bone marrow. It is most commonly seen as a solitary lytic lesion in cancellous bone from proliferated plasma cells, and virtually never as a solitary extramedullary lesion. Ensuring that this is a solitary bone lesion is one of the main uses for imaging. Rarely, a plasmacytoma presents in extraosseous tissues; this is an extraosseous plasmacytoma.
Still another is Waldenström macroglobulinemia (WM), a rare disorder of lymphoplasmacytic lymphomatous infiltration of bone marrow with an IgM monoclonal gammopathy in the blood. Patients typically present with bulky lymphadenopathy, hepatosplenomegaly, neuropathy, hyperviscosity syndrome, bleeding disorders, and constitutional B symptoms.
At present, there are 2 clinically accepted systems of staging active MM. The Revised International Staging System (R-ISS) uses a combination of laboratory values, fluorescence in situ hybridization, and genetic markers, and describes no role for imaging ( Table 1 ).
| Stage | Features |
|---|---|
| I | Serum beta2-microglobulin level <3.5 mg/L, and Serum albumin level of 3.5 g/dL or greater, and No high-risk chromosomal abnormalities [del(17p) and/or t(4;14) and/or t(14;16)] and Normal LDH level (less than the upper limit of normal range) |
| II | Anyone not in stage I or III |
| III | Serum beta2-microglobulin level >5.5 mg/L), and High-risk chromosomal abnormalities (as defined above) or high LDH level |
The more recently introduced Durie-Salmon System Plus (DSS Plus) staging system ( Table 2 ) takes into consideration serum creatinine, hemoglobin, serum calcium, blood and urine immunoglobulins, as well as imaging, such as number of lytic lesions on skeletal radiography (the old Durie-Salmon system), which can be upstaged by the number of focal lesions on whole-body MR imaging or PET scan using F-18 fluorodeoxyglucose (FDG).
| Stage | Durie-Salmon | Upstaged Under DSS Plus if # of Lesions by MR or PET is |
|---|---|---|
| I | Hgb level >10 g/dL, and Serum Ca level <12 mg/dL, and Normal skeletal radiographs or solitary bone plasmacytoma, and IgG level <5 g/dL, IgA level <3 g/dL, and Urine light chain M component <4 g/d | 0–4 |
| II | Fits neither stage I nor III | 5–20 |
| III | Hgb level <8.5 g/dL, or Serum Ca level >12 mg/dL, or Advanced lytic bone lesions, or IgG level >7 g/dL, IgA level >5 g/dL), or Bence Jones protein >12 g/24 h | >20 |
| B | Cr level >2 mg/dL | Cr level >2 mg/dL and/or EMD on PET or MR imaging |
Introduction
Multiple myeloma is a rare cancer, with an estimated 30,330 new cases in 2016 and 12,650 deaths, representing 1.8% of all cancers in the United States. It is more common among men and people of African American descent, and portends a poor 5-year survival rate of 48%.
Multiple myeloma (MM) is the most advanced of the plasma cell dyscrasias, which are a set of white blood neoplasms characterized by monoclonal proliferation of plasma cells that typically lead to secretion of monoclonal myeloma proteins (M-proteins) in serum. M-proteins are composed of 2 heavy polypeptide chains of the same class (such as immunoglobulin [Ig] A or IgM) and 2 light polypeptide chains of the same type (such as kappa or lambda). Although they have been challenging to classify in a way that is biologically accurate and clinically useful, the current World Health Organization classification places them under mature B-cell neoplasms and subcategorizes them as monoclonal gammopathy of undetermined significance (MGUS), solitary plasmacytoma of bone (SPB), MM or plasma cell myeloma, extraosseous plasmacytoma, and monoclonal immunoglobulin depositional diseases.
MM is a disseminated malignancy of monoclonal plasma cells. Technically, to have MM, patients must have clonal bone marrow plasma cells of 10% or greater or an extramedullary plasmacytoma, plus at least 1 myeloma-defining event. These events are commonly referred to by the mnemonic CRAB, which stands for elevated serum calcium level (1 mg/dL above normal or over 11); renal clearance less than 40 mL/min or creatinine clearance greater than 2 mg/dL; anemia as manifested by hemoglobin level less than 100 g/L or greater than 20 g/L below the lower limit of normal; and bone lesions with 1 or more osteolytic lesions on bone survey, computed tomography (CT), or PET/CT. Not all increased monoclonal protein levels in serum or urine qualify as MM.
MM usually evolves from a previous MGUS or the so-called smoldering myeloma, both of which are asymptomatic. A patient with MGUS has increased monoclonal immunoglobulin level, but has serum monoclonal protein level less than 30 g/L, clonal bone marrow plasma cells less than 10%, urine monoclonal protein less than 500 mg/d, and no myeloma-defining events or amyloidosis. If IgG or IgA level is at least 30 g/L, urinary monoclonal protein is greater than 500 mg/d, and clonal bone marrow plasma cells are 10% to 60%, but there are no myeloma-defining events or amyloidosis, the patient instead has smoldering MM, an asymptomatic intermediate stage between MGUS and symptomatic MM that remains fairly stable over long periods of time. Such patients are usually followed.
Another forme fruste of MM is the solitary plasmacytoma of bone, which is a solitary bone lesion with no or minimal marrow involvement, otherwise normal MR or CT, and no other organ damage as previously described. SPB, which constitutes about 5% of all cases of plasma cell myeloma, is the proliferation of monoclonal plasma cells without the involvement of bone marrow. It is most commonly seen as a solitary lytic lesion in cancellous bone from proliferated plasma cells, and virtually never as a solitary extramedullary lesion. Ensuring that this is a solitary bone lesion is one of the main uses for imaging. Rarely, a plasmacytoma presents in extraosseous tissues; this is an extraosseous plasmacytoma.
Still another is Waldenström macroglobulinemia (WM), a rare disorder of lymphoplasmacytic lymphomatous infiltration of bone marrow with an IgM monoclonal gammopathy in the blood. Patients typically present with bulky lymphadenopathy, hepatosplenomegaly, neuropathy, hyperviscosity syndrome, bleeding disorders, and constitutional B symptoms.
At present, there are 2 clinically accepted systems of staging active MM. The Revised International Staging System (R-ISS) uses a combination of laboratory values, fluorescence in situ hybridization, and genetic markers, and describes no role for imaging ( Table 1 ).
| Stage | Features |
|---|---|
| I | Serum beta2-microglobulin level <3.5 mg/L, and Serum albumin level of 3.5 g/dL or greater, and No high-risk chromosomal abnormalities [del(17p) and/or t(4;14) and/or t(14;16)] and Normal LDH level (less than the upper limit of normal range) |
| II | Anyone not in stage I or III |
| III | Serum beta2-microglobulin level >5.5 mg/L), and High-risk chromosomal abnormalities (as defined above) or high LDH level |
The more recently introduced Durie-Salmon System Plus (DSS Plus) staging system ( Table 2 ) takes into consideration serum creatinine, hemoglobin, serum calcium, blood and urine immunoglobulins, as well as imaging, such as number of lytic lesions on skeletal radiography (the old Durie-Salmon system), which can be upstaged by the number of focal lesions on whole-body MR imaging or PET scan using F-18 fluorodeoxyglucose (FDG).
| Stage | Durie-Salmon | Upstaged Under DSS Plus if # of Lesions by MR or PET is |
|---|---|---|
| I | Hgb level >10 g/dL, and Serum Ca level <12 mg/dL, and Normal skeletal radiographs or solitary bone plasmacytoma, and IgG level <5 g/dL, IgA level <3 g/dL, and Urine light chain M component <4 g/d | 0–4 |
| II | Fits neither stage I nor III | 5–20 |
| III | Hgb level <8.5 g/dL, or Serum Ca level >12 mg/dL, or Advanced lytic bone lesions, or IgG level >7 g/dL, IgA level >5 g/dL), or Bence Jones protein >12 g/24 h | >20 |
| B | Cr level >2 mg/dL | Cr level >2 mg/dL and/or EMD on PET or MR imaging |
Imaging with fluorodeoxyglucose PET and magnetic resonance: current recommendations
Historically, a radiographic skeletal survey was used to assess plasma cell neoplasm. Over the last several decades, CT, FDG-PET/CT, and MR imaging have shown incremental value in the management of patients with MM, at baseline, surveillance, assessing therapy response, and often for surveillance and radiation therapy treatment planning. FDG-PET/MR is a new technique, and as such there are no recommendations by either the National Comprehensive Cancer Network (NCCN) or International Myeloma Working Group (IMWG), the two major groups producing guidelines in this area, for its use in staging or after therapy. However, the 2 underlying modalities, FDG-PET and MR, have been individually recommended for use in various settings. In general, both authorities recommend the use of MR, whereas only the NCCN recommends use of PET/CT. Many European practices use whole-body MR imaging, which is not routinely done in the United States.
Current NCCN recommendations recommend MR and PET/CT in several situations, usually as separate options for the same indication. MR and PET/CT are listed as options for following up smoldering myeloma, and for following up solitary extraosseous disease after radiation therapy. Whole-body MR or PET/CT is used to assess for bone disease and to discern active from smoldering myeloma in the case of a negative bone survey. Active MM is diagnosed when there is at least 1 focal lesion on MR greater than 5 mm, or 1 focal hypermetabolic osteolytic lesion on PET/CT along with clonal cells of at least 10% in the bone marrow. In addition, MR and PET/CT can be used to assess treatment response in active myeloma to either initial chemotherapy or stem cell transplant, as clinically indicated or when conventional imaging with radiographs or CT is inconclusive.
The IMWG, the main international myeloma group, recommends MR as part of the staging procedure for solitary bone plasmacytoma, and can upstage smoldering to symptomatic MM by finding at least 1 focal lesion larger than 5 mm. It also recommends MR in symptomatic MM as the gold standard for detection of bone marrow involvement (with prognostic information based on the pattern), for evaluating painful lesions for spinal cord compression, for differentiating benign from malignant vertebral fractures, and for accurate visualization of soft tissue extension and extramedullary plasmacytoma. PET is still not described as being useful for diagnosis or routine follow-up, although it can sometimes detect otherwise occult disease.
The American College Of Radiology does not have separate appropriateness criteria addressing MM, but it is mentioned as a potential cause for metastatic bone disease; specifically, in the case of patients with MM presenting with acute back pain, in which case lumbar spine MR is recommended after radiography.
Applications of PET and magnetic resonance imaging in plasma cell dyscrasias
There has been little academic work specifically on the use of PET-MR for MM; most studies and review articles have examined the 2 modalities separately. There is a recent study directly comparing standardized uptake values (SUVs) for PET/MR and PET/CT, showing that maximal SUV (SUV max ) values are lower on PET/MR but nonetheless tightly correlated with PET/CT (allowing the possibility that PET/MR can still be used with a conversion factor). As discussed previously, authorities are clear that MR is useful for staging, and differ on the role of PET. In most cases, evidence exists for the use of both modalities, with slight differences in sensitivity and specificity suggesting areas where synergy may be possible.
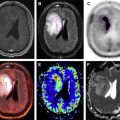
In general, positive MR for MM shows lesions with loss of signal on T1-weighted images, increased signal on T2-weighted images, and delayed contrast enhancement. The overall pattern may be described as normal, focal (focal lesions), diffuse (change of signal of the entire fatty bone marrow), or variegated/salt-and-pepper pattern (heterogeneous appearance of the entire bone marrow). A positive PET scan shows areas of increased uptake over and above the surrounding bone marrow (note that bone marrow has a low level of diffuse baseline uptake even when normal, and detecting diffuse involvement may be difficult). An example of a negative study is shown in Fig. 1 : PET activity is low and bone marrow signal is normal on all sequences, with no contrast enhancement when intravenous contrast is used.
Given that 2 modalities are being used, it is possible for one to be positive and the other to be negative. An example of this is shown in Fig. 2 : MR shows focal lesions, but they are not FDG avid. This finding is generally considered to be an example of indolent or quiescent disease. The converse is shown in Fig. 3 : the MR shows the usually low-risk variegated pattern, but the intense FDG uptake (together with the clinical picture) suggests recurrent disease.
Staging
Like MR, PET can detect marrow involvement (sensitivity 59%–85%, specificity 79%–92%) and extraosseous disease, usually presenting as an FDG-avid soft tissue mass. In terms of comparison with other modalities, PET has shown superiority compared with whole-body radiography for lesion assessment according to a recent meta-analysis, but the same meta-analysis suggests the same for MR (and CT). MR can also depict the spinal cord and nerve roots, which PET lacks the spatial resolution to do. Another study showed superior sensitivity and specificity of MR to PET (59% vs 68% and 75% vs 83%) for staging but also showed 100% specificity and positive predictive value when modalities were concordant, suggesting possible synergy between modalities. One study has shown superiority of PET to MR outside the spine and pelvis, whereas others have shown greater sensitivity of MR, particularly for the diffuse pattern of involvement. A study comparing PET/CT with MR in patients with smoldering MM and MGUS showed that PET upstaged more patients with smoldering MM than MR did, but few patients with smoldering MM had MR, so the result is uncertain. MR, specifically diffusion-weighted imaging (DWI), may be more sensitive than PET/CT for pretreated patients specifically.
However, PET/CT has known limitations. It may fail to detect smaller lytic skeletal lesions (less than about 10 mm) or diffuse spine involvement, and may produce false positives because of other causes of FDG uptake, such as other malignancies, inflammation or infection, steroid use, brown fat, or postsurgical changes (note that, in the case of other malignancies and even of inflammation or infection, although the uptake may not represent MM, it may still be clinically useful information). MR also occasionally produces false-positives at the site of bone marrow biopsy, atypical hemangioma, or bone infarction.
There has been interest in using tracers other than the standard FDG for staging. C-11 choline has shown better ability to detect diffuse bone marrow infiltration and may be more sensitive for myelomatous bone lesions in general. 18F-fluorocholine may be able to detect more lesions in suspected relapsed or progressing MM. 18F-fluoro -alpha-methyltyrosine (FAMT) has shown more uptake, and C-11 methionine has detected more lesions and shown more uptake, and correlates more strongly with biopsy-proven bone marrow involvement, than FDG. However, most of these remain experimental only, because they require either a cyclotron on site or an active radiopharmacy. F-18 NaF, which is US Food and Drug Administration (FDA) approved for bone metastases in other tumors, is relatively insensitive in MM, often detecting lytic lesions already visible on CT, although it may be useful to gauge bone formation after bortezomib chemotherapy.
Prognosis
PET has been shown to predict prognosis, in MGUS, in smoldering myeloma, and in active myeloma, with more than 3 to 4 FDG-avid lesions, as well as an SUV max of more than 4.2 and the presence of extramedullary disease, indicating lower progression-free and/or overall survival ; a higher metabolic tumor volume is a risk indicator as well. Liver, lung, and muscle involvement in particular decreases survival. An SUV max more than 3.5 in a spinal lesion combined with diffuse or multifocal involvement on MR may suggest an impending fracture. Increased metabolic activity in fat may be a sign of progression from MGUS to MM.
MR has prognostic value as well, with diffuse marrow replacement (and contrast enhancement) having a worse prognosis than variegated or normal patterns and asymptomatic patients being more likely to progress if they have MR-visible lesions. Overall survival is also adversely affected by a large number of focal lesions. Delayed contrast enhancement also seems to reflect degree of pathologic bone marrow involvement, changes in microcirculation, and prognosis in MM and smoldering MM, so optimal prognostic ability may be obtained using both modalities.
Response to Therapy
Myelomatous bone lesions regress only rarely, even after complete remissions, so conventional CT and radiography are difficult to use for monitoring. Where imaging is thought to be necessary, MR and PET have been used as methods of monitoring response to disease. In general, MR lesions, if resolving, regain the appearance of surrounding bone, whereas PET lesions lack avidity above surrounding bone.
PET/CT has been used as a method of monitoring response to therapy. SUV max has been shown to correlate with plasma cell ratios on bone marrow biopsy as well as proportion of plasma cells expressing CD38/CD138 ; furthermore, 3 or fewer lesions after day 7 of induction therapy and suppression of lesions after induction as well as after completion of treatment are all good prognostic indicators. In patients with an SUV max greater than 4.2 after treatment, PET is particularly effective for detecting relapse in follow-up.
MR has also been used, with resolution of focal lesions as a good prognostic factor. Delayed contrast-enhanced imaging is under investigation as a method of monitoring response; although it does not seem to delineate additional lesions, it does correlate with vessel density and serum markers of disease activity, much like PET SUV. Some studies have used DWI as a metric as well, because it also correlates with plasma cell ratio, shows changes after therapy, and may be able to predict response, in some cases quantitatively. There has also been interested in using advanced quantitative metrics on diffusion-weighted images to assess response. Combining delayed contrast enhancement and DWI may be the most useful option. Arterial spin labeling, another MR technique, seemed to be effective in a small study of 10 patients. However, in the few studies directly comparing the 2 modalities, PET/CT seems to have the advantage here, identifying more patients with a good response to treatment and doing so more quickly ; in the same studies showing greater sensitivity for MR.
In addition, PET/CT may be useful for assessing disease activity after autologous stem cell transplant; sensitivity is lower than with MR at 55%, but a negative scan after transplant correlates with a longer survival ; this is also true for allogeneic stem cell transplant. The sensitivity of MR for remission is better at 64%, with the number of focal lesions also having prognostic significance. Dynamic contrast-enhanced imaging has been used here as well, with bone marrow enhancement after induction correlating with response to stem cell transplant. One of the few studies to compare MR and PET directly showed a clear sensitivity-specificity trade-off between the modalities, with PET showing a 50% sensitivity but an 86% specificity, whereas MR showed an 80% sensitivity but a 38% specificity for remission.
PET has been shown to change management in 49% of cases (more than any known tumor), but this does not mean management has been changed correctly.
In short, PET and MR show different levels of sensitivity and specificity for the same applications, opening the possibility of synergy using PET-MR.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree