Chapter 5 Radiography for the Critical Care Patient
For a safe and logical approach, the radiologist should first determine the nature and location of all the support devices, such as tubes and lines (Table 5-1). Incorrect positioning is an important cause of morbidity. A systematic review of the lungs, pleura, and mediastinum ensures that all important observations are made. Interpretation may be difficult, because the radiographic features are often nonspecific, and it is therefore important to have as much clinical information as possible and the aid of prior radiographs. Additional radiographic studies, such as decubitus views, ultrasound, or CT, often help to clarify a difficult interpretation or a suspected clinical problem.
Table 5-1 Correct Positioning for Tubes and Lines
| Tube or Line | Location |
|---|---|
| Endotracheal tube | 5 to 7 cm above the carina |
| Nasogastric tube | Side holes or tip below the left hemidiaphragm in the stomach |
| Central venous pressure catheter | Superior vena cava |
| Pulmonary artery line | Pulmonary artery within 2 cm of the hilum |
| Intra-aortic counterpulsation balloon catheter | Just below the superior aortic knob contour |
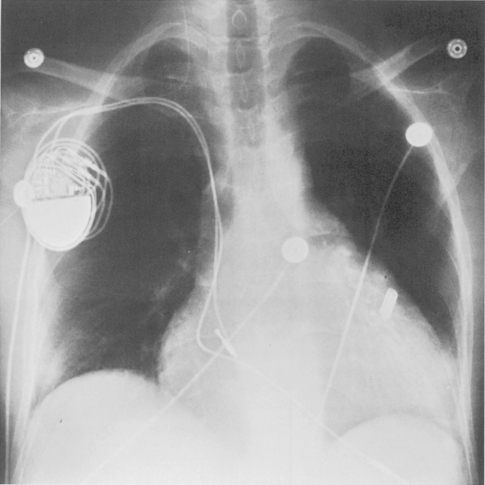
| Cardiac pacemaker (right ventricular lead) | Posteroanterior view: projected over the cardiac apex; lateral view: lies anterior and inferior (behind sternum) |
| Automatic implantable cardioverter-defibrillator | Proximal lead: superior vena cava; distal lead: right ventricle; patch: left chest wall or on pericardial surface |
| Pleural drainage tubes | Midaxillary, sixth to eighth interspace directed anterosuperiorly (pneumonectomy) or directed posteroinferiorly (effusion) |
SUPPORT DEVICES
Endotracheal Tube
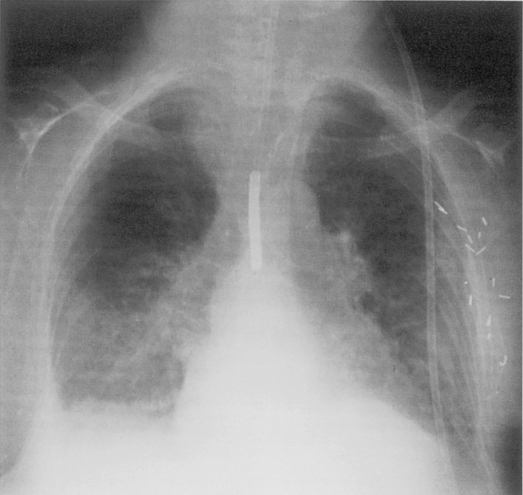
Malposition occurs in about 15% of placements, with emergency intubation having the highest complication rates. Physical examination is an unreliable guide to correct tube location, and a chest radiograph is required for confirmation. Correct placement of the endotracheal tube can be determined by the location of the tube tip relative to the carina. Ideally, the tip should be within 5 to 7 cm of the carina, with the head in the neutral position (i.e., inferior border of the mandible projected over the C5 and C6 vertebra). During flexion and extension of the cervical spine, the tip of the endotracheal tube may vary by a distance of 2 cm. The endotracheal tube should be at least 3 cm distal to the cords. A location that is too high may result in inadvertent extubation or may damage to the vocal cords. A position that is too low results in endobronchial intubation, occurring on the right more frequently than the left (Fig. 5-1). This may result in overinflation and possibly pneumothorax on the intubated side and atelectasis of the opposite lung.
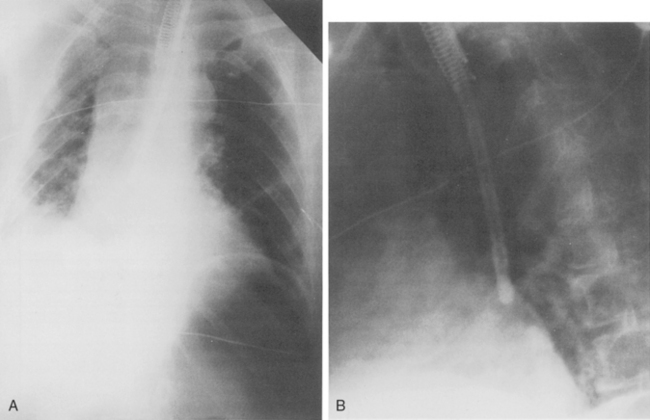
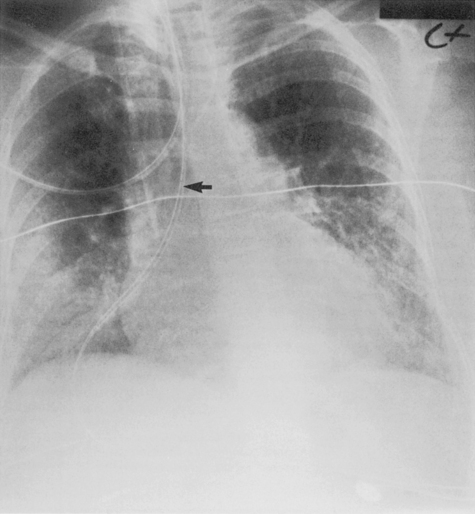
Esophageal intubation may be recognized by the margins of the endotracheal tube lying lateral to the tracheal air column and gaseous gastric distention. A right posterior oblique chest radiograph displaces the trachea to the right of the esophagus and allows recognition of the esophageal intubation (Fig. 5-2). The optimal width of the tube should be one half to two thirds of the width of the tracheal lumen, and the inflated cuff should not distend the tracheal wall. After a tracheostomy, the tube tip is ideally situated between one half and two thirds of the distance from the stoma to the carina. The width of the tube should be approximately two thirds of the width of the trachea, and the tip should not be wedged against the tracheal wall.
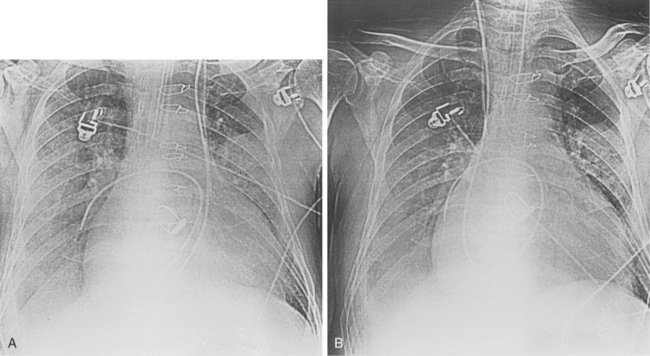
Tracheal laceration due to the endotracheal tube may result in pneumomediastinum, pneumothorax, or subcutaneous emphysema, singly or in combination. The tip of the endotracheal tube may be deviated to the right of the tracheal lumen. Inflation of the cuff by more than 2.8 cm (normal diameter is 2 to 2.5 cm) can cause tracheal laceration, as can positioning of the lower margin of the cuff at less than 1.3 cm from the tube tip (normal distance is 2.5 cm). Tracheal stenosis may occur at the tracheostomy stoma or at the endotracheal tube tip (Fig. 5-3). At the stoma, stenosis is caused by the formation of granulation tissue or by fibrosis with destruction of the tracheal cartilage. At the cuff site, stenosis results from a circumferential scar that is 1 to 4 cm long and that is typically 1.5 cm below the stoma.
Nasogastric Tube and Feeding Tube
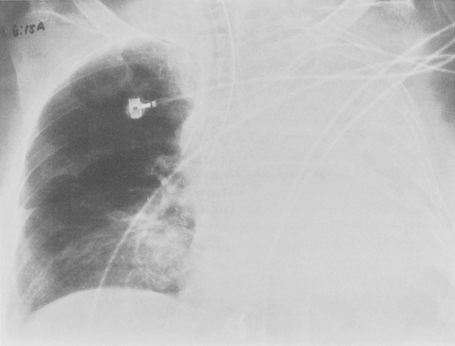
Incorrect positioning of a nasogastric tube is the most common tube complication (Fig. 5-4). Radiographic confirmation of correct positioning is mandatory before suction or feedings begin. The tube may be seen lodged within the tracheobronchial tree or coiled with the larynx or pharynx. More commonly, the tube lies too high in the esophagus above the gastroesophageal junction (Fig. 5-5). On many tubes, the side holes extend for a distance of 10 cm from the tip, and at least 10 cm of tubing should be seen within the stomach. Side holes above the gastroesophageal junction place the patient at risk for aspiration of gastric contents. Feeding tubes should be positioned in the duodenum to reduce the risk of gastroesophageal reflux of feedings and aspiration. The enteroflex tube is inserted over a wire, and perforation of the esophagus or stomach is a potential hazard. The stiff stylet may inadvertently enter the lung and cause a pneumothorax (Fig. 5-6). Complications associated with the nasogastric and feeding tubes are listed in Box 5-1.
Central Venous Catheter
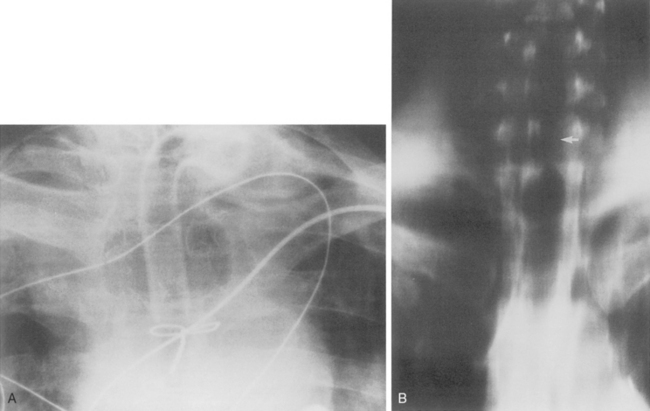
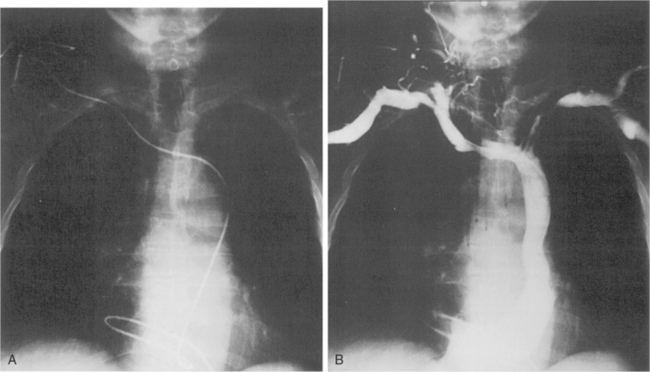
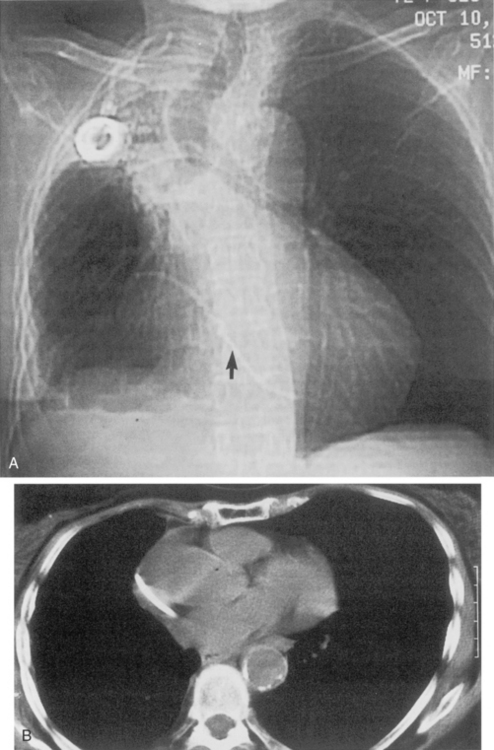
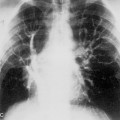
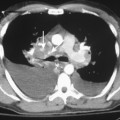
Central venous catheters are used routinely in the management of critically ill patients for venous access and measurement of intravascular blood volume (i.e., central venous pressure). Up to 40% of catheters are malpositioned. The catheters are usually placed through the subclavian or internal jugular vein. The optimal site for the catheter tip is within the superior vena cava, identified on the frontal view as at the level of the first anterior intercostal space. A catheter within the brachiocephalic veins produces inaccurate central venous pressure measurements due to interference by the proximal venous valves, and positioning within the right atrium is associated with a risk of cardiac perforation and arrhythmias. A catheter that follows a left anterior paramediastinal course is most likely in a left-sided superior vena cava (Fig. 5-7). This venous anomaly occurs in 0.3% of the population, and is usually associated with a right-sided superior vena cava. The left superior vena cava drains into the right atrium by way of the coronary sinus (Fig. 5-8). Catheter placement in an arterial vessel is usually clinically suspected because of the pulsatile flow through the catheter. This may be confirmed on the chest radiograph by the course of the catheter following the major arterial vessels.
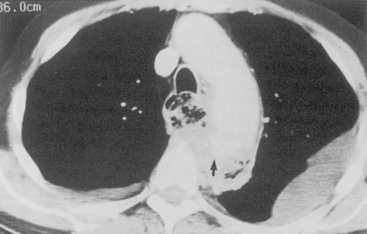
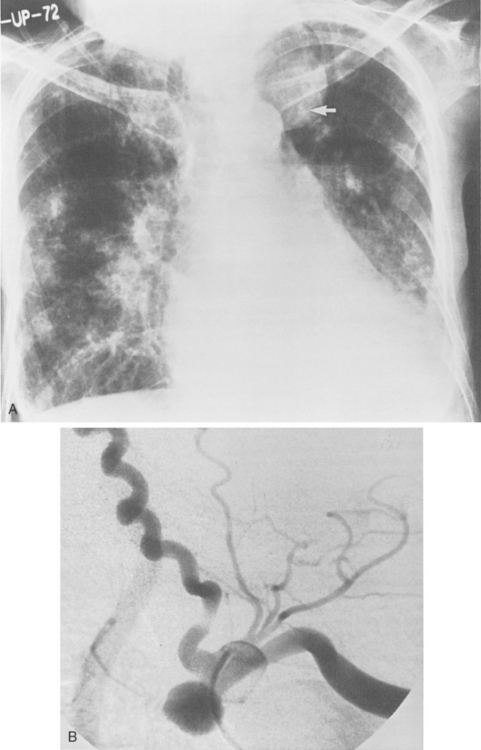
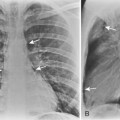
Inadvertent puncture of the subclavian or carotid artery may result in an extrapleural hematoma, recognized as a small apical opacity or as mediastinal widening due to more extensive bleeding. Rarely, a pseudoaneurysm of these vessels may develop (Fig. 5-9).
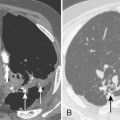
Catheter fragmentation with subsequent central venous embolization is estimated to occur in 1% of catheter placements (Fig. 5-10). Many cases are unrecognized clinically and may be detected by the astute radiologist. The fragments typically migrate through the central veins and right heart chambers and into the pulmonary artery and its branches. Death, arrhythmias, cardiac or vessel perforation, sepsis, mycotic aneurysm, and pulmonary emboli may result. In many cases, percutaneous retrieval devices can successfully remove the fragment.
Complications of line insertion in part reflect the expertise of the operator. For this reason, the femoral vein approach has gained popularity. There is no risk of a pneumothorax and access is direct and technically easier, and the puncture site is readily compressible. Bleeding from an inadvertent puncture of the femoral artery is easily controlled. Concern over the potentially increased risk of infection and thrombosis using this approach has proved unwarranted. Complications associated with the central venous catheter are reviewed in Box 5-2.
Percutaneous intravascular central catheters (PICCs) are particularly useful for long-term access. They are small (2 to 5 F in diameter) and are routinely placed by way of the antecubital veins. Because of their fine caliber, they may be difficult to visualize radiographically, particularly in the mediastinum. There is obviously no risk of a pneumothorax, and the risk of infection and thrombosis is low. Because these lines are very flexible and may become displaced, they should be monitored by routine radiographs (Fig. 5-11).
Pulmonary Artery Catheter
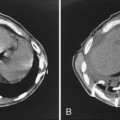
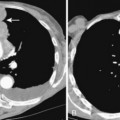
Pulmonary hemorrhage, another complication, has a similar radiographic appearance and is more common in patients with pulmonary arterial hypertension and in those receiving anticoagulation (Fig. 5-12). It may be caused by pseudoaneurysm formation, a rare but potentially fatal complication resulting from rupture of the pulmonary artery (Fig. 5-13
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree