Normal Anatomy
The main pulmonary artery arises from the right ventricle at the level of the pulmonic valve and travels approximately 5 cm before bifurcating into the left and right pulmonary arteries. The right pulmonary artery passes anterior to the right main bronchus and divides into the truncus anterior, which extends into the right upper lobe of the lung, and the interlobar branch, which divides into segmental arteries extending into the right middle and right lower lobes. A branch of the interlobar artery supplies the posterior segment of the right upper lobe in approximately 90% of patients. The left pulmonary artery is typically a shorter vessel and passes cephalad to the left main bronchus; it continues as an interlobar artery and extends into the left upper and lower lobes of the lung through segmental branches. A separate branch supplying the left upper lobe may arise from the left pulmonary artery before continuing as the interlobar artery.
Comparisons of pulmonary arterial size in physiologically normal patients and in those with pulmonary arterial hypertension (PAH) resulted in suggested measurements for the upper limits of normal vessel diameters. These values are 28.6 mm for the main pulmonary artery, 28 mm for the left pulmonary artery, and 24.3 mm for the right pulmonary artery. Distal to the origin of the upper lobe bronchi, the ratio of the sizes of the pulmonary artery and adjacent bronchus is approximately 1.3:1 to 1.4:1. This ratio approaches 1:1 in the periphery. Pulmonary arterial branching is dichotomous, with approximately 17 separate divisions from the bifurcation of the main pulmonary artery. Three main types of pulmonary arteries have been described: elastic, muscular, and transitional. Elastic arteries measure greater than 0.5 mm and are composed of internal and external elastic laminae around central circular muscle and connective tissue. These vessels receive the blood ejected from the right ventricle. Muscular arteries measure less than 0.5 mm and are composed of fewer internal and external elastic laminae around more substantial central circular muscle and connective tissue. These vessels course with the peripheral airways to the level of the terminal bronchioles. Transitional arteries are characterized by composition between the elastic and muscular types. Pulmonary arterioles typically measure less than 0.15 mm, but arterioles as small as 300 μm may be seen in the periphery on high-resolution computed tomography (HRCT).
Acute Pulmonary Embolism
Pathogenesis
Acute pulmonary embolism (PE), one of the most common causes of cardiovascular morbidity and mortality, ranks behind only myocardial infarction and stroke in prevalence. Approximately 300,000 patients die of PE each year. Because patients with PE may be asymptomatic, the diagnosis often goes undetected and may be made only at autopsy. The most common presenting clinical symptoms include dyspnea, pleuritic chest pain, tachypnea, and tachycardia. Only one half of the patients with PE present with one component of the classic triad of chest pain, dyspnea, and hemoptysis.
PEs are typically the result of dislodged thrombus that originally formed within the deep veins of the calf. Approximately 79% of patients with PE have evidence of thrombus formation, and PE occurs in approximately 50% of patients with deep venous thrombosis (DVT).
Common clinical manifestations of DVT include leg pain, warmth, and swelling. Acute medical illness may be the most common clinical situation in which PE is encountered. Surgical procedures for hip fracture, knee and hip replacement, and cancer, as well as trauma and spinal cord injury, are associated with a high risk of PE. Other risk factors include prolonged immobilization, advanced age, cerebrovascular accident, pregnancy, and genetic or acquired thrombophilia. PE involving the lower lobes of the lung has a higher incidence.
The D-dimer test measures the levels of a specific derivative of cross-linked fibrin and is commonly used to evaluate the probability of DVT and PE. However, this test is nonspecific, and elevated levels may be encountered in trauma, inflammatory states, infection, malignancy, and pregnancy, as well as during the postoperative period. The clinical probability of PE can be assessed with one of several clinical prediction scores, the most commonly used of which are the Canadian (Wells) prediction score and the Revised Geneva score. Imaging studies are not indicated in patients with low to moderate pretest probability because the pretest probability of DVT and PE is low. However, further evaluation with imaging is recommended if the pretest probability is high.
The strongest prognostic implications for short-term mortality are derived from the hemodynamic status of the patient at the time of presentation. The mortality rate in normotensive patients without evidence of right ventricular dysfunction at the time of presentation is approximately 2%. The short-term mortality rate of massive PE, characterized by arterial hypotension or shock, ranges from 15% to 30% and increases to 65% in patients presenting with cardiac arrest.
Imaging Evaluation
Chest Radiography
The chest radiograph is often the first imaging examination performed in the evaluation of a patient with suspected PE. Although no specific abnormalities may be detected on the chest radiograph in a patient with PE, other potential causes of symptoms simulating PE may be detected. These include abnormalities such as pneumonia, pulmonary edema, and pneumothorax.
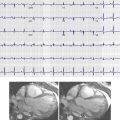
Many classic signs of PE on chest radiography have been described, but they are infrequently encountered. These include the Westermark sign, which is characterized by lucency within all or a portion of a lung and represents oligemia secondary to vasoconstriction distal to the site of PE ( Fig. 42-1 ). Pulmonary infarction, which is encountered approximately 10% to 15% of the time, may appear as a peripheral consolidation adjacent to the pleura that is also known as the Hampton hump ( Fig. 42-2 ). Additional imaging manifestations of infarction include atelectasis, pleural effusion, and diaphragmatic elevation. Focal enlargement and abrupt tapering of the central pulmonary artery comprise the Fleischner sign. However, these imaging findings are nonspecific for the diagnosis of PE, and additional imaging is usually required.


Ventilation-Perfusion Scintigraphy
Ventilation-perfusion (V/Q) scintigraphy is most valuable in the absence of cardiopulmonary disease and in the setting of a normal chest radiograph. The examination involves the intravenous injection of technetium-99m–macroaggregated human albumin (MAA) and the inhalation of either xenon-133 or aerosolized technetium-99m diethylenetriamine pentaacetate. PE is suggested by the appearance of a V/Q mismatch. However, other abnormalities such as lung cancer, vasculitis, fat emboli, and PAH may result in similar imaging characteristics.
The Prospective Investigation of Pulmonary Embolism II (PIOPED II) interpretation scheme is used in the evaluation of the V/Q scan. The diagnostic categories include normal, very low probability, low probability, intermediate probability, and high probability. This technique has a sensitivity of 98%, but a specificity of 10%, for the diagnosis of PE. A normal perfusion scan result virtually excludes the possibility of PE, whereas a high-probability scan is considered diagnostic of PE. However, most patients undergoing V/Q scanning have results that lie between these two categories and usually require an additional imaging test for definitive diagnosis.
Pulmonary Angiography
Once considered the gold standard for diagnosing PE, pulmonary angiography has largely been replaced by contrast-enhanced multidetector computed tomography (MDCT) at most institutions. The procedure involves percutaneously accessing the femoral vein and extending a catheter through the right atrium and ventricle into the pulmonary arteries. After the injection of intravenous contrast material, fluoroscopic images are obtained. The most common imaging manifestation of PE is a filling defect. Other findings include abrupt vessel cutoff and regions of oligemia with pruning of vascular branches.
Computed Tomography
Technique
MDCT has emerged as the gold standard for the detection of PE in many institutions and has supplanted pulmonary angiography and V/Q scintigraphy. Contrast-enhanced MDCT has the highest sensitivity and specificity for the detection of PE in the main, lobar, and segmental pulmonary arteries, with sensitivities ranging from 55% to 100% and specificities ranging from 83% to 100%. Other primary advantages of MDCT include its widespread availability, fast scan times, noninvasiveness and the ability to visualize the subsegmental pulmonary arteries. Additionally, CT can be used to evaluate the remainder of the thorax for possible alternative diagnoses resulting in symptoms mimicking PE such as pneumonia, congestive heart failure, and pneumothorax, as well as to evaluate the lungs and mediastinum for nodules and masses.
Careful selection of scan protocol and timing of contrast material administration are the most important factors in obtaining high-quality CT images through the thorax. Modern MDCT scanners are able to image the entire thorax in a single breath hold, thus resulting in shorter acquisition times. Shorter acquisition times lead to maximal spatial and temporal resolution and decreased artifacts secondary to respiratory and cardiac motion. The shortest rotation time and the highest pitch are recommended to maximize resolution. Dual source MDCT scanners, in which two tubes and detector configurations are present, have been shown in studies to optimize temporal resolution and offer higher pitches than those available on standard MDCT scanners. The amount and rate of iodinated contrast material injected are greater than those administered for routine CT scanning of the thorax, although the details vary among institutions. Optimal timing of the scan acquisition following contrast injection, the most important factor in obtaining adequate opacification of the pulmonary arterial system, may be achieved through either bolus tracking technology or the use of a test injection. Following image acquisition, multiplanar reformations may be constructed with modern MDCT scanners to optimize visualization of the pulmonary arterial system.
Findings
Central hypodense filling defects within opacified pulmonary arterial branches are the most common imaging manifestations of PE. Sharp interfaces between the filling defects and the contrast material and acute angles between the filling defects and the vessel wall are usually present. Serpiginous filling defects within pulmonary arteries may result in a tram-track appearance, and occluded branches may dilate. Emboli bridging the main pulmonary arteries, known as saddle emboli, may be seen. Less specific findings include complete arterial occlusion and abrupt cutoff of vessels, which may be seen in either acute or chronic PE.
The most common imaging manifestations within the lung parenchyma are segmental and subsegmental atelectasis, which may obscure visualization of the adjacent lung parenchyma and the presence of PE within pulmonary arterial branches. Pulmonary infarction, reported to occur in 10% to 15% of cases, manifests as wedge-shaped opacities of solid, ground glass, or mixed density abutting the pleural surface in the lung periphery. Although insensitive, this finding is the most specific sign of acute PE. Pleural effusions, which may be transudative or hemorrhagic, have been reported to occur in approximately 30% of cases.
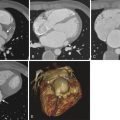
The term massive PE is used to characterize the association of right ventricular dysfunction and systemic hypotension with acute PE. These patients have been shown to benefit from additional systemic thrombolysis. Right ventricular dysfunction is manifested on contrast-enhanced MDCT as cardiac strain, the most specific feature of which is dilation of the right-sided cardiac chambers as compared with the left-sided cardiac chambers ( Fig. 42-3 ). This measurement is made at the point of maximal diameter, typically near the base of the heart at the level of the atrioventricular valves. Straightening of the anterior interventricular septum, bowing of the interatrial septum toward the left atrium, and right atrial dilation may also be seen. A less specific sign of right-sided cardiac strain is enlargement of the pulmonary arteries, most commonly the main pulmonary artery, to more than 3.5 cm. Dilation between 3.0 and 3.5 cm also suggests right-sided cardiac strain if the pulmonary artery is larger than the ascending aorta at the same level. Additional findings may include reflux of contrast into the inferior vena cava and diminished pulmonary vascularity.

Magnetic Resonance Imaging
Magnetic resonance imaging (MRI) provides an alternative way to evaluate for the presence of PE, but it is less commonly used than MDCT because of longer scan times and increased cost. Additionally, sensitivity for the detection of PE has been demonstrated only within the main, lobar, and segmental pulmonary arteries. Studies comparing the use of contrast-enhanced MDCT and multitechnique MRI have been performed. One of these studies, employing real-time MRI, MRI perfusion imaging, and contrast-enhanced magnetic resonance angiography (MRA) separately and in combination, demonstrated sensitivities and specificities ranging from 77% to 100% and 91% to 100%, respectively.
The Prospective Investigation of Pulmonary Embolism III (PIOPED III) trial evaluated the efficacy of using gadolinium-enhanced MRA to diagnose acute PE. Adequate studies were defined as those demonstrating adequate opacification of the pulmonary arteries to the subsegmental level. However, only 25% of the examinations performed were deemed adequate for diagnostic purposes averaged across all participating institutions. Technically inadequate examinations correctly identified PE in 57% of cases. Technically adequate examinations demonstrated a sensitivity and specificity of 78% and 99%, respectively. The combination of technically adequate contrast-enhanced MRA and magnetic resonance venography (MRV) had a sensitivity of 92%, higher than that of MRA alone, and a specificity of 96%, slightly lower than that of MRA alone. However, 52% of patients who had MRA and MRV had inadequate examinations. The investigators of PIOPED III concluded that MRA should be used for the evaluation of PE only if patients have contraindications to other tests, such as contrast allergy in the case of contrast-enhanced MDCT, and in those institutions that routinely perform the test and perform it adequately.
Chronic Pulmonary Embolism
Approximately 90% of PEs resolve fully without sequelae. In the remainder of cases, however, PEs may organize, retract, and recanalize. One of the most common imaging manifestations of chronic PE on contrast-enhanced MDCT is complete occlusion of a pulmonary arterial branch, which is usually smaller than the adjacent branches. In contrast to acute PE, the filling defects of chronic PE are eccentrically located within the pulmonary arteries and demonstrate obtuse angles with respect to the vessel wall ( Fig. 42-4 ). Thrombus may become incorporated into the vessel wall, and intraluminal webs and flaps may be visualized. Recanalized pulmonary arterial branches demonstrate contrast opacification of a smaller lumen when compared with the adjacent vessels, and the vessel walls may be thickened. Similar findings may be seen on MRI, although the limited spatial resolution of MRI may impair identification of residual disease if thrombus is incorporated into the wall of the vessel. Manifestations of chronic PE on MDCT, apart from those involving the pulmonary arteries, include the formation of prominent collateral vessels, usually of bronchial origin, and mosaic perfusion within the lung parenchyma (see Fig. 42-4 ). Although most pulmonary infarctions resolve completely over the course of several weeks, patchy opacities representing scar may persist at the site of infarction.

Chronic PE may result in the development of PAH, which may manifest clinically as dyspnea and fatigue. PAH may be suggested by enlargement of the main, left, and right pulmonary arteries on chest radiography. Additionally, MDCT may demonstrate dilation of the main pulmonary artery to more than 3 cm (see Fig. 42-4 ).
Pulmonary Arterial Hypertension
Pathogenesis
PAH is an abnormality characterized by elevated pulmonary vascular resistance and pulmonary artery pressure. It is defined as mean pulmonary artery pressure greater than 25 mm Hg at rest or greater than 30 mm Hg during exercise. Mean pulmonary capillary wedge pressure and left ventricular end-diastolic pressure are typically less than 15 mm Hg. The World Health Organization has classified PAH into five separate groups on the basis of mechanism. PAH has been designated as group 1 in the classification scheme and comprises idiopathic PAH (IPAH); PAH in the setting of collagen vascular disease, portal hypertension, congenital left-to-right cardiac shunts, and infection with the human immunodeficiency virus (HIV); and persistent PAH of the newborn. Group 2 includes pulmonary hypertension (PH) with left heart disease. Group 3 comprises PH secondary to lung diseases such as chronic obstructive pulmonary disease (COPD) and interstitial lung disease. Group 4 includes PH due to pulmonary emboli or other blood clotting disorders. Group 5 comprises PH secondary to blood disorders such as polycythemia vera, systemic diseases such as sarcoidosis and vasculitides, and metabolic disorders.
Another common scheme used to categorize the causes of PAH is to partition them into precapillary and postcapillary etiologic groups. In IPAH, the cause of PAH is unknown. The most common clinical presentation is dyspnea on exertion. Other presenting symptoms include fatigue, syncope, and chest pain. Women are affected more often than are men. Sporadic and familial forms of IPAH have been described. The familial form of IPAH is responsible for approximately 10% of cases and is inherited in an autosomal dominant fashion. Plexogenic pulmonary arteriopathy is the term given to describe the pathologic abnormalities involving the pulmonary arteries in IPAH, and it consists of medial hypertrophy, intimal proliferation and fibrosis, and necrotizing arteritis.
Most cases of PAH, however, are secondary to a known cause. Common precapillary causes of PAH include chronic thromboembolic disease, congenital heart diseases resulting in left-to-right shunts such as patent ductus arteriosus and atrial and ventricular septal defects, hepatic disease, infection with HIV, and drugs and toxins. Abnormalities of the lung parenchyma such as emphysema and interstitial lung disease are additional precapillary causes of PAH. Common postcapillary causes of PAH include left-sided cardiac failure, mitral stenosis, fibrosing mediastinitis, and pulmonary veno-occlusive disease. The diagnosis and assessment of the severity of PAH are most reliably made with right-sided heart catheterization, in which right-sided pressures may be directly measured. However, several noninvasive modalities are often used to screen for PAH. They can provide estimates of pulmonary arterial pressures at rest and at exercise, and they can demonstrate secondary imaging findings suggestive of the diagnosis. Echocardiographic findings that are consistent with right ventricular pressure overload include enlargement of the right ventricle and atrium, right ventricular hypertrophy, and global right ventricular dysfunction. Systolic flattening and increased thickness of the interventricular septum, as well as an abnormally increased ratio of the interventricular septum to the posterior wall of the left ventricle greater than 1, may be present.
The radiologic manifestations of IPAH are often indistinguishable from those of secondary PAH. The most common finding on chest radiography is enlargement of the main pulmonary artery, which appears convex. The left and right pulmonary arteries may also be enlarged, as well as the right atrium and right ventricle. Evaluation of the pulmonary arteries and right-sided cardiac chambers is more accurately performed with MDCT, and dilation of the main pulmonary artery to more than 3 cm suggests PAH ( Fig. 42-5 ). Pruning of the peripheral pulmonary arteries is usually present. Electrocardiogram-gated CT angiography has demonstrated substantially decreased distensibility of the pulmonary arterial wall, defined as the change in cross-sectional area between systole and diastole, in PAH. In chronic PAH, atherosclerotic calcifications may develop within the peripheral aspect of the central pulmonary arteries ( Fig. 42-6 ). Such calcifications are most commonly seen in patients with PAH secondary to Eisenmenger syndrome, in which a long-standing left-to-right shunt is reversed.



Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree