Fig. 40.1
Radiographic ossification and fusion of the tibial tuberosity, lateral radiographs of four different male individuals. (a) Eight-year-old boy, no ossification is detected at the tibial tuberosity (arrows). (b) 14-year-old boy, tongue-like ossification of the tibial tuberosity with small loose-calcified part (arrow). (c) 15-year-old boy, tongue-like ossification of the tibial tuberosity without loose-calcified parts. (d) 23-year-old adult male, ossification of the growth plate with fusion of the tibial tuberosity
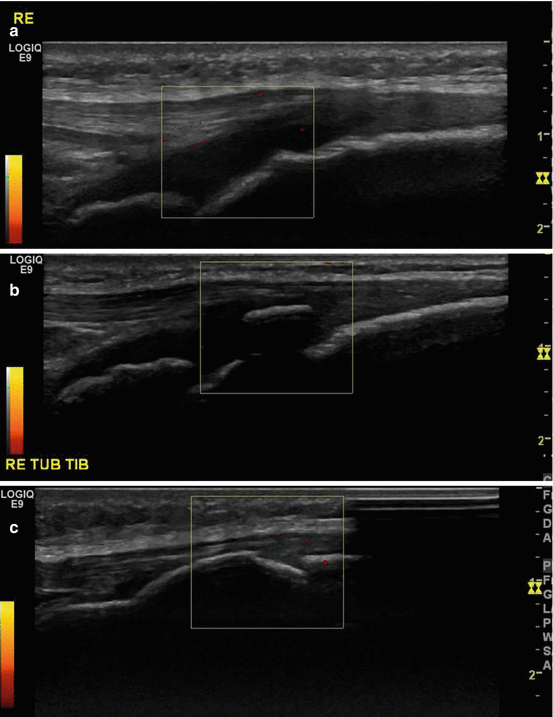
Fig. 40.2
Ducher ultrasound classification of osteochondrosis. (a) Cartilage attachment, characterized by a large amount of apophyseal cartilage and anechoic appearance without interspersed ossicles, with ultrasonographic band-like echogenic structures. (b) The second stage is the insertional cartilage, characterized by a progressive attachment of the collagen fibers onto the bone surface. A thin layer of cartilage is still visible, and the apophyseal surface appears smoother. (c) The third stage is the mature enthesis, characterized by a complete attachment of the collagen fibers onto the bone surface
40.2.3 Clinical Features
Pain at the knee is mild and intermittent, although in the acute phase, it can be severe and continuous in nature. Pain exacerbates after sporting activities involving jumping (basketball, volleyball, running) or on direct contact (kneeling) (Flowers and Bhadreshwar 1995). Physical examination usually finds tenderness directly over the area of the tibial tuberosity and local swelling. The pain can be reproduced with extension of the knee against resistance. Once the acute phase heals, the pain and tenderness subside, and the only positive physical finding may be an anterior mass. Most studies claim spontaneous resolution and self-limiting nature of the symptoms with recovery expected in about 90 % of the patients. In approximately 10 % of the patients, the symptoms continue unabated into adulthood, despite conservative treatment (Gholve et al. 2007).
40.2.4 Incidence and Prevalence
Osgood-Schlatter disease affects approximately one in five adolescent athletes (Hogan and Gross 2003). It is more prevalent in males than females. However, more and more girls are affected, because over the years more girls are participating in sports and girls become taller, so they have a growth spurt equally like boys. Osgood-Schlatter is seen in males between 12 and 15 years and in females between 8 and 12 years of age, although other studies mention other ages for girls, between 11 and 13 years of age (Duri et al. 2002). The difference in age is due to the earlier onset of puberty and the associated earlier epiphyseal plate closure (Gholve et al. 2007). The condition is more common in patients that experience a rapid growth during adolescence (Vreju et al. 2010). It frequently presents bilaterally 20–30 % (Bloom et al. 2004; Wall 1998).
40.2.5 Diagnosis
The diagnosis is mostly made clinical, but imaging methods are often used to confirm it. Plain radiographs of the knee are recommended in all unilateral cases of Osgood-Schlatter disease to rule out other conditions such as acute tibial apophyseal fracture, infection, or tumor. A lateral view of the knee with the leg internally rotated 10–20° shows irregularity of the apophysis with separation from the tibial tuberosity in early stages and fragmentation in the later stages (Fig. 40.3). A persistent bony ossicle may be visible in a few cases after fusion of the tibial epiphysis (Gholve et al. 2007). The isolated appearance of an irregular ossification center in the absence of surrounding inflammatory changes is a normal development variant and should not be the only criterion used to make the diagnosis (Dupuis et al. 2009).
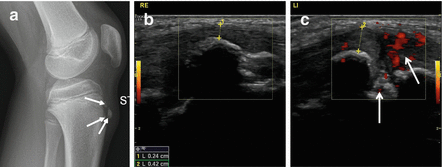

Fig. 40.3
Radiograph and ultrasound of Osgood-Schlatter disease. Thirteen-year-old boy with chronic pain at the left tuberositas tibiae. (a) Lateral radiograph of the left knee shows soft tissue swelling at the tuberositas tibiae with fragmentation of the ossifications at the tuberositas tibiae (arrows). (b) Ultrasound midsagittal imaging plane at the level of the normal right tuberositas tibiae and (c) of the left tuberositas tibiae showing soft tissue thickening in front of the tuberositas and angiogenesis at the noncalcified parts (arrows)
MRI may assist in the diagnosis of an atypical presentation. The role in the diagnosis is currently limited. On MRI, the normal infrapatellar tendon appears as a homogeneous band of low-signal intensity on all sequences. Hirano et al. studied the MRI appearance of Osgood-Schlatter. They proposed five stages of the disease on MRI: normal, early, progressive, terminal, and healing stage. In the normal stage, the MRI scan is normal although the patient has developed symptoms. The early stage does not reveal any MRI evidence of inflammation or avulsed portion of the secondary ossification center. There may be edema-like changes around the tibial tuberosity, but a normal MRI is not ruling out early Osgood-Schlatter. In the progressive stage, a partial tear in the secondary ossification center is detected. If the tear is extended, the anterior parts that consist of bone and cartilage are avulsed. The terminal stage is characterized by the presence of separated ossicles (Fig. 40.4). The avulsed part is pulled superiorly and separated completely. The ossicle is formed from this avulsed part. The healing stage shows an osseous healing of the tibial tuberosity without separated ossicles. The intervening region may heal subsequently by fibrocartilage. If the gap is small, the bridge of fibrocartilage may ossify. However, larger gaps may remain, with the avulsed fragments maturing to form separated ossicles within the patellar tendon (Hirano et al. 2002; Gholve et al. 2007).
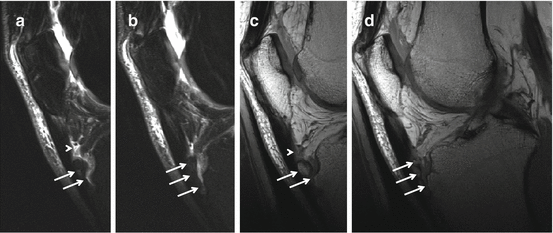

Fig. 40.4
MRI in Osgood-Schlatter disease. Adult, 56 years old, male with long-standing complaints at the right tuberositas tibiae that started during adolescence. Fragmented aspect of the tuberositas tibiae with increased T2 time surrounding the bone fragments (arrows), thickening of the distal patellar tendon, and fluid signal at the deep infrapatellar bursa (arrowheads). (a, b) Sagittal intermediate TE with FS and (c, d) sagittal PD WI. A and C have identical slice position and thickness and (b, d) have identical slice position and thickness
Ultrasonography is more and more used to diagnose Osgood-Schlatter. It has a superior spatial resolution over MRI and provides a rapid, cheap, accessible imaging technique to assess the infrapatellar tendon. On ultrasonography, the parallel tendon fibrils are echogenic. Osgood-Schlatter disease is characterized by pretibial soft tissue swelling, cartilage swelling, fragmentation of the tibial tuberosity’s ossification center, thickening at the insertion of the patellar ligament, and inflammation of the deep infrapatellar bursa (Figs. 40.3, 40.5, 40.6 and 40.7). Calcifications are easily detected by ultrasonography compared to MRI. On ultrasonography, Osgood-Schlatter can be divided into three types. Type one is characterized by delamination of the internal ossification center, resulting in an igloo-like deformation of the physeal part of the tibial tuberosity (Fig. 40.5). Other signs are a deep infrapatellar bursitis and/or fibrosis due to a shared arterial supply with the tibial tuberosity, inflammation of the patellar ligament secondary to the main injury and deep infrapatellar bursitis, and a superficial infrapatellar bursitis secondary to deep infrapatellar bursitis or secondary to the main injury. The prognosis of type one is very favorable as it leaves minimal disturbance to the shape of the tuberosity and the state of the patellar ligament. Type two is characterized by a delamination tear or fracture of the epiphyseal part of the tibial tuberosity and a significant anterior displacement of the proximal attachment of the patellar ligament due to the displacement of the fractured cartilage (Figs. 40.6 and 40.7). Other signs are a deep infrapatellar bursitis due to bleeding from the torn cartilage or ossification center, inflammation or fibrosis of the patellar ligament secondary to the main injury and the deep infrapatellar bursitis, and a superficial infrapatellar bursitis secondary to the deep infrapatellar bursitis and the inflammation involving the enthesis zone of the patellar ligament. The prognosis of type two is moderately favorable as it tends to create significant bursal fibrosis including impairment of dynamic behavior of the fatty apron of the deep infrapatellar bursa (Hoffa’s fat pad). However, it doesn’t leave a significant scarring within the patellar ligament. Type three is characterized by a delamination tear of the ossification center resulting in an irregular deformation of the tuberosity; there is a cartilage fracture with or without significant anterior displacement of the proximal attachment of the patellar ligament. The cartilage fracture has to be located at least partially within the footprint of the patellar ligament insertion. Other signs are, first, a deep infrapatellar bursitis or fibrosis due to an injured arterial supply within the tibial tuberosity, the bleeding from the patellar ligament, and the fractured cartilage; second, an inflammation of the patellar ligament; and last, a superficial infrapatellar bursitis and a focal scarring or possible bone formation due to a tear in the patellar ligament arising from its tibial insertion. For this type, the prognosis is unfavorable with a high probability of chronic symptoms due to fibrosis of the deep infrapatellar bursa and ectopic bone formation within the patellar ligament’s scar (Czyrny 2010) (Fig. 40.6).
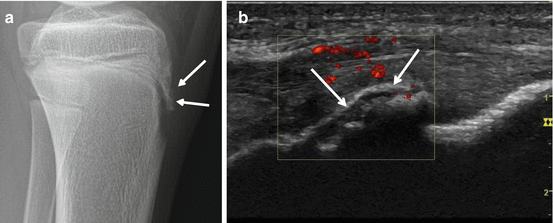
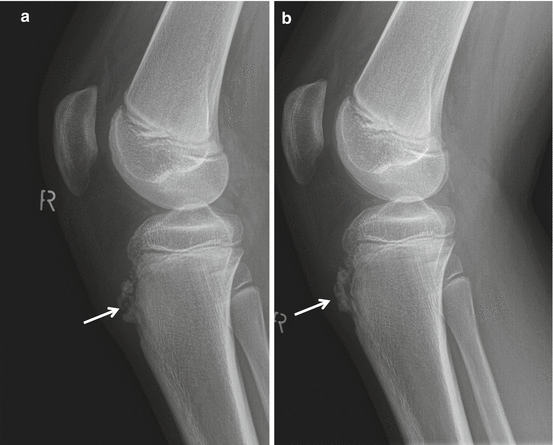
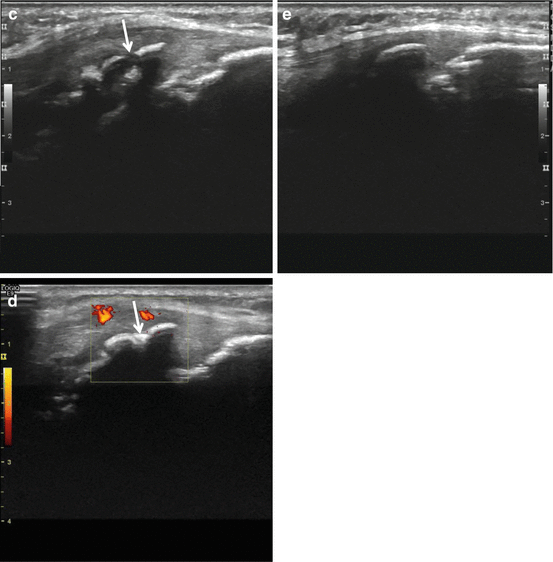
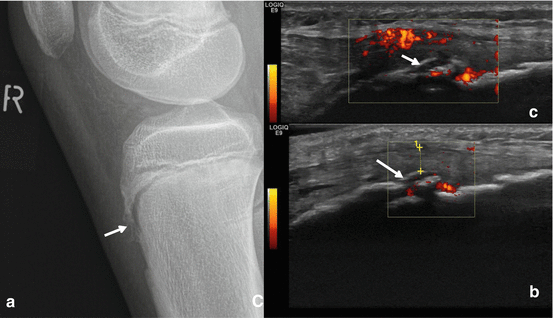

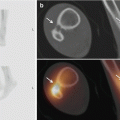
Fig. 40.5
Radiograph and US of Osgood-Schlatter, Igloo aspect. Thirteen-year-old boy soccer player with fracture of the left tuberositas tibiae. Radiographically, (a) a shell-like fracture fragment is documented (arrows). On ultrasound, (b) the shell-like fragment presents like an igloo with shine through of the deeper calcified parts of the tibial tuberosity. The noncalcified parts of the tibial tuberosity are hypervascular on power Doppler ultrasound


Fig. 40.6
US and radiograph of Osgood-Schlatter disease. Boy, 11 years old, with right-side Osgood-Schlatter disease, lateral radiographs, and ultrasound examinations (a and c) with follow-up three months later (b and d. Fractured aspect of the tuberositas tibiae on radiograph (a) and ultrasound (c) (arrows) with thickening of the patellar tendon. Left tibial tuberosity for comparison (e). Three months later, ossification at the fracture line (arrows) of the tuberositas tibiae on radiograph (b) and ultrasound (d). Decreased thickness and angiogenesis of the noncalcified parts

Fig. 40.7
Radiograph and US of Osgood-Schlatter disease; US demonstration of angiogenesis and combined fractured and igloo aspect. Twelve-year-old boy with right-sided Osgood-Schlatter disease. Fragmented tuberositas tibiae (a) with igloo aspect on ultrasound (b and c arrows) and angiogenesis at noncalcified parts of tuberositas. Nonfavorable clinical evolution with increased pain and swelling, follow-up ultrasound examination 6 weeks (c) later with increased angiogenesis and increased fragmentation
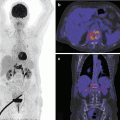
Bone scintigraphy is not generally used in clinical suspicion of osteochondroses. Regarding Osgood-Schlatter, only a case report is available describing a young adult male in which the activity status of the disease correlated better with the scintigraphic activity status; actually, this is better and with less radiation burden achieved with ultrasound with Doppler imaging (Namey and Daniel 1980).
40.3 Sinding-Larsen-Johansson Syndrome
Sinding-Larsen-Johansson Syndrome (SLJS) is affecting the patellar apex at the proximal insertion of the patellar tendon accompanied by radiographic evidence of fragmentation or a calcification at the patellar apex. It has a similar etiology as Osgood-Schlatter disease in that it is the result of repetitive microtrauma and excessive prolonged stress occurring at a well-defined skeletal region which is mechanically and biologically weak when stress exceeds intrinsic resistance (Figs. 40.8, 40.9, 40.10 and 40.11). However, in this condition, the stress is directed at the attachment of the patellar tendon on the inferior pole of the patella and not at a cartilaginous growth center. Some authors classify SLJS as jumper’s knee in pediatric setting, although jumper’s knee is a tendinopathy of the patellar tendon (Dupuis et al. 2009; Hogan and Gross 2003).
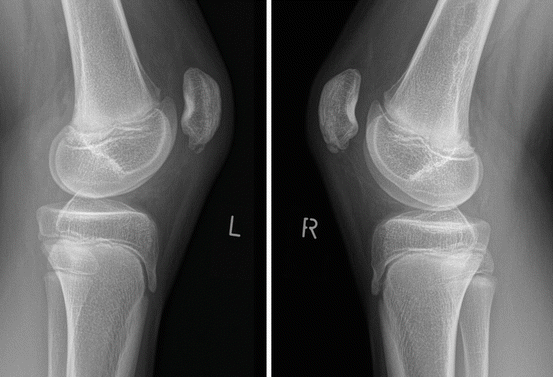
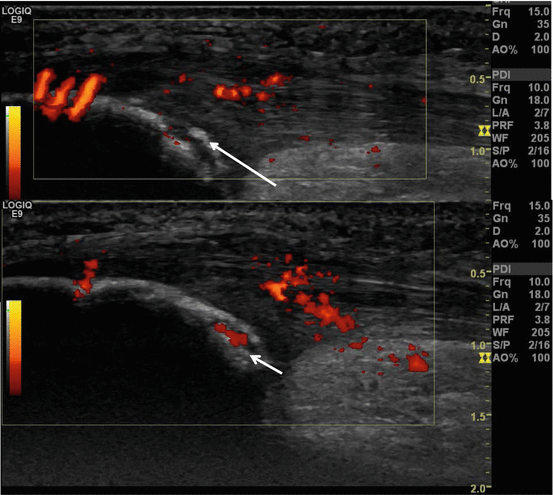
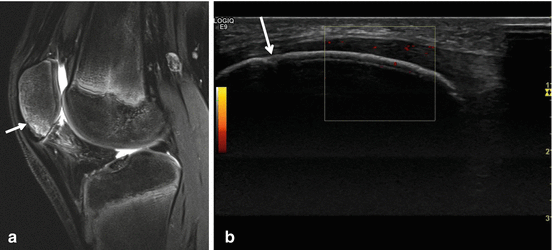
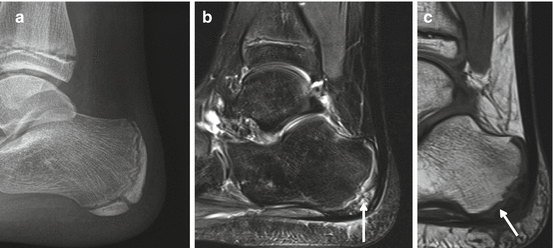

Fig. 40.8
Radiographs of SLJS. Fifteen-year-old boy with chronic complaints at both patella apices. Sinding-Larsen-Johansson disease of both patella, fragmented aspect of the apex of the patella on radiographs in advanced disease

Fig. 40.9
US of SLJS. Eight-year-old girl with chronic pain at the left patella apex. Sagittal ultrasound image at the level of the left patella apex with loose fragment (arrow) and angiogenesis (short arrow) at the level of the patellar tendon origin and the cartilaginous part (short arrow)

Fig. 40.10
SLJS, MRI, and US. Twelve-year-old boy with Sinding-Larsen-Johansson disease of both patella. MRI and US examinations. Sagittal MRI TSE intermediate TE with FS, increased SI at the patella apex and origin of the patellar tendon. Minor cortical irregularity at the mid-third of the patella (a, arrow) distending into the patella apex isolating a bone fragment. Only minor abnormalities on US with demonstration of the minor irregularity at the anterior cortical lining of the patella (b, arrow), minor thickening and angiogenesis at the prepatellar part of the patellar tendon

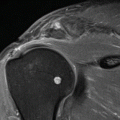
Fig. 40.11
MRI of Sever’s disease. Nine-year-old boy with pain at tuber calcanei since a few weeks, ultrasound and radiographs were inconclusive (a), MRI, sagittal images, TSE intermediate TE with FS (b) and SE T1 WI (c) demonstrating bone marrow edema at the apophysis with increased T2 time and reduced SI on T1 (arrows)
40.3.1 Clinical Features
SLJS is clinically characterized by pain localized at the distal pole of the patella, swelling of the infrapatellar soft tissues, and functional limitation. Patients complain of activity-related pain, especially with jumping, running, kneeling, or with climbing stairs.
40.3.2 Incidence and Prevalence
40.3.3 Diagnosis
Like Osgood-Schlatter disease, the diagnosis of SLJS is mostly based on clinical findings. SLJS results in calcification or ossification of the patellar tendon in the late stages of the disease, which can be often demonstrated on plain radiography. MRI (Fig. 40.10) demonstrates edema at the inferior pole of the patella and in the proximal portion of the patellar tendon and adjacent soft tissues (Dupuis et al. 2009). Ultrasonic findings are the same as those in Osgood-Schlatter disease: cartilage swelling, patellar tendon swelling at its proximal insertion, and patellar fragmentation at its distal pole (Figs. 40.9 and 40.10) (De Flaviis et al. 1989).
40.4 Sever’s Disease
Sever’s disease or apophysitis of the calcaneus is a common cause of heel pain in children and adolescents. The condition is overuse related in children between the age of 8 and 10 years in girls and 10 and 12 years in boys (Madden and Mellion 1996).
40.4.1 Etiopathogenesis
Sever’s disease is an osteochondrosis. The apophysis of the os calcis is an epiphyseal plate that develops along the posterior border of the bone. The Achilles tendon inserts on this calcaneus apophysis. The growth plate is weak and subject to injury. The most significant etiologic factor is overuse and microtrauma in sports (Michelli et al. 1987). Additionally, Sever’s disease can be caused by bursitis or necrosis of the apophysis (Mathieson et al. 1988). Sever’s disease occurs in children aged from 8 to 12 years although Heneghan and Wallace recorded cases from 10 to 13 years (Heneghan et al. 1985).
40.4.2 Clinical Features
The athlete complains of heel pain, which is aggravated by activity and frequently occurs bilateral. Physical exam reveals well-localized tenderness of the posterior heel at the site of insertion of the Achilles tendon (Hogan and Gross 2003).
40.4.3 Normal Development of the Achilles Tendon Insertion
Until the age of 3 years, no ossification of the secondary ossification center of the calcaneus is visible. From 4 to 6 years old, early signs of ossification of the secondary ossification center are present. In children from 7 to 11 years, a hypoechoic gap representing the growth plate cartilage can be seen between the posterior bony outline of the calcaneus and the ossified secondary ossification center at the calcaneal tuberosity. Among adolescents from 12 to 18 years of age, the apophyseal cartilage between the posterior bony contour of the calcaneus and the secondary ossification center of the calcaneal apophysis can appear as a hypoechoic gap (Fig. 40.8). The apophyseal growth plate fuses toward the end of skeletal growth (Grechenig et al. 2004).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree