Physiologic Basis of Inflammation Imaging
Achim H. Kaim
Gustav K. von Schulthess
Imaging of septic and sterile inflammation is frequently used in the management of these diseases, be it to decide on the best therapeutic approach or to monitor disease course under therapy. Inflammation can be acute or chronic, the former showing predominantly neutrophil granulocyte infiltrates, whereas in the latter macrophages predominate. Various imaging methods are available for inflammation imaging, most notably magnetic resonance imaging (MRI), which has been used widely because of its excellent soft tissue contrast and its sensitivity to tissue edema and hyperemia, which are hallmarks of acute infection. MRI is less specific in chronic inflammation. Of the various nuclear imaging methods, the most relevant specific methods involve the labeling of granulocytes. Drawbacks are again good suitability for imaging of the acute phase only and a lack of spatial resolution and anatomic detail. The development of combined gamma camera–computed tomography (CT)—that is, single-photon emission computed tomography (SPECT)–CT systems—allows digital image coregistration of anatomic maps with conventional nuclear medicine studies.
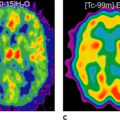
Fluorodeoxyglucose (FDG) positron emission tomography (PET) promises to overcome some of the shortcomings of the other methods, and this is owing to the so-called metabolic burst reaction, which granulocytes and macrophages undergo when they are fighting inflammation. This makes FDG-PET potentially useful for acute and chronic inflammation imaging. Furthermore, FDG-PET labels only activated granulocytes, and thus hematopoietic marrow contains little label, making the axial skeleton also accessible to inflammation imaging with PET. The spatial resolution of PET is substantially higher than that of conventional methods. The introduction of anatomic-metabolic imaging with PET-CT has been a great step forward, although the literature is still too limited to make definitive statements on the usefulness of this method in inflammation imaging. Although FDG-PET seems to be very useful in inflammation imaging in general, the logistics of the method make its use easier in chronic than in acute inflammation.
Pathophysiology
Inflammation is described as being acute or chronic depending on its duration. These two phases of an inflammatory process are microscopically characterized by different tissue changes. In acute inflammation, an early vascular response with vasodilatation is found, and exudation of plasma proteins from the peripheral vascular bed owing to damage of the endothelium occurs, which in turn leads to accumulation of edema fluid within the first 10 to 15 minutes. Moreover, increased endothelial permeability allows leukocytes to escape from the blood into tissue parenchyma. Early, these are predominantly neutrophil granulocytes, although a few eosinophils and monocytes emigrate as well. Neutrophils are particularly attracted by bacteria and dying tissue. As acute inflammation subsides after a few days, the number of cells escaping from the blood stream decreases rapidly and the injured tissue returns to normal. Acute infection inflammation does not always resolve, but inflammation may remain active for weeks or months, thereby now becoming chronic inflammation. The clinical character of the inflammation changes
with a decrease of symptoms and episodic fever. In the tissue, the vascular congestion and the interstitial edema are less prominent. The neutrophils in the exudates die or escape back into the blood and are replaced by lymphocytes and macrophages. The inflamed tissue becomes increasingly scarred and fibrotic (1,2).
with a decrease of symptoms and episodic fever. In the tissue, the vascular congestion and the interstitial edema are less prominent. The neutrophils in the exudates die or escape back into the blood and are replaced by lymphocytes and macrophages. The inflamed tissue becomes increasingly scarred and fibrotic (1,2).
Inflammation may be septic or aseptic and most frequently involves the musculoskeletal system, the cardiovascular organs, and the lungs, but any organ may be affected. The histologic alterations that occur in the first few hours and days of the inflammatory process change with time, and later more chronic stages of inflammation are considerably different. Since current imaging modalities reflect certain morphologic and functional changes, this dynamic development of an inflammatory process with time has to be kept in mind when interpreting imaging studies.
Diagnosis
In most cases, the diagnosis of an inflammatory disorder is a clinical one. The early diagnosis of acute infection is based on signs and symptoms of inflammation (rubor, calor, dolor, edema, functio laesa, raised body temperature, increased white blood cell count [WBC], and C-reactive protein [CRP]). Suspected disorders on certain anatomic sites such as the great vessels or visceral organs cannot be exclusively confirmed by clinical examination and warrant further radiologic workup. Generally, the aim of imaging is to confirm the clinical suspicion, define the grade and extent of inflammation, and detect complications such as abscess or fistula formation in infection.
In cases of chronic inflammatory disorders, the generalized signs of inflammation are frequently masked: the body temperature is normal or only slightly elevated, and hematologic testing (WBC, erythrocyte sedimentation rate [ESR], C-reactive protein [CRP]) is highly variable. Imaging represents a particular challenge in these patients, since subsequent treatment is strongly influenced by the imaging results. The main questions, which have to be answered from a clinical point of view, are as follows:
Presence of active inflammation versus aseptic noninflammatory changes, for example, granulation tissue
Extension of the inflammation
Regional anatomy
Development of local complications (abscess, sinus tracts, necrotic tissue)
Intensity of inflammation prior/during anti-inflammatory medication (therapy monitoring)
Imaging of inflammation can be accomplished with various imaging techniques. Computed tomography (CT) and magnetic resonance imaging (MRI) are most frequently used and provide anatomic maps with high spatial resolution. A certain degree of functional information can also be obtained by the use of standard x-ray or MR contrast media. MRI in particular has been recognized as a very useful modality for the detection of inflammatory disorders because of its capability to demonstrate anatomic details and pathologic changes of soft tissues and bone marrow with high spatial resolution and contrast. The common criteria used for delineation of inflammatory changes on MRI include the presence of an edema pattern on T2-weighted images and contrast enhancement after intravenous gadolinium application on T1-weighted images. Thus, MRI visualizes accumulation of interstitial fluid, hyperemia, and increased vascular permeability owing to the endothelial damage in inflammation. Vascularized granulation tissue in chronic inflammatory changes exhibits a similar signal and contrast behavior on MRI and cannot be reliably differentiated from noninflammatory aseptic changes after surgical or conservative treatment of any underlying disease (3,4,5).
Conventional Nuclear Medicine Imaging
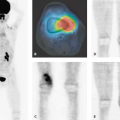
Although radiologic techniques provide excellent structural spatial resolution, nuclear medicine procedures can assess the degree of disease activity based on physiologic and metabolic changes caused by underlying pathologies. Radionuclide imaging has proven its usefulness in the evaluation of patients suspected of harboring an infection, although the role varies with the individual and specific setting. The development of integrated single-photon emission computed tomography (SPECT)–computed tomography (CT) cameras has overcome to a certain extent the lack of spatial resolution in conventional nuclear medicine and allows detailed correlation of functional-metabolic information with high-resolution anatomic maps in CT by immediate digital image coregistration and fusion (6). By combining a dual-head gamma camera with low-end and increasingly mid- to high-end CT systems, sequential acquisition of SPECT and CT data has become possible in one session without moving the patient. Inflammation scintigraphy is carried out with radiopharmaceuticals, which all label one of the consecutive steps of the host response to the injury. Defense mechanisms include physical barriers against the penetration of germs, release of cytokines, regulation of adhesion molecules, activation of the complement cascade, and infiltration with inflammatory cells, most notably neutrophil granulocytes, lymphocytes, and macrophages, which undergo a so-called white cell burst reaction when actively fighting inflammation. The choice of the best radiopharmaceutical depends on the grade of inflammation, duration of infection, availability, cost, and radiation exposure. Currently used radiopharmaceuticals comprise the following:
99mTc-radiolabeled nanocolloids and human immunoglobulins (HIG)
67Ga-citrate
111In and 99mTc-HMPAO-labeled leukocytes
99mTc-radiolabeled murine monoclonal antigranulocyte antibodies (MAB)
18F-deoxyglucose (FDG) for positron emission tomography (PET)
Further comments on some properties and preparation of these radiopharmaceuticals are given in Chapter 18.
99mTc-Radiolabeled Nanocolloids and Human Immunoglobulins (HIG)
Nanocolloids (NC) are radiolabeled with 99mTc. This radiopharmaceutical has a favorable radiation dosimetry, high availability, and acceptable image resolution. Examination time is short (60 minutes), and the substance is easy to handle because neither time-consuming preparation, nor special facilities, nor technical expertise are needed. The mechanism of uptake in infection is the detection of increased vascular permeability with an interstitial extracellular distribution of the nanomolecules. Moreover, since nanocolloids are phagocytized by the reticuloendothelial system (RES), phagocytosis by local and activated macrophages may also contribute to local uptake. Phagocytosis of the reticuloendothelial cells leads to a physiologic uptake in liver, spleen, and bone marrow. The usefulness of NC in suspected acute infection of the extremities is well proven; however, clinical experience documents a limited value in the body trunk and a relatively poor specificity with false-positive results in chronic inflammation (7), which requires a highly specific second examination.
HIG are also labeled with 99mTc and characterized by good availability, simple labeling, and a single in vivo injection. The increased permeability of vascular endothelium mainly contributes to HIG accumulation in the interstitial space, resulting in unspecific uptake in chronic inflammation.
67Ga-Citrate
67Ga-citrate has been used in infection imaging for more than 30 years. Several factors contribute to the uptake of this tracer in infection (8). About 90% of circulating 67Ga is in the plasma and bound to transferrin. Increased blood flow and increased vascular membrane permeability result in increased delivery and accumulation at inflammatory foci. 67Ga also binds to lactoferrin, which is present in high concentrations in inflammation. Other mechanisms are uptake into leukocytic lysosomes and endoplasmatic reticulum and direct accumulation by certain bacteria. The normal biodistribution of 67Ga, which can be variable, includes bone, bone marrow, liver, gastrointestinal and genitourinary tracts, and soft tissues. It has been widely replaced by other radiopharmaceuticals because of the unfavorable physical characteristics (high radiation exposure, long examination time, reduced availability, and high energy gamma rays with subsequent low spatial resolution).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree