A wide spectrum of incidental bowel findings can be seen on CT, including but not limited to, pneumatosis intestinalis, diverticular disease, non-obstructive bowel dilatation, transient small bowel intussusception, and submucosal fat. Radiologists should be aware that such findings are almost always benign and of little clinical significance in the absence of associated symptoms. Conversely, vigilance must be maintained when evaluating the bowel, because malignant neoplasms occasionally come to clinical attention as incidental imaging findings. When suspicious incidental bowel wall thickening is detected, the radiologist can alert the clinical team to the finding prior to the patient becoming symptomatic, potentially leading to definitive management at an early, more curable stage.
Key points
- •
Incidental bowel findings commonly are discovered on computed tomography (CT); most are benign and require no further work-up or management; however, some are clinically relevant. In particular, bowel wall thickening may reflect underlying neoplasm and should be brought to the attention of the referring clinician.
- •
There are a wide variety of causes of pneumatosis intestinalis, with clinical presentations ranging from asymptomatic to life-threatening bowel ischemia.
- •
Bowel dilatation most often is due to mechanical obstruction; however, the radiologist should be aware of the causes of acute and chronic nonobstructive bowel dilatation.
- •
Bowel intussusception in an adult should raise suspicion for a neoplasm acting as the lead point; however, a transient self-resolving intussusception can be suggested in the setting of a short segment intussusception with no upstream bowel dilatation or visible lead point.
- •
The fat halo sign can be a CT sign of inflammatory bowel disease; however, it probably is seen more frequently as an incidental and clinically insignificant finding.
Introduction
Incidental findings in the bowel commonly are encountered on CT examinations and encompass a diverse array of pathology, reflective of the unique spectrum of disease encountered in each segment of the intestine. Similar to incidental findings in other organ systems, most of these findings are benign and require no further work-up. Examples of benign disease covered in this chapter include diverticulosis, most cases of intramural fat, asymptomatic pneumatosis, nonobstructive bowel dilatation, and transient intussusception. A minority of incidental findings are clinically relevant or initially indeterminate, requiring further patient management and often necessitating additional imaging examinations and interventions. For example, malignant tumors of the gastrointestinal (GI) tract may have a long latency period before becoming symptomatic and frequently are detected on CT as incidental findings. Determination of the clinical relevance and optimal management of incidental findings often poses a dilemma for the radiologist. In recent years, there has been a proliferation of studies reviewing incidental findings on CT as well as position papers from the American College of Radiology written for the purpose of providing guidelines for management. Unfortunately, many of these articles do not include discussions or data regarding incidental bowel findings nor is there yet an American College of Radiology white paper dedicated to management of bowel findings. The goal of this article is to review the various incidental findings that can be detected on CT and MR imaging, discuss their clinical implications, and give examples from the literature in order to provide a perspective on management.
Wall thickening
Incidental bowel wall thickening (IBWT) is a relatively frequent finding and can be focal or regional. Although IBWT may be inflammatory, infectious, ischemic, or neoplastic in etiology, its clinical relevance when detected incidentally remains unclear, particularly when identified on CT (or occasionally on MR imaging or ultrasound [US]) performed for apparently unrelated reasons. Furthermore, there is no consensus in the literature, to the authors’ knowledge, on how to approach this finding.
The first challenge for radiologists is to determine to the best of their ability whether the suspected wall thickening is truly pathologic, or spurious, due to under-distention. Trends toward decreased use of positive oral contrast at many centers can make evaluation of the bowel difficult, especially when under-distended ( Figs. 1–3 ), and CT examinations performed without intravenous contrast do not allow for analysis of mural enhancement. Additionally, false-positive and false-negative results for an intestinal mass can result from stool or other intraluminal contents either obscuring or mimicking a mass.



The thickness of the wall of the GI tract varies between small bowel and large bowel and also is affected by multiple factors, including the degree of luminal distention. Normal small bowel wall is thin, measuring between 1 mm and 2 mm, when the lumen is well distended. When the bowel is partially collapsed, it can measure between 2 mm and 3 mm in thickness, which frequently is used as the upper limit of normal. The jejunum commonly appears falsely thickened when under-distended due to a greater fold density than the ileum. The normal wall of the colon is very thin—often barely perceptible—and when distended should measure less than 3 mm. , The wall generally is considered abnormal if there is thickening greater than 5 mm. Normal bowel wall also enhances predictably following intravenous contrast administration. Enhancement typically is uniform and symmetric and also usually is more pronounced in the mucosal layer.
When bowel wall thickening is detected, additional CT findings need to be analyzed for complete assessment and to formulate an effective differential diagnosis. Salient features include degree of thickening, symmetry, length of affected segment, mural attenuation, and any associated perienteric abnormalities. ,
The primary entity of concern in a patient with a short segment of thickened bowel is neoplasia, which is uncommonly but not rarely detected as a purely incidental finding on CT. For example, in a study of 1175 emergency department patients in whom abdominal/pelvic CT was performed, incidental findings were detected in 700, including 15 patients with colonic wall thickening, which yielded 4 colon cancers and 5 colorectal polyps. Additionally, 11 gastric lesions were detected, which yielded 1 gastric cancer.
In studies that have investigated the significance of IBWT diagnosed on CT, there is a high rate of disease found on endoscopic examination. A meta-analysis evaluating incidental colonic wall thickening on CT, and which included 9 studies and 1252 patients, found that 73% of patients had abnormalities at colonoscopy, with a cancer rate that ranged from 14% to 27%. Eskaros and colleagues found that 64% of patients with incidental colonic wall thickening on CT had a corresponding abnormality on colonoscopy. Colitis was the diagnosis in a majority of cases; however, in a subset of patients in which a mass was suspected on CT, this was confirmed in 100% (12/12) of cases. Uzzaman and colleagues found abnormalities on optical colonoscopy at the exact site of IBWT seen on CT in 58% of patients; in 36/95 of these patients, the abnormality was a malignancy. Documentation of the characteristics of the wall thickening would help in assessing the clinical relevance of IBWT in predicting the presence of a colonic neoplasm. Shorter segment of wall thickening, irregular or eccentric thickening, and greater degree of wall thickness all are factors that favor a neoplastic process; however, such analysis is lacking in most studies. Note that routine abdominal CT has been shown to be moderately sensitive for detection of colorectal neoplasia. Ozel and colleagues found routine abdominal CT to be 72% sensitive and 84% specific in diagnosing invasive colorectal cancer.
There are fewer studies in the literature that have looked at the significance of esophageal or gastric wall thickening compared with colonic wall thickening on CT, to the authors’ knowledge. Cai and colleagues found endoscopic abnormalities in 22/27 patients with distal esophageal wall thickening on abdominal CT, with esophagitis, varices, and hiatal hernia diagnosed in approximately equal frequency. Tellez-Avila and colleagues found abnormalities on endoscopy in 6/19 patients with incidental gastric wall thickening, all of which were adenocarcinomas.
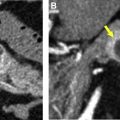
The appendix also can be a site of incidental tumors, such as a neuroendocrine tumor (NET) (the appendix is the most common site in the GI tract for NET), mucinous tumor, or adenocarcinoma ( Fig. 4 ).

In contrast to the more infiltrative appearance of wall thickening commonly encountered with gastric or colonic adenocarcinoma, an incidentally found circumscribed masslike focal process suggests an alternative diagnosis on CT, such as a NET, or GI stromal tumor (GIST), which arises most commonly in the stomach ( Fig. 5 ), followed by the small bowel.

Lipomas are benign submucosal tumors found throughout the GI tract, most commonly in the colon, and easily are diagnosed on CT as fat attenuation intramural or intraluminal masses. Although these generally can be ignored, lipomas occasionally can be the cause of complications, such as intussusception or bleeding. Tumors greater than 2 cm are more likely to ulcerate, leading to acute or chronic anemia.
Non-neoplastic entities also can be the cause of an incidental mass or wall thickening in the GI tract. Bowel wall thickening from chronic diverticulosis is a common finding, particularly in the sigmoid colon on CT, and usually can be distinguished from cancer by the longer length of involvement and presence of diverticulosis. Wall thickening in the setting of cirrhosis can be due to portal hypertensive colopathy, a benign condition with no clinical relevance in and of itself. CT shows wall thickening, usually involving the right colon, indistinguishable from a colitis; diagnosis can be made based on the presence of findings indicative of cirrhosis and portal hypertension and lack of symptomatology.
Heterotopic pancreas can occur in the stomach, duodenum, or jejunum and usually is discovered as an incidental small nodule or mass on CT or MR imaging, although rarely it can be the cause of acute pancreatitis ( Fig. 6 ).

In an estimated 12% to 37% of cases of endometriosis, endometriotic implants can involve the bowel, usually bowel segments in the dependent pelvis, involving in decreasing order of frequency, the rectosigmoid, appendix, cecum, and terminal ileum. These implants appear as serosal masses and may be large enough to be visualized on cross-sectional imaging ( Fig. 7 ). A diagnosis can be suggested when there is involvement of pelvic bowel loops and in the setting of a known history of endometriosis, although primary GI tract tumor and metastatic disease are in the differential diagnosis. Concurrent findings of endometriosis, such as complex adnexal cyst(s) or hydrosalpinx, if present, can assist the radiologist in arriving at the correct diagnosis.

Splenosis, a condition defined by heterotopic autotransplantation of splenic tissue due to traumatic or iatrogenic cause, can mimic a primary intestinal tumor in cases of an implant detected along the serosal surface of the bowel.
Pneumatosis intestinalis
Pneumatosis intestinalis (PI) is a radiological finding that refers to gas in the subserosal or submucosal layers of the GI tract. , The etiology commonly is associated with serious underlying pathology, which may require immediate surgical intervention but also can be seen with benign conditions requiring no intervention. With the widespread increase of imaging, the prevalence of PI, in particular benign pneumatosis, is higher than previously thought. Given that incidental findings can prompt additional diagnostic and therapeutic intervention in some cases, which can be harmful to the patient, it is important to understand the differential diagnosis and associated clinical significance of PI. Identifying which patients can be managed conservatively and which require urgent intervention requires an understanding of the various etiologies of PI, assessment of the clinical context, and the recognition of associated radiological findings.
PI is classified into primary PI and secondary PI. Primary PI, or pneumatosis cystoides intestinalis (pneumatosis coli), is less common (15%), is idiopathic, and has the morphology of well-circumscribed cysts or bubbles within the walls of the bowel. It has an incidence of approximately 0.03%. A majority of cases (approximately 85%) of PI are secondary to an underlying disease process, including mesenteric ischemia, bowel necrosis, trauma, inflammatory bowel disease (IBD), malignancy, autoimmune conditions (scleroderma, dermatomyositis), infection ( Clostridium difficile , human immunodeficiency virus, or cytomegalovirus), chronic obstructive pulmonary disease, postoperative changes (laparotomy, laparoscopy, peritoneal dialysis, and so forth), or medications (corticosteroids, immunosuppressants, and chemotherapeutic agents). , , The pathogenesis of PI is not fully understood but likely is multifactorial and involves the disruption of mucosal integrity and the migration of gas either via direct dissection into the wall or through translocation of gas-producing bacteria from the lumen into the submucosal space, and/or increased thoracic pressure, causing alveolar rupture and gas diffusion via perivascular or perilymphatic routes. ,
Benign PI appears as intramural cystic/bubble-like gas collections on CT ( Fig. 8 ) is often and asymptomatic. Studies have shown that benign PI often is confined to the right colon. As a result, patients with PI confined to the right colon, without signs of peritonitis or sepsis, in the appropriate clinical context, and in the absence of worrisome imaging findings (discussed later) can be treated with supportive care. A theory proposes that immunosuppressive or steroid therapies may induce lymphoid depletion in Peyer patches, which impairs the GI defense mechanism, reduces peristalsis, and compromises the intestinal wall integrity resulting in PI. , An association with tapering and discontinuation of steroids has been associated with improvement of PI.