Renal masses are commonly encountered on cross-sectional imaging examinations performed for nonrenal indications. Although most can be dismissed as benign cysts, a subset will be either indeterminate or suspicious; in many cases, imaging cannot be used to reliably differentiate between benign and malignant masses. On-going research in defining characteristics of common renal masses on advanced imaging shows promise in offering solutions to this issue. A recent update of the Bosniak classification (used to categorize cystic renal masses) was proposed with the goals of decreasing imaging follow-up in likely benign cystic masses, and therefore avoiding unnecessary surgical resection of such masses.
Key points
- •
Incidental renal masses and other processes are commonly encountered on routine cross-sectional imaging examinations. Most will be benign cysts; a minority are indeterminate and will require a dedicated renal mass imaging. Renal cell carcinoma (RCC) is now most commonly discovered as an incidental imaging finding.
- •
Changes to the existing Bosniak classification for cystic renal masses in the 2019 proposed revision include incorporation of MR imaging findings, eliminating the need for further imaging in likely benign cystic masses, and definitions of structural components within a cyst.
- •
Despite gains in knowledge regarding imaging features of renal masses, there are still no reliable criteria to our knowledge that can be used to determine whether a soft tissue mass in the kidney is benign or malignant, with the differential diagnosis including RCC, lipid-poor angiomyolipoma, and oncocytoma. Machine learning with attention to radiomics shows promise in identifying features that can provide a more specific and accurate diagnosis.
- •
Intravenous contrast-enhanced computed tomography (CT) and MR imaging continue to be the primary modalities for characterization of the indeterminate or suspicious renal mass; however, other modalities will likely take on a greater role in renal mass imaging, including contrast-enhanced ultrasound and dual-energy CT.
Introduction
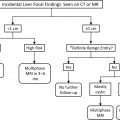
Incidental findings on abdominal cross-sectional imaging examinations are frequently encountered in solid organs, bowel, blood vessels, bone, and the abdominal and pelvic wall. The kidney is one of the more common locations for encountering an incidental finding. Most kidney masses and other incidental findings, for example, scarring and calculi, can be either safely ignored or confidently diagnosed on routine cross-sectional imaging. A small percentage will need further imaging with a specific renal mass imaging examination, or referral for urologic management. The purpose of this article is to outline the spectrum of incidental renal masses in the kidney, both common and uncommon, to inform the reader on data and guidelines that can help to diagnose and triage these findings, and to discuss recent developments and future directions in renal imaging.
Commonly encountered renal masses and characterization with imaging
Renal Cysts
Renal cysts are ubiquitous in the adult population. Their prevalence increases with age, with cysts found in 30% to 50% of patients by the seventh decade. Therefore, the most commonly encountered mass in the kidney will overwhelmingly be a benign renal cyst. Mensel and colleagues showed a prevalence on MR imaging of 27%. O’Connor and colleagues showed the prevalence of incidental renal masses on nonenhanced CT to be 17%.
Most simple renal cysts are easily diagnosed on conventional gray-scale ultrasound imaging. The ultrasound criteria for defining a simple, uncomplicated renal cyst include well-defined, thin, smooth walls, increased posterior acoustic enhancement, and no internal debris or septa.
On computed tomography (CT), a simple cyst can be diagnosed in a homogeneous mass of variable size measuring −9 to 20 HU, as long as there is an imperceptible wall, and no septa, nodules, or calcifications. Although in the past such cysts were considered to be incompletely evaluated on nonenhanced CT alone, recent literature supports the presumption that these masses are benign.
A limitation in the diagnosis of small cysts on intravenous (IV) contrast-enhanced CT is “pseudoenhancement.” Pseudoenhancement is an artifact that causes increased HU measurements adjacent to avidly enhancing tissue, such as renal parenchyma. , Pseudoenhancement introduces inaccuracy into the analysis of enhancement, and the potential to misdiagnose a benign, nonenhancing cyst as an enhancing mass. This phenomena may be more frequently encountered on newer generation multidetector CT scanners than on earlier scanners.
A minority (6%) of cysts are hemorrhagic or proteinaceous. Although benign, hemorrhagic cysts may sometimes be difficult to characterize accurately on imaging examinations because of changes in echogenicity (ultrasound), attenuation (CT), and signal (MR).
On ultrasound, hemorrhagic cysts can have a complex appearance due to internal echoes from the presence of blood. Color and spectral Doppler can help in the evaluation of such a cyst by demonstrating lack of blood flow; however, a cystic mass with internal echoes often requires further evaluation with IV contrast-enhanced CT or MR imaging.
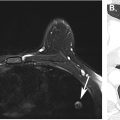
The increased attenuation of a hemorrhagic cyst on CT often overlaps with that of a soft tissue mass, including renal cell carcinoma (RCC); however if sufficiently dense enough, it can be confidently diagnosed as a benign cyst on nonenhanced CT alone. Jonisch and colleagues in a study of 105 patients, 54 with renal cell carcinoma and 51 with high attenuation cysts showed a specificity of greater than 99.9% in the diagnosis of a hemorrhagic cyst in a homogeneous small renal mass uniformly measuring 70 HU or greater on nonenhanced CT ( Fig. 1 ).

MR imaging can be used to accurately diagnose simple cysts, even subcentimeter cysts, by the presence of uniform high T2 signal equivalent to other fluid-containing structures. Nonenhanced MR imaging also has utility for diagnosing a hemorrhagic or proteinaceous cyst. In a study looking at unenhanced T1-weighted MR images in 134 patients, 84 with hemorrhagic cysts and 50 with RCC, Davarpanah and colleagues found that a homogeneous hyperintense renal mass with a smooth border and signal intensity ratio 2.5× greater than renal parenchyma was likely to be a benign hemorrhagic cyst, with a confidence level greater than 99.9% ( Fig. 2 ).

Renal Cell Carcinoma
RCC now most often comes to attention as an incidental finding on abdominal cross-sectional imaging performed for other purposes. Clinical presentation with signs and symptoms of RCC is becoming less frequent.
Eighty percent of RCCs are clear cell carcinomas (ccRCC). ccRCC has a more unfavorable prognosis than other RCC subtypes, with a higher frequency of metastatic disease and decreased survival. ccRCC is a hypervascular tumor on imaging, and typically enhances rapidly and intensely, especially in the corticomedullary phase ( Fig. 3 ). , On MR imaging, ccRCC frequently demonstrates high T2 signal, which is often heterogeneous because of necrosis, cystic degeneration, hemorrhage, or a combination of these findings. Most ccRCCs will show drop out on chemical-shift imaging due to the presence of microscopic fat ( Fig. 4 ).


The second most common renal malignancy is papillary renal cell carcinoma (pRCC), comprising 10% to 15% of RCCs, including a typically indolent and slow-growing Type 1 pRCC and an aggressive Type 2. pRCCs are typically hypovascular, demonstrating weak enhancement on CT and MR imaging, which can lead to the potential pitfall of misdiagnosis as a hemorrhagic or proteinaceous cyst on CT in a minority of cases. pRCC tends to have lower T2 signal intensity than other RCC subtypes due to intratumoral hemorrhage and papillary architecture ( Fig. 5 ).

Chromophobe RCC (chrRCC) is the third most common RCC subtype, accounting for approximately 5% of RCC, and has the most favorable prognosis. chrRCC has enhancement kinetics that are intermediate relative to ccRCC and pRCC ( Fig. 6 ).

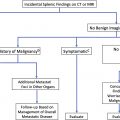
Malignant Renal Masses Other than Renal Cell Carcinoma
Malignant masses not of primary renal origin include transitional cell carcinoma, metastatic disease, and secondary lymphoma.
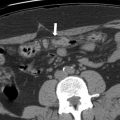
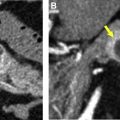
Transitional cell carcinoma (TCC) accounts for 10% of renal masses, and can mimic a centrally located RCC, especially when most of the tumor involves the renal parenchyma rather than the collecting system. Infiltrative TCCs tend to preserve the renal contour ( Fig. 7 ), although less commonly can present as an eccentric tumor distorting the renal contour similar to RCC.

Renal metastases account for only 0.9% of renal masses, but when they do occur they are mostly carcinomas (80.8%), typically solitary (77.5%), with lung cancer being the most common primary site. ,
Renal lymphoma usually occurs in the setting of widespread nodal or extranodal lymphoma, has various presentations, and can be unilateral or bilateral, as well as solitary, multiple, or diffuse and ill defined.
Benign Solid Renal Tumors
A limitation of all imaging modalities is the inability to differentiate RCC from a benign solid soft tissue attenuation renal mass. Approximately 40% of solid renal tumors smaller than 1 cm, and approximately 20% of tumors smaller than 3 cm, will be of a benign histology. The most commonly encountered benign solid masses are angiomyolipoma (AML) and oncocytoma, which represented 44% and 35% of resected benign renal masses in one surgical series, respectively.
AMLs are benign tumors composed of fat, smooth muscle, and vascular tissue in varying proportions. Most AMLs will contain fat in sufficient quantity for detection on CT and MR imaging, allowing for definitive diagnosis. Macroscopic fat is only rarely seen in RCC, and are often associated with calcifications, which are seldom seen in AMLs. Four percent to 5% of AMLs contain either minimal (3%–10% mature fat microscopically) or no fat, and may resemble a nonspecific solid soft tissue mass on cross-sectional imaging (lipid-poor AML).
An AML may be diagnosed by direct visualization of fat on CT ( Fig. 8 ), or on MR imaging by loss of signal on frequency selective fat-suppressed sequences ( Fig. 9 ), or the presence of an “India ink” artifact between the mass and normal renal parenchyma ( Fig. 10 ). Loss of signal on out-of-phase (OOP) images may be seen in an AML due to small areas of fat within the tumor; however, this finding is not useful in differentiating from RCC, as OOP dropout can be seen in all renal cortical tumors, most frequently in ccRCC due to intracellular lipid. , More than 20% loss of signal intensity in a nodule without macroscopic fat has been shown to have high specificity for ccRCC. , However, individual pixel analysis of CT images is not useful for making the diagnosis, as ccRCC has been shown to have more pixels in the negative HU range than AML.



Lipid-poor AMLs have attenuation similar to skeletal muscle on nonenhanced CT, and are more hyperattenuating than most RCCs; however, there is overlap between the density measurements of lipid-poor AMLs with chromophobe and other subtypes of RCC. Low T2 signal on MR imaging is a feature suggestive of an AML, but is also commonly seen in papillary RCC. Therefore, although such findings are suggestive of a lipid-poor AML, they do not obviate the need for biopsy.
Oncocytomas account for approximately 7% of renal masses, and more than half of all benign solid renal tumors smaller than 4 cm. These tumors share a common progenitor cell with chrRCC, and therefore have overlapping histologic and imaging features. Characteristic imaging features of oncocytoma include peripheral location, lack of calcifications, well-circumscribed borders, and heterogeneous T2 signal on MR imaging. Enhancement is typically less than renal cortex at all phases of dynamic contrast-enhanced imaging. Findings of a central stellate scar, seen in 50% to 61% of oncocytomas, typically with associated spoke-wheel enhancement, can suggest the diagnosis, but can also be seen in RCC, particularly in chrRCC. , Segmental contrast inversion, an interesting feature whereby portions of a tumor that enhance avidly on early-phase imaging washout on later phases, whereas areas that enhance weakly on early phase show increased enhancement on delayed images ( Fig. 11 ), can be seen in oncocytomas, although the frequency of this observation greatly varies in the literature, and segmental contrast inversion can also be seen in chrRCC. ,

Despite the preceding advances in defining imaging characteristics of minimal-fat AML and oncocytomas, these tumors are currently unable to be reliably differentiated from RCC with any imaging modality, to our knowledge, and require either biopsy or surgical removal for definitive diagnosis.
Cystic Renal Masses
Cystic renal cancers account for 3% to 14% of renal cell carcinoma. There is overlap in the imaging appearance of benign and malignant cystic renal masses, with the differential diagnosis including cystic degeneration of a usually solid RCC subtype, multilocular cystic renal cell carcinoma, multilocular cystic nephroma, mixed epithelial stromal tumor, hemorrhagic cysts, and atypical cysts, all of which may contain thickened, nodular septations, enhancing nodules, or a combination of these findings. ,
Although portions of a cystic malignancy may measure less than 20 HU, in most cases cystic tumors will demonstrate some degree of heterogeneity, allowing for differentiation from a simple cyst. Rarely, necrosis within a papillary RCC may be extensive enough that it may resemble a cyst on imaging ( Fig. 12 ). Corwin and colleagues found that 2.8% of papillary RCCs were found to be homogeneous and less than 20 HU in attenuation on nonenhanced CT ( Fig. 13 ). Fifteen percent of clear cell carcinomas may have cystic components, which can be due to multilocular cystic growth, origin from the wall of a preexisting cyst or cystic necrosis. Multilocular cystic renal carcinoma is a subtype of clear cell RCC and is a true cystic neoplasm, which is defined by strict pathologic criteria. These tumors exhibit benign behavior, and distant metastases have seldom, if ever, been reported, to our knowledge. , Regardless of the exact tumor histology, cystic renal cancers tend to have less aggressive tumor biology and significantly improved survival compared with solid RCC. , , ,


Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree