, Valery Kornienko2 and Igor Pronin2
(1)
N.N. Blockhin Russian Cancer Research Center, Moscow, Russia
(2)
N.N. Burdenko National Scientific and Practical Center for Neurosurgery, Moscow, Russia
In men, stomach cancer is detected two times more often than in women. According to IARC, the maximum incidence of stomach cancer was observed in men in Japan (Kutuki et al. 1996; Fan et al. 2004). In Russia, SC is second only to lung cancer, 48.8 thousand new cases of gastric cancer are reported annually, and 45,000 patients die from this disease each year. According to Davydov and Axel (2014), mortality from gastric cancer is one of the highest in Russia, ranking second among men (12.0%) after lung cancer and in women (10.0%) after breast cancer. Patients with SC have lower survival rates as compared to lung cancer, breast cancer, and kidney cancer (Go et al. 2011). In Western Europe and the USA, the 5-year survival rate ranges within 25%. In Japan, known for success in the treatment of gastric cancer, the figure is 66% among patients with primary gastric cancer and 68.2% among patients who underwent radical surgery (Verlato et al. 2012).
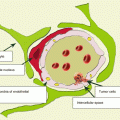
Most stomach carcinomas are adenocarcinomas and signet ring cell carcinomas. In 75% of newly diagnosed patients, the disease is identified in stages III–IV; the frequency of detection of early forms of gastric cancer does not exceed 10–20%. One of the potentially important markers that reflect the activity of metastases of stomach cancer is nm23 protein (nonmetastatic cell protein) (Ji et al. 2002; Lee et al. 2003).
The final destination of regional metastatic gastric cancer, regardless of the tumor site, is a para-aortic lymph collector. Even with a small depth of tumor invasion into the stomach wall, for example, to the level of its muscle membrane or submucosal layer, the frequency of lymphogenic metastases may increase from 15 to 40% (Lewis et al. 2008).
Stomach cancer metastasizes to the brain even more often than colorectal cancer. Kim (1999) noted the development of metastatic brain lesions in only 13 (0.2%) of 8080 patients with gastric carcinoma. In turn, York (1999) reported a 0.7% frequency of SC metastases to the brain in 23 of 3320 patients. Gastric cancer patients with brain metastases amount to not more than 1% of all cases of brain metastases, both according to autopsy findings and clinical manifestations (Zimm et al. 1981; Graf et al. 1988). Metastases in the brain develop 1–23 months after the diagnosis of gastric cancer, with the overall survival of patients of about 9 weeks. In our study, the proportion of stomach cancer metastases in the brain was 0.4% of all metastatic lesions.
There are no specific radiological signs that would allow to differentiate gastric cancer metastases in the brain from secondary tumors from other primary sites.
17.1 Clinical Cases
See Figs. 17.1, 17.2, 17.3, 17.4, 17.5, 17.6, 17.7, 17.8, 17.9, 17.10, and 17.11.
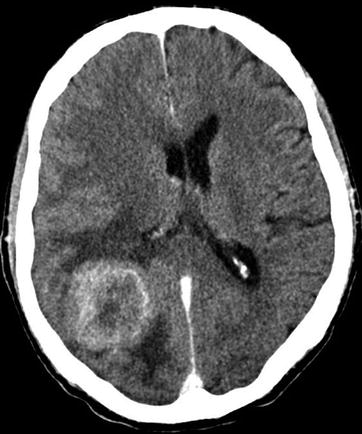
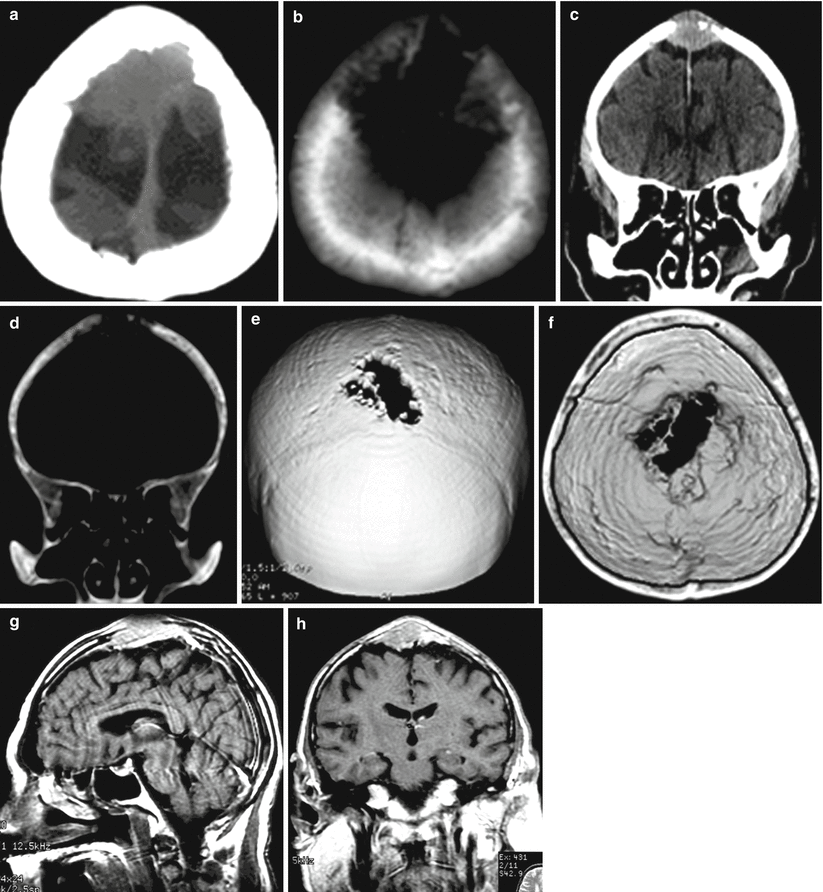
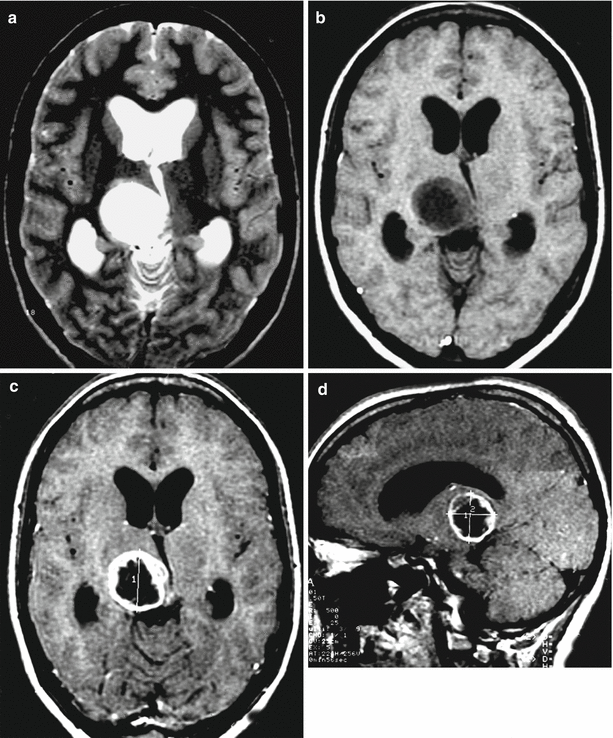

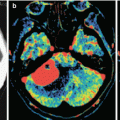
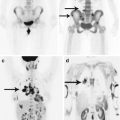
Fig. 17.1
A stomach cancer metastasis in the brain. On axial CT after intravenous CA administration, in the right parietal-occipital region, there is a large heterogeneous space-occupying lesion with an intense and uneven contrast media accumulation mainly on the periphery, with signs of a pronounced perifocal edema in the form of low-density areas (ring enhancement). The lateral ventricles are deformed

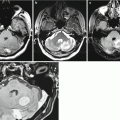
Fig. 17.2
A stomach cancer metastasis in the brain. A space-occupying lesion with an irregular shape, unclear contours, and intra- and extracranial spread, with the involvement of the dura mater and the superior sagittal sinus, is identified in the posterior part of the squama of the frontal bone. On CT in the axial (a, b) and coronary (c, d) projections, as well as on the 3D reconstruction (e, f), a portion of osteodestruction is determined. After intravenous CA administration on CT (c) and T1-weighted MRI in the sagittal (g) and coronary projections (h), there is intense accumulation of the contrast agent in the soft tissue of the metastasis. The metastasis grows into the superior sagittal sinus

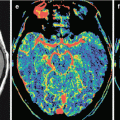
Fig. 17.3




A stomach cancer metastasis in the brain. In the projection of the right thalamus, there is a rounded space-occupying lesion with signs of central necrosis. On T2-weighted MRI, the lesion has a high signal (a); there is no perifocal edema. On T1-weighted MRI (b), the solid part of the metastasis has a moderately low signal, while its cystic part has a hypointense signal. After CA administration (c, d), the solid part of the tumor is intensely contrast enhanced
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree