Introduction
The United States (US) Food and Drug Administration (FDA) has overseen medical device marketing in the US since May 28, 1976, when the Medical Device Amendments to the Federal Food, Drug and Cosmetic Act were passed. The system that was established has first and foremost the public safety in mind, focusing on regulations for the marketing of medical devices in the US. While safety is the first priority, the FDA also has purview to mandate evidence of efficacy for any devices it chooses, but certainly for devices that are either novel or high risk. A key point is that the FDA regulates the medical device industry which designs, manufactures, and sells medical devices to physicians and hospitals. The FDA does not typically regulate hospitals or individual physicians. Physicians operate with certifications from the state and their medical society along with privileges to practice medicine at a specific medical facility or hospital. This is the concept of the “practice of medicine.” Dating back to 1908, the “practice of medicine” derived its name from the custom of administering so-called “medicines.” Today, the “practice of medicine” is defined as diagnosing, treating, operating, or prescribing for any human disease, pain, injury, deformity, or physical condition. Licensed physicians or healthcare professionals with appropriate state and hospital privileges can treat patients as they see fit under the “practice of medicine.”
A medical device is defined by the FDA as follows:
Per Section 201(h) of the Food, Drug, and Cosmetic Act, a medical device is: An instrument, apparatus, implement, machine, contrivance, implant, in vitro reagent, or other similar or related article, including a component part, or accessory which is:
- 1.
recognized in the official National Formulary, or the US Pharmacopoeia, or any supplement to them,
- 2.
intended for use in the diagnosis of disease or other conditions, or in the cure, mitigation, treatment, or prevention of disease, in man or other animals, or
- 3.
intended to affect the structure or any function of the body of man or other animals, and which does not achieve its primary intended purposes through chemical action within or on the body of man or other animals and
- 4.
which does not achieve its primary intended purposes through chemical action within or on the body of man or other animals and which is not dependent upon being metabolized for the achievement of its primary intended purposes. The term “device” does not include software functions excluded pursuant to section 520(o).
The key takeaways from the definition include that a medical device is meant for diagnosis or in the cure, mitigation, treatment, or prevention of disease. Medical device products range from simple gauze and surgical tape to very complex total joint implants and pacemakers. In the US, diagnostic imaging equipment such as ultrasound devices, computed tomography (CT) systems, and magnetic resonance imaging (MRI) machines are also included and regulated as medical devices.
Medical Device Classification
The FDA uses a classification system based on risk of the device and the regulatory controls necessary to provide a reasonable assurance of safety and effectiveness. Medical devices are categorized into one of three classes—Class I, II, or III ( Table 9.1 ). All device manufacturers, regardless of device class, must follow “general controls,” i.e., have a quality system, maintain records, and perform duties such as adverse event reporting in addition to registering and listing the device with the FDA.
| Class I | Class II | Class III | |
|---|---|---|---|
| Risk level | Low | Moderate | High |
| Type of FDA submission | Typically none | 510(k) | Premarket approval (PMA) |
| Clinical data required? | No | Mostly no | Yes |
Low-risk devices fall into Class I and typically do not require premarket notification or approval from the FDA. If a manufacturer would like to sell a Class I product which does not require premarket notification or premarket approval (PMA), they must comply with general controls, register as a manufacturer with the FDA, and list the products with the FDA prior to selling. It is estimated that just under half of all medical devices fall into the Class I category.
Class II devices contain higher, but still moderate levels of patient risk and are typically subject to premarket notification. Premarket notification for Class II medical devices typically involves filing a 510(k) application which provides safety data and compares the product to a known, previously approved “predicate device.” The predicate device must have the same indications for use and risk profile as the subject device. The use of predicate devices is key to the way the FDA has worked for over three decades. Again, just under half of all products fall into the Class II category, including most implants (e.g., total knee implants, facial fracture plating) and diagnostic imaging systems such as CT scanners or MRI machines.
Class III devices are considered high-risk devices and may be life-supporting, life-sustaining, or important in preventing impairment of human health. If the risk to the patient is high and/or there is little known about similar devices in the market, the FDA will require more data and assurance about the device. Class III devices represent less than 10% of all devices on the market and this category is reserved for either high-risk devices or devices without adequate historical market data. These devices typically require a PMA application to be filed and most times the FDA requires clinical data in support of these PMAs. When the PMA is cleared by the FDA, it is cleared for both safety and efficacy. Efficacy (or effectiveness) is something that the lower Class I and II categories cannot claim because their submissions typically are not forced to contain these data. Only a product cleared through the PMA pathway can be claimed to be “FDA-Approved.” All other clearances should be referred to as “marketing clearance” or “FDA-cleared” as it is inappropriate to use the term “approved” with devices which are not cleared through the PMA pathway.
Another key to the FDA’s way of looking at the medical device market is that they clear or approve a specific medical device for a specific intended use. Here it is helpful to lookup specific FDA Product Codes (aka procodes) which list a specific medical device, its classification, and whether or not it typically requires a 510(k), a PMA, or is Exempt. Many of these product codes are agnostic to whether the product is personalized, this product code could refer to a patient-matched or a non–patient-matched product, depending on the applicant and product.
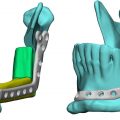
Let us take for example a cranioplasty device, product code GXN. The title of the device is “Plate, Cranioplasty, Preformed, Non-Alterable.” Looking at the listing, a more formal description in the Federal Register for product code GXN is found at 21 CFR Part 882.5330. From the listing we can tell that this device is a Class II device that typically requires a 510(k) and is overseen by the Neurology review panel within the FDA. If Company A files a 510(k) and obtains clearance for a product in the GXN product category with the FDA, then this product will be a patient-matched cranioplasty product. They use an indication for use statement of “<Product Name> is designed individually for each patient and is intended to replace bony voids in the cranial skeleton.” Indications statements are typically fairly short and to-the-point, although most are more than a single sentence. After getting the FDA’s marketing clearance for their product, Company A can market the product freely within the US for this indication. If Company A were to market this product to fill voids in another areas of the skeletal structure (e.g., the pelvis), this would be outside of the cleared indications for the product. The FDA considers this practice misbranding and “off-label use” of the device. The FDA regulates companies and prohibits them from off-label promotion but the FDA does not regulate physicians who safely use devices off-label in order to provide a solution that will positively impact patient care. Many shades of gray are present with regard to off-label usage of medical devices, but this only applies when the device is a medical device and has some cleared indication for use. In this case, if a second company, Company B, wants to sell the exact same product for cranial reconstruction, they would also need to go through the 510(k) process with the FDA. They could use Company A’s product as a predicate if they so desired. Preparing a 510(k) and choosing a predicate device is beyond scope for this chapter but there are many factors that would go into choosing a predicate, including access to the device and/or test data so as to use it for direct comparison to the new device.
Novel products called De Novo products are products which do not have an adequate predicate since they are new and follow an expanded pathway as compared to filing a traditional 510(k). Most times De Novo products end up in the Class II category. Future products after the first product is cleared through the De Novo pathway can be cleared using the De Novo product as a predicate with a traditional 510(k).
Another pathway for a Class III device targeting a small population is the Humanitarian Device Exemption pathway. These devices are cleared for limited numbers of patients per year and typically require submission of clinical data to which the FDA gives approval for safety and “probability of effectiveness.”
Medical Device Regulations
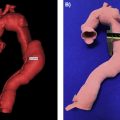
FDA regulations, which only apply to the medical device industry, include commercially available, 3D printed medical devices consisting of instrumentation (e.g., surgical guides to assist with proper placement of a device), implants (e.g., cranial plates or joint arthroplasty components), and external prostheses (e.g., lower extremity prosthetics). Between January 2010 and April 2016, more than 80 3D printed medical devices received 510(k) clearance, with the majority (83%) using powder bed fusion printing techniques.
Anatomic models have historically been regarded by the medical device industry as very low–risk medical devices. Many questioned early on whether an anatomic model is actually a medical device at all. Starting around 2000 several companies providing services in this area submitted themselves to the FDA’s scrutiny and listed products under a product code HWT—Template for Clinical Use, regulation number 888.4800. HWT devices consist of a pattern or guide such as that intended to assist with selecting or positioning of orthopedic implants or guiding the marking of tissue before cutting.
Although this was not a perfect fit for patient-specific 3D printed anatomic models, it seemed justifiable since these models are used for planning purposes. HWT is a Class I Exempt device, with the exemption allowing the product to bypass premarket notification or approval. The manufacturers of these devices are still required to abide by general controls, including company registration, device listing, maintaining a quality system, and adverse event reporting. Importantly, the fact that the manufacturer must register and list the device gives the FDA the authority to audit the manufacturer at any time it wishes. Companies that sold 3D printed anatomic models to physicians and hospitals used HWT as a “home” for anatomic models since approximately 2000. If the FDA disagreed with this classification, then they certainly did not make it known publicly, at least not until 2017.
In August 2017, an important public meeting was held at the FDA’s White Oak Campus (Silver Spring, MD) by and between the FDA and the Radiological Society of North America (RSNA) 3D Printing Special Interest Group (SIG). The meeting, which brought together different stakeholders (e.g., regulatory personnel, manufacturers, and healthcare professionals), was focused on 3D printed anatomic models and regulatory implications. The stated goals and objectives of the meeting were, as described on the FDA website,
- (1)
To provide a forum for open discussion between experts in 3D printing anatomic models from the clinical, industry, hospital, and regulatory field.
- (2)
To map out priorities for developing scientific evidence, identifying critical quality attributes, and building best practices for the safe clinical use of 3D printed anatomic models derived from medical imaging data.
Both of these goals were met during the course of this meeting, and a few very interesting clarifications were made by the FDA. First, the FDA clearly explained that HWT was not the appropriate product code for a 3D printed anatomic model. They stated that a 3D printed anatomic model, if marketed for diagnostic use, was considered a Class II medical device and should be listed under product code LLZ, “System, Image Processing, Radiological.” LLZ is typically related to software for PACS and image processing, and several of the software historically used in image processing for 3D printing are cleared under product code LLZ. The FDA reiterated that the existing software cleared for image processing stopped short of the actual 3D printing and as such could not claim that the outputs of these software packages were “diagnostic use” anatomic models. In addition, the FDA clarified that they see the image processing software as the most important and critical portion of the workflow for creating a “diagnostic use” anatomic model.
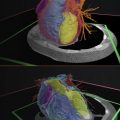
The term “diagnostic use” has been frequently misconstrued in the context of patient-specific 3D printed anatomic models. We will recall the word diagnosis appearing in the definition of a medical device. Many in the medical field have believed diagnosis was more of a literal word concerning the definitive predilection of the patient’s medical condition. Radiologists diagnose from medical images on a daily basis. Indeed the FDA clarified that their interpretation of the term diagnostic use was broader by far than classification of illness. Diagnostic use of a 3D printed anatomic model was defined by the FDA as shown in Fig. 9.1 .