Anatomy, embryology, pathophysiology
Please see Chapter 23 for discussion.
Techniques and protocols
Please see Chapter 23 for discussion.
Specific disease processes
Benign solid lesions
Oncocytoma
- ◼
An oncocyte is a large transformed epithelial cell with fine granular eosinophilic cytoplasm. Oncocytomas arises from distal tubules or collecting ducts of the kidney.
Epidemiology
- ◼
3% to 7% of all renal neoplasms.
- ◼
Present in the sixth to seventh decade of life.
- ◼
Male predilection (2:1).
- ◼
3% are bilateral, 5% are multicentric.
Clinical presentation
- ◼
75% are asymptomatic and incidentally diagnosed.
- ◼
Uncommonly presents with flank mass, pain, or hematuria.
Computed tomography
- ◼
Well-defined solid mass with smooth margins and central stellate scar. This is nonspecific; renal cell carcinoma (RCC) with central necrosis can appear the same.
- ◼
Noncontrast computed tomography (CT): isodense to hyperdense relative to normal renal parenchyma.
- ◼
Nephrographic phase: hypodense relative to normal renal parenchyma ( Fig. 24.1 ).

Fig. 24.1
Axial contrast-enhanced computed tomography in the nephrographic phase shows a solid enhancing mass with central low attenuation scar in the upper pole of the right kidney.
(From Sahani DV, Samir AE. Abdominal Imaging , ed 2. Philadelphia: Elsevier; 2017.)
Magnetic resonance imaging
- ◼
Low signal on T1-weighted image, high signal on T2-weighted image.
- ◼
Shows homogeneous enhancement with contrast.
- ◼
Central scar will show low T1 and T2 signal, in contrast to central necrosis, which has high T2 signal.
- ◼
Central scar does not enhance ( Fig. 24.2 ).

Fig. 24.2
T1-weighted postcontrast axial magnetic resonance image with fat suppression in the same patient as in Fig. 24.1 demonstrates enhancement of the left renal oncocytoma with no enhancement of the hypointense central scar.
(From Sahani DV, Samir AE. Abdominal Imaging, ed 2. Philadelphia: Elsevier; 2017.)
Angiography
- ◼
Spoke wheel arrangement of vessels.
- ◼
Homogeneous dense tumor blush.
- ◼
Sharply demarcated from the kidney.
Differential diagnosis
- ◼
Main consideration is RCC.
Treatment
- ◼
Oncocytoma cannot be reliably distinguished from RCC on cross-sectional imaging. It is also difficult to distinguish from other oncocytic tumors (e.g., chromophobe RCC) on histological evaluation from needle biopsy. Therefore, treatment is typically with partial or total nephrectomy.
Angiomyolipoma
- ◼
Benign renal tumor, belonging to the PEComa family (perivascular epithelioid cell tumors) of mesenchymal tumors. They are composed of varying amounts of blood vessels, fat, and smooth muscle.
Epidemiology
- ◼
Majority are sporadic (80%).
- ◼
Female predilection (4:1).
- ◼
20% associated with tuberous sclerosis (more often larger, multiple and bilateral).
Clinical presentation
- ◼
Often incidental.
- ◼
Risk of bleeding is proportional to size of lesion when larger than 4 cm.
- ◼
Intratumoral or perirenal hemorrhage may cause mass effect leading to flank pain, nausea/vomiting, and fever.
- ◼
Computed tomography
- ◼
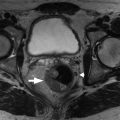
Presence of intratumoral fat (<–20 HU) is nearly pathognomonic for angiomyolipoma (AML) ( Fig. 24.3 ).

Fig. 24.3
Unenhanced computed tomography of the abdomen shows a large fat-containing mass in the left kidney with layering hemorrhage. Findings are compatible with an angiomyolipoma.
(From Sahani DV, Samir AE. Abdominal Imaging , ed 2. Philadelphia: Elsevier; 2017.)
- ◼
About 5% of AMLs have insufficient fat to be recognized on CT. RCC cannot be entirely ruled out in this case.
- ◼
Hemorrhage may obscure the fatty component of the lesion.
Magnetic resonance imaging
- ◼
Variable areas of high signal on T1 and T2 depending on fat content.
- ◼
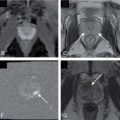
Use fat suppression techniques and chemical shift imaging to distinguish intratumoral fat. India ink artifact at the mass/kidney interface within the renal mass suggests AML (note, however, that clear cell RCC can also show signal loss on opposed-phase imaging due to intracellular lipid content) ( Fig. 24.4 ).

Fig. 24.4
In phase (A), opposed phase (B), and fat-suppressed (C) T1-weighted magnetic resonance imaging of the abdomen demonstrates a small exophytic mass along the posterior cortex of the left kidney. The mass shows type II chemical shift artifact (India ink artifact in B) and signal dropout on fat-suppressed imaging (C) because of the macroscopic fat present in this angiomyolipoma.
(From Sahani, D.V, Samir, A.E. Abdominal Imaging, ed 2. Philadelphia, Elsevier, 2017.)
Ultrasound
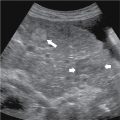
- ◼
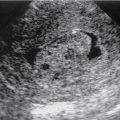
Well-defined hyperechoic mass ( Fig. 24.5 ).

Fig. 24.5
Sagittal ultrasound of the right kidney shows a small well-circumscribed homogeneously hyperechoic solid renal mass, consistent with angiomyolipoma.
(From Sahani DV, Samir AE. Abdominal Imaging , ed 2. Philadelphia: Elsevier; 2017.)
- ◼
AML is more likely than RCC to have posterior acoustic shadowing.
Angiography
- ◼
Demonstrates clusters of saccular microaneurysms or macroaneurysms.
Differential diagnosis
- ◼
RCC with perirenal fat entrapment or fat necrosis.
- ◼
Rare: myolipoma, liposarcoma, lipoma, oncocytoma, and Wilms tumor.
Treatment
- ◼
If small and asymptomatic, no treatment necessary.
- ◼
Larger lesions at risk of hemorrhage → prophylactic embolization (>4 cm).
Juxtaglomerular tumors
- ◼
Renin-producing tumors of juxtaglomerular cells usually arising beneath the renal capsule.
Epidemiology
- ◼
Female predilection.
- ◼
Patients younger than those with essential hypertension.
Clinical presentation
- ◼
Hypertension.
- ◼
Polydipsia.
- ◼
Polyuria.
- ◼
Hypokalemia secondary to hyperaldosteronism.
Computed tomography
- ◼
Nonenhanced CT: isodense relative to normal renal parenchyma.
- ◼
Hypovascular on contrast-enhanced imaging.
Ultrasonography
- ◼
Echogenic mass.
Differential diagnosis
- ◼
Renal artery stenosis.
- ◼
Other renin-secreting tumors.
- ◼
Mass effect on renal artery or renal parenchyma from an RCC or Wilms tumor.
Treatment
- ◼
Surgical excision is curative.
Adenoma
- ◼
Develop in the proximal convoluted tubule and are of unknown etiology.
Epidemiology
- ◼
Most are identified at autopsy and are of little clinical significance.
- ◼
Same age group as RCC.
- ◼
Male predilection.
Clinical presentation
- ◼
Usually asymptomatic.
Imaging
- ◼
Indistinguishable from RCC on imaging.
Malignant lesions
Renal cell carcinoma
- ◼
Most common renal malignancy (85%–90% of cases).
- ◼
Accounts for 3% of all malignancies.
- ◼
Subtypes (in order of decreasing metastatic potential): Clear cell, papillary, chromophobe.
- ◼
Renal medullary carcinoma is a rare aggressive subtype in younger (mean age 22 years) patients with sickle cell trait. Mean survival after surgery is only 15 weeks.
Epidemiology
Environmental risk factors
- ◼
Smoking.
- ◼
Unopposed estrogen exposure.
- ◼
Obesity.
- ◼
Occupational exposure to petroleum, heavy metals and asbestos.
- ◼
Hypertension (and its treatment).
- ◼
Acquired cystic kidney disease secondary to chronic hemodialysis.
Hereditary risk factors
- ◼
Von Hippel-Lindau.
- ◼
Tuberous sclerosis.
- ◼
Uterine leiomyosarcoma/RCC syndrome.
- ◼
Birt-Hogg-Dube syndrome.
- ◼
Familial clear cell carcinoma.
- ◼
Median age at diagnosis → mid-60s (except for medullary carcinoma).
- ◼
Male predilection (2:1).
Clinical presentation
- ◼
Classically, hematuria, flank pain, and flank mass.
- ◼
In men, can present with new unilateral (usually left) varicocele from obstruction or thrombus of the left renal vein.
- ◼
RCCs can secrete hormones (erythropoietin, parathyroid hormone, prolactin, adrenocorticotropic hormone) resulting in paraneoplastic syndromes.
Radiography
- ◼
Peripheral rim calcifications are usually because of benign cysts (80% of cases) rather than RCCs (20% of cases) ( Fig. 24.6 ).