Background
Despite major advances in other imaging modalities, invasive cardiac catheterization remains a critical component of cardiovascular care. More than 1.1 million cardiac catheterization procedures, in addition to approximately 1.3 million percutaneous coronary interventions (PCIs), were performed in the United States in 2006. Tremendous advances in digital imaging and procedural equipment have occurred since the last American College of Cardiology/American Heart Association (ACC/AHA) guidelines were published in 1999. This chapter aims to provide the reader with an overview of the practical fundamentals of performing invasive coronary angiography.
Indications for Performing Coronary Angiography
Selective catheterization of the left coronary artery and the right coronary artery (RCA), combined with intraarterial injection of radiopaque contrast media, allows radiographic imaging of the coronary arterial system. A complete assessment of coronary artery bypass grafts (arterial and venous) also entails selective cannulation and contrast media injection. Critical information concerning the presence (and burden) or absence of obstructive coronary artery disease (CAD) may be obtained from invasive coronary angiography. However, coronary angiography is a luminogram, which shows only the contrast-filled arterial lumen and densely calcified plaque. Noncalcified or lightly calcified nonobstructive atherosclerotic plaque may remain undetected in the absence of luminal narrowing. Although coronary angiography has undoubtedly had an impact on many aspects of cardiovascular care, the widespread availability of this imaging technique has perhaps influenced the management of patients with acute coronary syndrome the most. For example, primary PCI is the superior treatment modality for patients with acute ST-segment elevation myocardial infarction.
Detailed guidelines concerning indications for coronary angiography are summarized in Box 2-1 . This list is not meant to be exhaustive and serves merely to highlight some of the most common clinical indications for invasive coronary angiography. The reader is directed to the ACC/AHA guidelines for coronary angiography for further information. As can be seen from these comprehensive guidelines, risk stratification plays an important role in assessing patient suitability for invasive coronary angiography. A study of patients without known CAD who underwent coronary angiography from January 2004 through April 2008 examined almost 400,000 patients enrolled in the ACC National Cardiovascular Data Registry. The investigators defined obstructive CAD as stenosis of 50% or more of the diameter of the left main coronary artery (LMCA) or stenosis of 70% or more of the diameter of a major epicardial artery. No CAD (defined as <20% stenosis in all vessels) was found in 39.2% of all patients. Among those patients with a positive result on noninvasive testing, the number was 41%. The investigators concluded that better risk stratification is required in clinical practice to ensure a higher diagnostic yield.
Patients With Known or Suspected Coronary Artery Disease Who Are Symptomatic or Have Stable Angina
Class 1
CCS class III and IV angina in patients receiving medical therapy
Presence of high-risk criteria on noninvasive testing
After cardiac arrest or in patients with sustained monomorphic VT or nonsustained polymorphic VT
Class IIa
CCS class III or IV angina that improves with medical therapy
Progressive worsening abnormalities on serial noninvasive testing
Patients with angina who are unsuitable for noninvasive testing
CCS I or II unresponsive to or in patients intolerant of medical therapy
Certain occupations with abnormal but not high-risk stress tests or those deemed high risk
Class IIb
CCS class I or II angina with known ischemia in the absence of high-risk criteria on noninvasive testing
Asymptomatic man or postmenopausal woman with two or more major clinical risk factors and an abnormal stress test without high-risk criteria without known CAD
Asymptomatic patients with prior MI with normal left ventricle and ischemia in the absence of high-risk criteria
Follow-up evaluation after cardiac transplant
Non–cardiac transplantation workup in patients 40 years old or older
Class III
Angina in patients who refuse revascularization
Angina in patients unsuitable for coronary revascularization or in whom revascularization is unlikely to improve the quality or duration of life
Screening test for asymptomatic patients
Postrevascularization status without evidence of ischemia
Coronary calcification on noninvasive testing without criteria listed earlier
Indications in Patients With Nonspecific Chest Pain
Class I
High-risk findings on noninvasive testing
Class IIb
Recurrent admissions for chest pain with abnormal (but not high-risk) or equivocal noninvasive test findings
Class III
All other patients with nonspecific chest pain
Indications in Acute Coronary Syndrome
Class I
High or intermediate risk for adverse outcomes in patients with UA unresponsive to medical therapy
High risk with UA
High or intermediate risk with UA that stabilizes with medical therapy
Initially low-risk UA that has high-risk features on noninvasive testing
Suspected Prinzmetal variant angina
Class IIb
Low−short-term-risk UA without high risk on noninvasive testing
Class III
Recurrent chest discomfort suggestive of UA without objective signs of ischemia with a normal angiogram within 5 years
UA in patients not deemed suitable for revascularization for whom revascularization will not lead to improved quality or length of life
Postrevascularization Ischemia Indications
Class I
Suspected acute failure of coronary angioplasty or stenting after PCI
Recurrent angina or high-risk criteria on noninvasive testing within 9 months of percutaneous revascularization
Class IIa
Recurrent symptomatic ischemia within 12 months of CABG
High-risk criteria after CABG
Recurrent angina after revascularization refractory to medical therapy
Class IIb
Asymptomatic post-PCI patients with suspected restenosis but without noninvasive high-risk test features
Recurrent angina in absence of high-risk features on noninvasive testing more than 1 year after CABG
Asymptomatic post-CABG patients with deterioration in noninvasive test parameters in the absence of high-risk features
Class III
Post-CABG symptoms in patients not deemed suitable for repeat revascularization
Routine angiography in asymptomatic patients after PCI or surgery
Acute ST-Segment Elevation Myocardial infarction
Class I
ST-elevation MI in patients who are eligible for primary or rescue PCI
Patients with cardiogenic shock who are suitable for revascularization
Patients for surgical repair of ventricular septal rupture or severe mitral regurgitation
Ongoing hemodynamic or electrical instability
Class III
Coronary angiography not indicated in patients deemed unsuitable for revascularization (e.g., because of severe comorbidities)
Acute Coronary Syndrome Unstable Angina and Acute Non–ST-Segment Elevation Myocardial Infarction
Class I
Diagnostic angiography with intent to perform revascularization in UA and non–ST-segment elevation MI with refractory angina or hemodynamic or electrical instability
Patients directed toward an early invasive strategy
Class IIb
As part of an invasive strategy for patients with chronic kidney disease
Class III
Diagnostic angiography as part of an invasive strategy not indicated in patients deemed unsuitable for revascularization (e.g., because of severe comorbidities)
Patients with acute chest pain and a low likelihood of acute coronary syndrome
Patients who refuse an invasive strategy
Perioperative Evaluation for Noncardiac Surgery
Class I
Patients with known or suspected CAD
High-risk features on noninvasive testing
Angina refractory to medical therapy
UA
Equivocal noninvasive test results in high–clinical risk patients undergoing high-risk surgery
Class IIa
Multiple intermediate–clinical risk features and planned vascular surgery
Ischemia on noninvasive testing without high-risk features
Equivocal noninvasive test result in intermediate–clinical risk patient with planned high-risk noncardiac surgery
Urgent noncardiac surgery after recent acute MI
Class IIb
Perioperative MI
Stable CCS III or IV angina and planned low-risk surgery
Class III
Low-risk noncardiac surgery, in patients with known CAD without high-risk features on noninvasive testing
Asymptomatic patients after coronary revascularization and excellent exercise capacity (≥7 METS)
Patients deemed unsuitable for revascularization (e.g., because of severe comorbidities)
Patients With Valvular Heart Disease
Class I
Before valve surgery or balloon valvotomy in patients with chest discomfort or noninvasive ischemia, or both
Before valve surgery in patients with multiple risk factors for CAD
Infective endocarditis with evidence of coronary embolization
Class IIb
During LHC before atrioventricular valve or mitral valve surgery in patients without symptoms or noninvasive features of CAD
Class III
Before surgery for infective endocarditis in patients with no evidence of coronary embolization or risk factors for CAD
Asymptomatic patients without planned surgery
Before cardiac surgery when preoperative LHC is unnecessary and patients have neither risk factors for nor evidence of CAD
CABG, Coronary artery bypass grafting; CAD, coronary artery disease; CCS, Canadian Cardiovascular Society; LHC, left heart catheterization; MET, metabolic equivalent; MI, myocardial infarction; PCI, percutaneous coronary intervention; UA, unstable angina; VT, ventricular tachycardia.
Patient Evaluation before Angiography
Before performing diagnostic coronary angiography, the physician should be fully aware of the patient’s complete medical history and indications for proceeding to PCI if necessary. In particular, the physician must have a detailed discussion regarding the risk-to-benefit ratio of angiography with or without PCI before the patient receives sedatives. This discussion may not always be possible in the emergency setting, in which prompt revascularization with minimal delay is mandatory (e.g., ST-segment elevation myocardial infarction). Nevertheless, a focused history can elucidate key details such as allergies, risk of bleeding, and increased risk of potential adverse effects (e.g., contrast-induced nephropathy [CIN], arterial access difficulties), in addition to providing the opportunity to allay patient’s concerns and obtain informed consent. The patient should also be given a brief step-by step-account of what he or she can expect to experience in the cardiac catheterization laboratory.
Physical examination should include, at a minimum, detailed vital signs (e.g., blood pressure, heart rate) and evaluation of cardiovascular, respiratory, and peripheral arterial systems. The results of these evaluations will influence arterial access choice, level of sedation, and suitability to proceed with percutaneous revascularization if deemed necessary. Results of basic laboratory tests such as renal function, complete blood count, coagulation profile, and liver function parameters should be available and reviewed. Abnormalities may require case deferral, depending on the clinical setting. Box 2-2 shows contraindications to cardiac catheterization.
Coagulopathy
Anemia
Electrolyte abnormalities
Contrast allergy
Worsening renal dysfunction
Decompensated heart failure
Infection around planned arteriotomy site
Uncontrolled severe hypertension
Cardiac Catheterization
Coronary angiography is performed in a fully equipped specially designed catheterization laboratory (see Fig. 2-3 , A ). For minimally invasive cardiac procedures to be performed in a safe and efficient manner, a dedicated, fully trained multidisciplinary team including cardiac nurses, technicians, and an interventional cardiologist is required. Common femoral arterial sheath placement using the modified Seldinger technique remains the most frequently employed access route for coronary angiography in the United States. Despite a surge of interest in using radial artery access, ACC National Cardiovascular Data Registry data suggest that less than 2% of all cardiac catheterization procedures are performed using the radial approach. Safe arterial access before any cardiac catheterization procedure is of critical importance to the success of the entire diagnostic or subsequent PCI procedure. A thorough knowledge of this procedure and its potential complications ( Table 2-1 ) is mandatory for all operators. Femoral arterial access complications may lead to limb viability compromise and even death in severe cases.
| Diagnostic Cardiac Catheterization: Vascular Complications | 0.40% Overall Complication Rate |
|---|---|
| PCI | |
| Femoral access hematoma (>6 cm) Retroperitoneal hematoma Pseudoaneurysm Atriovenous fistula Infection | 5-23% 0.15-44% 0.5-6.3% 0.2-2.1% <0.1% |
What Is the Risk Associated With Arterial Access in My Patient?
Several patient- and procedure-specific features may be used to estimate an increased risk of vascular access complications in patients who subsequently require PCI ( Box 2-3 ). Patients undergoing vascular access for diagnostic angiography alone are deemed at low risk for complications (<1% complication rate). These procedures usually entail the use of smaller arterial sheaths, often with no systemic anticoagulation. Other characteristics of low risk include elective procedures, male sex, normal renal function, and short procedure duration. Patients at moderate risk (1% to 3%) of vascular access complications include those undergoing routine PCI. In addition, increasing age, female sex, renal impairment, use of larger arterial sheaths (6 to 7 Fr), anticoagulation, and antithrombotic treatments are associated with greater risk. High-risk (>3%) features are coexisting peripheral arterial disease (PAD), renal and coagulation abnormalities, increasing age and sheath size (≥8 Fr), and female sex. Furthermore, emergency and challenging procedures such as primary or multivessel PCI and complex prolonged procedures increases the vascular access complication profile. Procedures that require even larger arterial sheaths, such as balloon aortic valvuloplasty (≥10 Fr), may carry an access complication rate of 11% to 17%.
Advanced age
Female sex
Smaller body surface area
Congestive heart failure, coagulopathy, chronic obstructive pulmonary disease, renal impairment, liver dysfunction, immunosuppression, prior coronary artery bypass grafting
Urgent or emergency procedure
Multivessel coronary artery disease
Intraaortic balloon pump counterpulsation use
Antiplatelet therapy (e.g., antiglycoprotein IIb/IIIa agents)
Increased lesion complexity
Guide to Obtaining Arterial Access
The anatomic landmarks necessary for safe common femoral arterial access are illustrated in Figure 2-1 , A , and should be identified before puncture. The inguinal ligament usually lies inferior to the inguinal skin crease and is crossed by the femoral artery at the one third medial–two thirds lateral point. The relation of the inguinal skin crease to the midfemoral head is routinely assessed radiographically with artery forceps (see Fig. 2-1 , B ). This assessment is particularly important because considerable variation in distance may exist between the inguinal skin crease and the inferior border of the femoral head, especially in obese patients.

An example of equipment commonly used is shown in Figure 2-3 , B. After informing the patient of what to expect, the clinician administers 10 to 12 mL of 2% lidocaine subcutaneously, initially with a 25-gauge needle and then with a 22-gauge needle over the chosen skin puncture site. Care must be taken always to aspirate before injection, to avoid intraarterial or venous lidocaine administration. Once sufficient local anesthetic has been given, all remaining agent is discarded, to avoid mistaken intravascular administration at a later stage during the procedure. A small superficial nick using the tip of a number 11 scalpel is made; this nick can then be enlarged with blunt dissection using mosquito forceps. Care must be taken to avoid trauma to the femoral artery, especially in very thin patients.
Remembering the relation of the skin entry site to the inferior femoral head, the operator holds an 18-gauge needle to the skin, usually at a 30- to 45-degree angle, while the left index and middle fingers palpate the chosen arteriotomy site. The femoral pulse is usually approximately 2 cm inferior to the inguinal ligament. The needle is then advanced slowly toward the chosen arteriotomy site until resistance is met and the needle is felt to puncture the anterior wall. Brisk arterial flow should then be encountered. Care must be taken not to puncture the posterior wall of the artery because this may potentially lead to serious complications such as hematoma or retroperitoneal hemorrhage, especially if a subsequent PCI necessitating full anticoagulation is performed. Furthermore, bleeding from a back wall puncture may initially be unrecognized until hemodynamic compromise occurs.
Using his or her right hand, the operator advances a long 0.035-inch J-tipped wire through the needle while the left hand carefully fixes the needle in position within the artery. Use of a long 0.035-inch guidewire with a soft atraumatic tip, such as the Hi-Torque Versacore guidewire (Abbott Vascular, Abbott Park, Ill), is advisable, especially in patients with known or likely PAD. The wire should advance smoothly, without resistance. If any resistance is encountered, the wire should be withdrawn and spun while it is advanced slowly under fluoroscopic guidance, thus ensuring that the wire tip moves freely and is not subintimal. If this maneuver is not possible, the wire should be withdrawn, and pulsatile needle blood flow should be confirmed. If the needle blood flow is poor or absent, the operator may try carefully repositioning the needle by changing the angle of entry. If satisfactory blood flow cannot be achieved, the best approach is to withdraw the needle completely and maintain thorough hemostasis with firm manual compression for 5 to 6 minutes or until oozing ceases.
Once the first 5 to 10 cm of the guidewire is advanced, then the operator continues to advance the wire under fluoroscopic guidance to the descending thoracic aorta. The needle is removed, and pressure is applied with the left hand to maintain hemostasis over the arteriotomy site, while the guidewire is cleaned with a sterile, nonfibrous swab and loaded with the chosen sheath. The sheath is then advanced into the artery smoothly, with gentle rotation to reduce resistance if necessary. When any extra resistance is encountered, the sheath should not be advanced without examining its position and that of the guidewire under fluoroscopy. This is also the case if any undue discomfort is experienced by the patient. After allowing the sheath to bleed back, the operator should consider performing check femoral angiography in the right anterior oblique (RAO) projection with the guidewire in situ (see Fig. 2-1 , C ). The guidewire may be directed medially by using a towel to aid identification of the arteriotomy site under fluoroscopy. When a long sheath is used, it is advanced approximately 10 cm before femoral angiography and then readvanced over the reinserted dilator and indwelling guidewire.
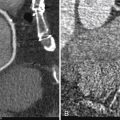
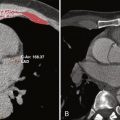
Checking femoral angiography at the outset of the procedure serves many uses. It identifies femoral artery anatomy and the presence of significant PAD ( Box 2-4 ; see Fig. 2-1 , D [e.g., calcification, tortuosity]), and it may prove vital in minimizing vascular complications if the patient requires PCI, in which larger sheaths may be needed. It also aids identification of possible complications, such as pseudoaneurysm formation in the case of low arterial puncture, and likely difficulty with manual compression and risk of hematoma or retroperitoneal hemorrhage in the case of high arterial puncture. For example, Figure 2-1 , E , demonstrates a high puncture with bladder compression from an extensive retroperitoneal hemorrhage. Femoral angiography is advised to assess for suitability before arterial closure device use.
- 1.
Aim to use the common femoral artery on the side with less PAD, if possible (stronger pulse, lesser claudication or objective evidence of lower PAD burden on the basis of arterial noninvasive testing, computed tomography angiography, or prior angiography).
- 2.
Identify key anatomic landmarks carefully as in item 1.
- 3.
Perform arterial entry under fluoroscopic guidance.
- 4.
Use a 0.035 steerable long guidewire with an atraumatic tip (e.g., Versacore wire, Abbott Vascular).
- 5.
Use a long sheath (23 cm at a minimum).
- 6.
Perform femoral angiography on successful sheath placement.
- 7.
Always exchange catheters over the guidewire by using the desheathing technique.
- 8.
When significant atherosclerotic disease is present at the arteriotomy site, avoid all active arteriotomy closure devices.
PAD, Peripheral arterial disease.
Radial Access
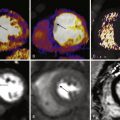
The radial approach for coronary angiography was initially described in 1989 by Campeau. Three years later, elective PCI through this approach was detailed by Kiemeneij and associates. Radial access as the default strategy has been embraced widely in many centers because of perceived benefits regarding patient comfort, early mobilization, and discharge, together with a significant reduction in morbidity and mortality after PCI in some series. Jolly and colleagues demonstrated a greater than 70% reduction in major bleeding rates with radial versus femoral access for diagnostic angiography or PCI. Significant bleeding as a consequence of femoral access vascular complications has been shown to have a negative impact on long-term survival mainly because of increasing 30-day mortality. Doyle and associates reviewed outcomes in more than 17,900 patients and found that greatest risk was seen with patients requiring transfusion of 3 or more units of blood. The most effective method of reducing the rate of major hemorrhage appears to be to employ the transradial approach. By virtue of its anatomy, the radial artery is very superficial and is not surrounded by any large veins or nerves, thus facilitating easy puncture and hemostasis. In addition, when the palmar arch is patent, the radial is not functionally an end artery, and so the hand should not be susceptible to ischemic complications should occlusion occur ( Fig. 2-2 , A and B ). Given these data, the obvious question is this: Why is the radial approach for arterial access so infrequently used?



Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree