Testicular
Acute testicular pain represents about 0.5% of ED complaints.1 Causes of acute testicular pain include trauma, epididymitis, orchitis, torsion of the testicular appendage, and hernia; however, testicular torsion is the diagnosis of the greatest concern in the emergency setting.
The traditional teaching was that most patients presenting to an ED or urgent care with a complaint of acute testicular pain had testicular torsion.2 This misconception has been dispelled and it is now known that the most common etiology of acute testicular pain is epididymitis.2 However, the evaluation of acute testicular pain presents a considerable challenge for emergency providers, since 50% of patients presenting with testicular torsion have delayed seeking care for >6 hours and are at high risk of losing the torsed testicle.3
The issue of acute testicular pain is further complicated by the high potential for litigation associated with infertility after testicular loss due to torsion or disruption of the testicle from severe trauma. When the diagnosis of testicular torsion is missed, the majority of patients have been incorrectly diagnosed with epididymitis.4
 CLINICAL CONSIDERATIONS
CLINICAL CONSIDERATIONS
High-resolution color Doppler ultrasonography has become widely accepted as the test of choice for evaluating acute scrotal pain, replacing scintigraphy in most institutions.5 While scintigraphy requires less technical skill on the part of the radiologist consulted to evaluate the patient, there are major drawbacks to the technique. Scintigraphy is a time-consuming process that can add an hour or more to the evaluation of a patient who may already be several hours into the testicular torsion process.5 Furthermore, the resultant hyperemia of the scrotal skin during testicular torsion can mask a lack of blood flow to the testicle itself and lead to a misdiagnosis in less experienced hands.6 This nuclear medicine study also provides no information regarding testicular anatomy, which is a critical issue if pathology other than torsion is present. MRI is a promising imaging modality for detecting acute scrotal problems, including ischemia caused by torsion. However, MRI is expensive and time consuming.
Traditionally the history and physical examination were thought to be the keys to diagnosing or ruling out testicular torsion.3 However, the historical features of several disease processes may be similar. For example, the duration of pain in testicular torsion, epididymitis, orchitis, and torsion of a testicular appendage frequently overlap.7 Also, only 50% of patients with torsion have sudden onset of pain and about 20% of have pain associated with trauma or physical exertion, such as heavy lifting.8,9 Adding to the difficulty using the history is the fact that many young men do not provide an accurate history of trauma. The physical exam can be similarly misleading, because it is frequently limited by pain, edema, and patient compliance. In addition, findings such as absent cremasteric reflex, abnormal testicular lie (present in <50% of testicular torsion cases), and epididymal tenderness are not reliable in differentiating torsion from other causes of acute scrotal pain.10
The classic clinical features of epididymitis, such as dysuria and urethral discharge, are not consistent. Up to 50% of patients with acute epididymitis do not have dysuria or urethral discharge.4 Furthermore, many patients with epididymitis complain of acute onset of pain. This is probably due to poor recall or the need for a threshold of discomfort to be crossed prior to the patient’s recognition of pain. Another complicating factor is that if the body or tail of the epididymis is inflamed without affecting the head, the pain and swelling will be in a different location from where most clinicians expect with epididymitis. Most clinicians learn to feel for the head of the epididymis on their physical examination and do not realize that it may not be involved. Patients with testicular torsion who are initially misdiagnosed with epididymitis are most commonly misdiagnosed because of the presence of dysuria, pyuria, or urethral discharge, or because of a vague history of pain onset, giving the impression of a less acute process.1 Because of the time-sensitive nature of testicular torsion, ultrasound is an ideal diagnostic test for the evaluation of testicular pain.
 CLINICAL INDICATIONS
CLINICAL INDICATIONS
The clinical indications for point-of-care testicular ultrasound are as follows:
 Acute testicular pain
Acute testicular pain
 Acute scrotal mass
Acute scrotal mass
 Trauma
Trauma
ACUTE TESTICULAR PAIN
The most serious cause of acute testicular pain is testicular torsion. Testicular torsion can occur at any age; however, the majority of cases occur between 12 and 18 years of age. The most common underlying cause of testicular torsion is a congenital malformation known as bell clapper deformity, where the tunica vaginalis completely encircles the epididymis, spermatic cord, and testis instead of attaching to the posterolateral aspect of the testis. This allows the testicle to twist about its own axis compressing the vasculature and resulting in ischemia. The bell clapper deformity is most often found to be bilateral (50–80% of cases) upon surgical exploration.11 Further increasing the likelihood of testicular torsion is a history of an undescended testicle.12 Once blood flow is interrupted to the testicle, infarction and loss of the testicle can occur quickly. Testicular salvage rates are approximately 100% prior to 3 hours, 83–90% at 5 hours, 75% at 8 hours, and 50–70% at 10 hours. When a testicle has been torsed for longer than 10 hours, the rates of salvage decrease to 10–20%. After 24 hours, salvage of a testicle is rarely seen unless there has been intermittent detorsion.13 In general, urologists are reluctant to take patients to the operating room and explore a painful scrotum based solely on the clinical examination. Studies argue that such patients should not be routinely explored and that accurate diagnosis can be provided with Doppler ultrasound of the involved testicle.14
For the detection of acute testicular torsion, ultrasonography has a sensitivity of 80–98% and a specificity of 97–100%.15 Data regarding testicular ultrasound use by clinicians at the bedside are relatively limited.16–21 Testicular ultrasound carries a stigma of being a difficult and advanced level examination that will lead to dire consequences if misinterpreted, but the data do not support this notion. One study evaluated 36 patients with acute testicular pain who underwent point-of-care testicular ultrasound by an emergency physician, and demonstrated a sensitivity and specificity of 95% and 94%, respectively, for testicular torsion when surgical follow-up and radiology imaging were used as the criterion standard.16 All patients with testicular torsion were correctly identified. Other diagnoses found included epididymitis, orchitis, hemorrhage, and herniation. Another study demonstrated good accuracy of point-of-care ultrasound for a variety of testicular pathology encountered in the emergency setting.21
Patients at institutions without 24/7 radiology ultrasound capabilities will benefit the most from clinician-performed point-of-care testicular ultrasound. Also, when clinicians can quickly confirm lack of blood flow to a painful testicle they are more likely to attempt manual detorsion, which may significantly improve testicular salvage rates.
A major limitation for clinicians attempting to gain experience with testicular ultrasound is the infrequency of patients presenting with acute scrotal pain. However, one study demonstrated that testicular torsion could be easily simulated for training purposes by digital compression of the spermatic cord. This technique holds great promise as an educational tool that could quickly increase clinician confidence and expand the use of point-of-care testicular ultrasound.19
ACUTE SCROTAL MASS
Painless scrotal masses are unlikely to represent an acute disease process. Patients often describe that the involved testicle has been slowly enlarging for some time, but they had simply ignored it or grew accustomed to the difference until some factor finally made them seek medical assistance. Firm, nontender testicular masses commonly represent neoplasms that should be directed for expeditious follow-up with a urologist. When the patient presents complaining of pain from the testicular mass, it may be from hemorrhage into the neoplasm itself. The vast majority of soft, nontender testicular masses are hydroceles that may develop because of a number of disease processes or idiopathically. Many are congenital and result from a direct communication with the abdominal cavity. Hydroceles may result from trauma, infection, neoplasm, radiation therapy, and undiagnosed torsion.22 Hernias can present with complaints of acute scrotal mass, and are generally painful. The presentation can be similar to that of torsion if the scrotum is erythematous.
TRAUMA
Blunt trauma can lead to injury of the testicle or associated structures. Assault, athletic injury (about 50% of blunt trauma cases), bicycle crashes, and motor vehicle crashes are the most common causes of blunt testicular trauma.23 Injuries can be in the form of laceration, hemorrhage, or contusion of the testicle. Clinical features of testicular injury are a tender swollen testicle, often accompanied by ecchymosis. Accurate diagnosis by physical examination alone is often difficult secondary to marked swelling and pain in the traumatized scrotum, thus making ultrasound imaging a requisite. The goal of point-of-care ultrasound in patients presenting with acute trauma to the scrotum is to determine whether the testicle is injured and requires operative intervention. Although a contusion or focal hemorrhage may simply require follow-up, fracture through the capsule of the testicle necessitates surgical intervention. Furthermore, since trauma can lead to testicular torsion, blood flow should be evaluated as well. Ultrasound is sensitive for detecting major trauma to the testicle and, therefore, can be useful for screening patients. While the accuracy of ultrasound is operator dependent, the diagnosis of a significant testicular injury is relatively straightforward.24,25
 ANATOMICAL CONSIDERATIONS
ANATOMICAL CONSIDERATIONS
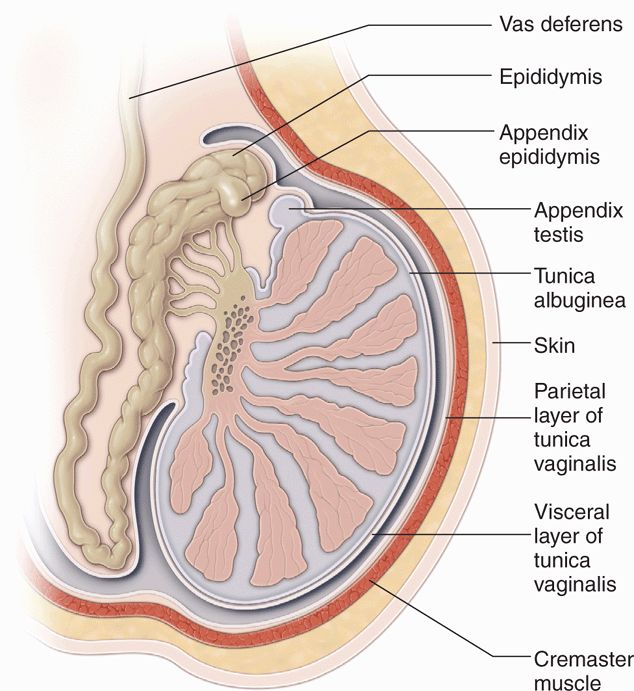
The scrotum is divided into two separate compartments by a midline septum called the median raphe. Each compartment of the scrotum contains a testis, epididymis, vas deferens, and spermatic cord. The normal adult testes are located in the scrotum and are oval in shape. Average testicular measurements obtained by ultrasound are 4 cm × 3 cm × 2.5 cm. Each adult testicle weighs between 10 and 19 grams. The size of the testicle changes with age; it increases in size up to puberty and decreases later in life. The testis is surrounded by a fibrous capsule known as tunica albuginea, which is covered by tunica vaginalis. The tunica vaginalis has two layers: the outer parietal and inner visceral, which merge at the posterolateral aspect of the testis (Figure 13-1). These two layers are separated by small amount of fluid. The tunica albuginea projects posteriorly into the testis to form an incomplete septum called mediastinum testis.
Multiple septations arise from the tunica albuginea and run through the testicle. These septations result in the separation of the testicle into multiple lobules. The testicular parenchyma is made up of numerous seminiferous tubules that converge toward mediastinum testis and open into rete testis, which drains into the epididymal head.26,27
The epididymis is a tubular structure located along the posterolateral aspect of each testis. It measures approximately 6–7 cm in length and consists of a head, body, and tail. The head of the epididymis lies superolaterally to the testis, the body adjacent to the posterolateral margin of the testis, and the tail at the inferior pole of the testis. The tail of the epididymis turns into the vas deferens as it travels superiorly out of the scrotum. The vas deferens, in turn, travels in the spermatic cord. The spermatic cord contains a number of structures, including the testicular artery, cremasteric artery, deferential artery, pampiniform venous plexus, lymphatic structures, nerves, and vas deferens. The appendix testis and the appendix epididymis are found toward the superior pole of the testis. The appendix testis, an oval structure about 5 mm in length, usually lies in the groove between the testis and the epididymis. The appendix epididymis is attached to the head of the epididymis and is of the same approximate dimensions as the appendix testis. The appendix epididymis has been identified unilaterally in 34% and bilaterally in 12% of testes.26,27
Most of the arterial blood supply received by a testicle comes from the abdominal aorta through the testicular artery. A small portion of the arterial supply comes from the deferential and cremasteric arteries, which anastomose with the testicular artery. The deferential artery, arising from the vesicular artery, supplies the epididymis and the vas deferens. The epididymis, however, is supplied predominantly by the superior epididymal artery, which is a branch of the testicular artery. The cremasteric artery arises from the epigastric artery and supplies the peritesticular tissues. There is variability in normal anatomy and, in general, anything that appears abnormal should be compared to the contralateral side.26
 GETTING STARTED
GETTING STARTED
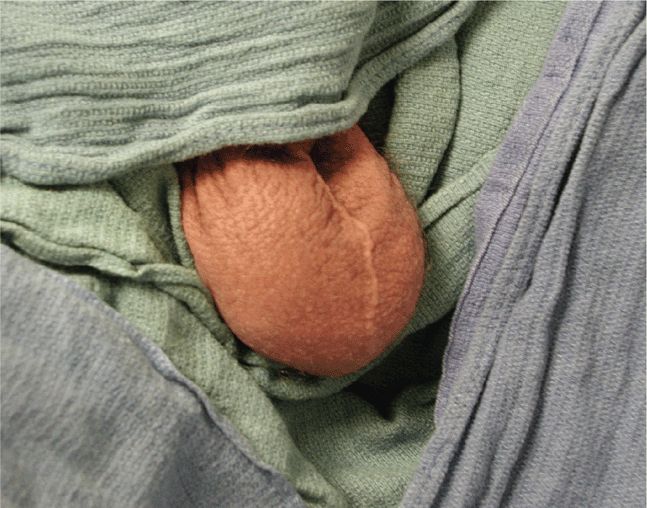
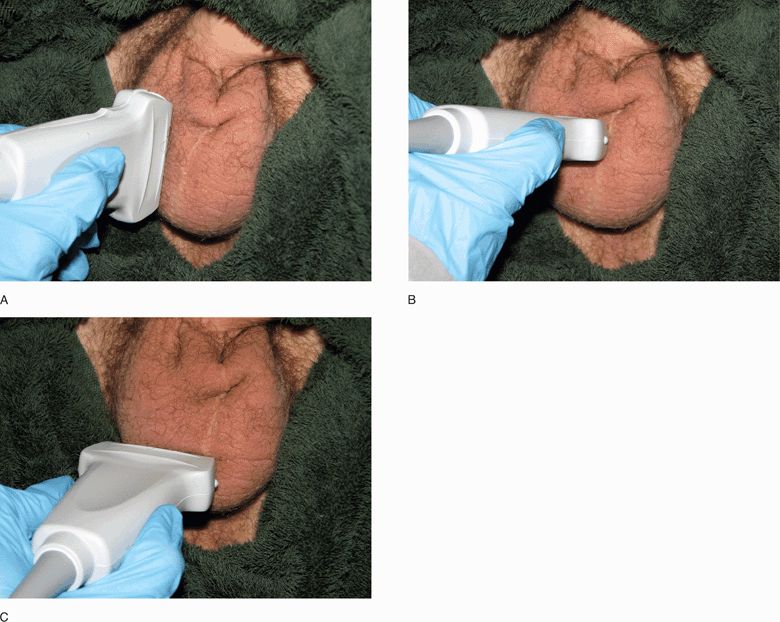
Ensure patient comfort when performing a testicular ultrasound examination. The main issue is having the patient lay still when performing power and pulsed-wave Doppler measurements. Both of these modalities are very sensitive to motion artifact. Thus, provide adequate analgesia and reassurance. Explain the examination details and its goals to the patient. Disrobe the patient from the waist down and scan in the supine position. A frog leg position allows good access to the scrotum and it may help to prop pillows or blankets under each bent knee to allow relaxation of the legs. Place the scrotum in a sling and support it on a rolled towel placed between the patient’s thighs to isolate the scrotum for scanning (Figure 13-2). Position the penis over the suprapubic region and cover it with a towel.
Figure 13-2. A properly exposed and draped patient with the scrotum supported in a sling of towels for improved patient comfort and visualization.
Place the ultrasound machine on the patient’s right. Use a generous amount of warmed ultrasound gel. Cold gel may make the patient more uncomfortable by eliciting a cremasteric reflex. In cases of significant inflammation or torsion, activation of the cremasteric reflex may cause considerable pain. Cold gel can also result in contraction and thickening of the scrotal skin, making it difficult to see scrotal contents. When evaluating the testes, select a high-frequency (7.5–10 MHz) broadband linear transducer, or a 5.0 MHz transducer for large masses or very edematous scrotum (Figure 13-3). The resolution provided by high-frequency linear transducers is crucial for examining not only the parenchyma of the testicle but also the blood flow within it. Some clinicians will even utilize 12–15 MHz transducers to obtain the best Doppler resolution. This may not be possible if significant swelling exists and the testicle itself is several centimeters from the skin surface. Select a small part or testes preset on the machine. In addition to a high-resolution transducer, color, power and spectral Doppler modes are required when evaluating a patient for torsion. Power Doppler, a nondirectional version of color Doppler, is highly sensitive for detecting vessels with low blood flow, and its signal is independent of Doppler angle correction. The disadvantages of power Doppler are its susceptibility to motion and omission of directional information. Power Doppler is used for evaluating testicular perfusion, where blood flow tends to occur through relatively small diameter vessels and at lower speed. Knowing direction of blood flow is of little help in most cases of testicular ultrasound. Pulsed-wave Doppler is also an important component of the evaluation especially when incomplete torsion is present. It allows for documentation of both venous and arterial blood flow within the testicle.
Figure 13-3. An example of a high-resolution linear array transducer that can be used for testicular sonography.
Power Doppler activation will bring up a box-like window the size of which may or may not be adjustable (Figure 13-4). This box can be moved around the screen to the portion of the testicle or scrotum of interest. If the size of the power Doppler box or window is adjustable, it may or may not be helpful to enlarge it, so that it covers the entire testicle. The drawback of enlarging the color box is that this decreases the frame rate, which may make it more difficult to detect higher velocity arterial blood flow. Typically, a balance has to be achieved between a power Doppler window that is too small, requires frequent movement and does not capture the overall blood flow of the testicle, and a window that is too large and does not allow the machine to refresh the image frequently enough. Simply turning on the power Doppler box may not be adequate if the presets on the machine are not set correctly. Adjusting the sensitivity setting as low as possible without having power Doppler signal everywhere will allow for more accurate detection of blood flow.
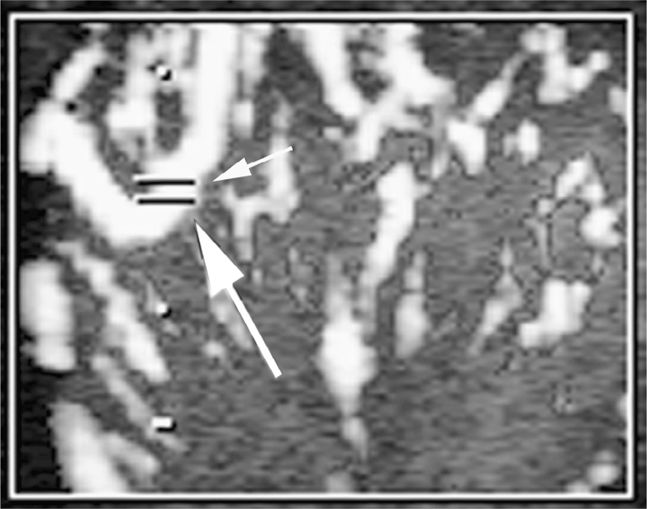
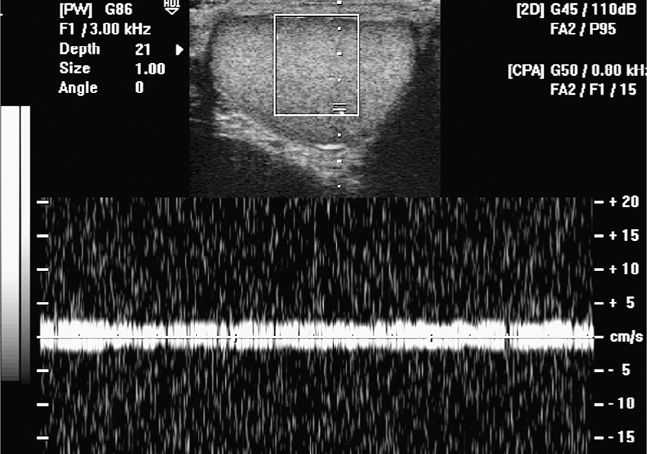
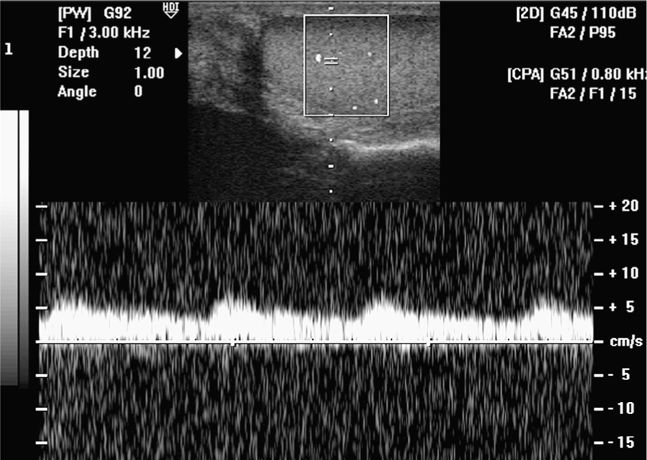
Once reliable power Doppler signals are visualized in the unaffected testicle, use the pulsed-wave Doppler. A small gate, often two parallel lines, will appear on the screen (Figure 13-5). It is guided with a trackball or touch pad to the power Doppler signals inside the power Doppler box. Typically, an update button will have to be pushed (or the pulsed-wave button may have to be pushed a second time) when the parallel lines are placed over the power Doppler signal. Once activated, a tracing will appear showing the pulsed-wave Doppler signal. Adjust the size of the sampling gate, otherwise a poor waveform will be obtained. Turning up the pulsed-wave gain is acceptable to a point where the background around the graph baseline becomes brighter and shows some noise. This will allow the exam to be more sensitive for detecting pulsed-wave venous and arterial flow. Venous flow in the testis is typically continuous with minimal variation in velocity (Figure 13-6). Arterial flow to the testes has pulsativity, with a low-resistance, high-flow waveform pattern (Figure 13-7). Sampling should occur in multiple areas in the testicle. Once the settings have been properly adjusted on the unaffected side, move the transducer to the painful testicle. If the machine’s settings are identical to those used for the unaffected testicle, the clinician will quickly appreciate if blood flow in the affected testicle is similar, decreased, or absent. In addition, gray scale comparison can quickly be made between the two testicles (Figure 13-8). A side-by-side comparison of the two testes can be helpful and illustrates any difference in power Doppler blood flow or echogenicity in a single image
Figure 13-6. A pulsed-wave Doppler tracing is obtained from a testicle. A typical venous waveform is shown.
Figure 13-7. A pulsed-wave Doppler tracing is obtained from a testicle. A typical arterial waveform with a systolic and diastolic flow component is shown.
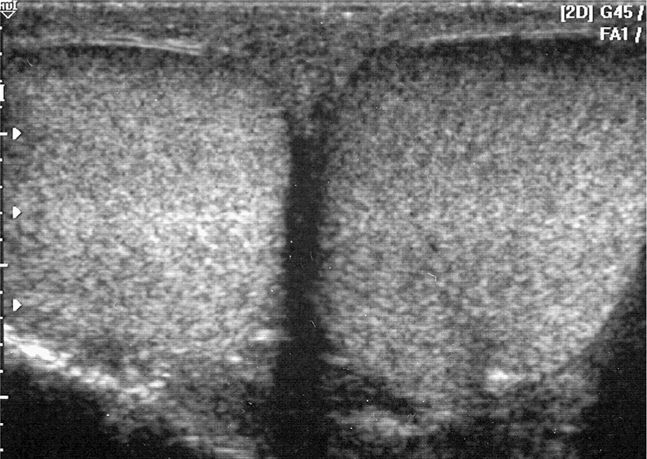
Figure 13-8. A side-by-side comparison image is shown. Both testicles are scanned at once with one linear transducer.
 TECHNIQUE AND NORMAL ULTRASOUND FINDINGS
TECHNIQUE AND NORMAL ULTRASOUND FINDINGS
Scan the scrotum and its contents in two orthogonal planes, along the longitudinal and transverse axes. Scan the asymptomatic side first to familiarize the patient with the process; it also provides a comparison of sonographic anatomy and blood flow. Anchoring the scanning hand to patient’s thigh will keep it from moving unintentionally. Scan the unaffected testicle in the longitudinal plane from medial to lateral (Figure 13-9A). At this point the indicator on the transducer is typically pointed toward the patient’s torso; however, if the testicle has an oblique lie, it is appropriate to orient the indicator toward the upper pole of the testicle. Pay attention to the gray scale abnormalities such as edema, focal masses, or disruptions, and measure the length and thickness of the testes. Evaluate the epididymis in this view. In the long-axis view with the indicator directed cephalad, the epididymis is seen on the left side of the screen. Any epididymal abnormalities will often become evident on the longitudinal views. Turning transverse, still on the unaffected side, scan the testicle from top to bottom, again looking for any gray scale abnormalities (Figure 13-9B and 13-9C). Measure the width in the central portion of the testicle. Scan the spermatic cord from the inguinal canal to the posterosuperior aspect of the testes. After evaluating the testes, assess the scrotal skin thickness and the remaining scrotal contents. Adjust the gain, depth, and resolution settings as appropriate for optimal gray scale image acquisition.
Figure 13-9. A longitudinal orientation to the transducer is shown on the right testicle (A). The transducer is held in transverse orientation of the right testicle and moved from upper pole (B) to lower pole (C).
Perform the Doppler examination after the gray scale evaluation of the scrotum to assess blood flow. Scan the asymptomatic side first to adjust Doppler settings to display low-flow velocities. Orient the transducer in the longitudinal plane of the testicle with the power Doppler feature turned on. Unless the machine presets happen to already be optimized for the testicular examination, some adjustments in gain, wall filter, scale, and pulse repetition frequency (PRF) will have to be made. Low wall filter (100 KHz), low PRF (1–2 Hz), and 70–90% color gain output settings are ideal for testicular scanning. The goal of these adjustments is to demonstrate as much blood flow as possible without introducing more than minimal artifact. Power Doppler is highly sensitive to any movement of the testicle or the clinician’s hand. Once areas of power Doppler signal are visualized, appreciate the amount of blood flow in the testicle. With experience it will be obvious when there is too much blood flow, as in cases of inflammation, or too little blood flow, as in possible ischemia. Even in experienced hands, however, it is prudent to reserve judgment until the two sides can be compared, as long as the other testicle is present for comparison. At this point, activate the pulsed-wave Doppler and place its sampling gate over areas of power Doppler signal until both venous and arterial waveforms are obtained in multiple areas within the body of the testicle. Blood flow seen on the edge of the testicle may be misleading and should not be reassuring. After evaluating testicular perfusion, a complete ultrasound examination of the scrotum includes confirmation of epididymal blood flow using power Doppler and spectral Doppler waveform analysis. Quantification of outflow resistance can be accomplished by determining the resistive index (RI). RI is defined as the peak systolic velocity minus the enddiastolic velocity, divided by the peak-systolic velocity, and is easily calculated by the ultrasound machine’s software.
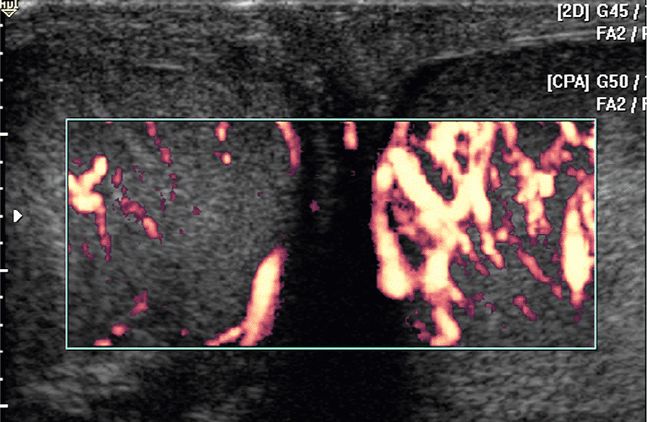
Repeat the entire process on the affected side; however, the examination is typically not finished at this point. In the majority of cases, it is helpful to perform a transverse scan of both testes simultaneously to identify differences in size, echogenicity, and blood flow (Figure 13-10). With the transducer turned horizontal through the midportion of both testes and indicator toward the patient’s right hip, scan the testes from upper to lower pole for comparison in gray scale. Turn on power Doppler to evaluate the testes. This will often mean enlarging the power Doppler box, thus slowing the motion on the screen. This view is ideal for demonstrating any similarities or differences between testes in both gray scale and power Doppler. This is especially illustrative in cases of torsion, with one side showing ample blood flow and the affected side showing no blood flow in the same power Doppler box (Figure 13-11A and 13-11B). Similarly, when one testicle has significantly increased blood flow due to inflammation and the other has normal blood flow, the diagnosis is difficult to dispute when such an image is obtained (Figure 13-12). Despite which order one chooses to perform the individual steps, perform the examination the same way each time for consistency.
Figure 13-10. The transducer is held in transverse orientation across both testicles. This generates a comparison view of both testicles on the screen for gray scale and Doppler evaluation.
Figure 13-11. Power Doppler evaluation (gray scale copy). An equal amount of flow is shown in this comparison view of both testicles with power Doppler (A). In this side-by-side view, no flow is seen in the right testicle, which is displayed on the left side of the screen (B). Torsion was confirmed in the operating room.
Figure 13-12. A side-by-side comparison view with power Doppler is shown. The left testicle, shown on the right side of the screen, has markedly increased flow in this example of orchitis.
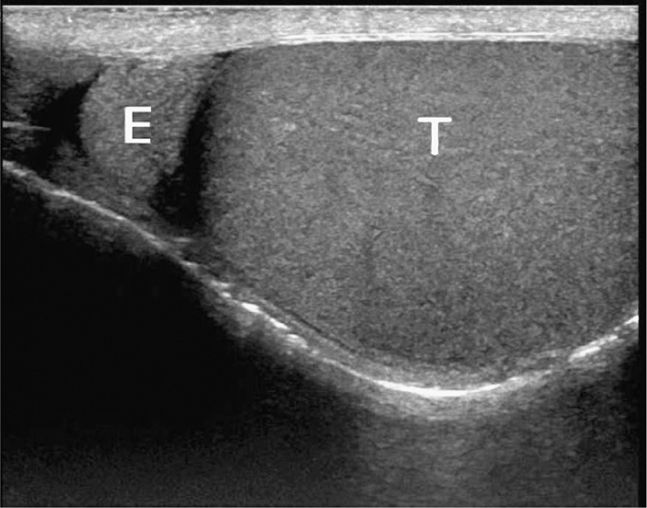
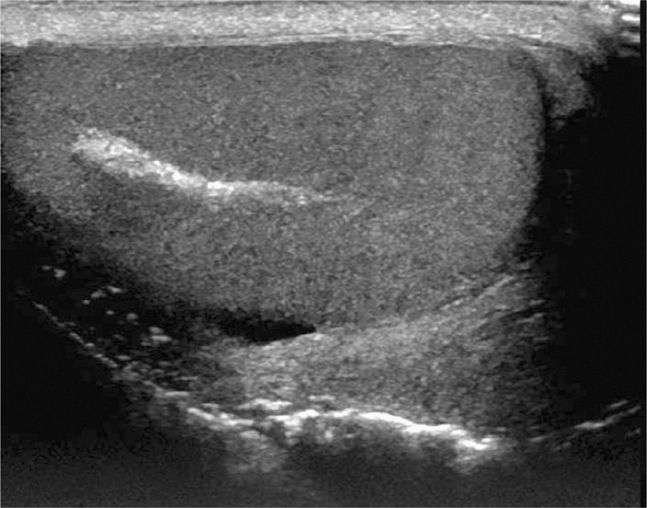
Sonographically, a testicle has mid-gray or medium-level echoes and appears quite homogeneous in echo-texture (Figure 13-13). The echogenicity of the testes is somewhat similar to liver and thyroid tissue. The normal scrotal wall thickness varies between 2 and 8 mm, depending on the state of contraction of the cremasteric muscle.26 The tunica albuginea is seen as one echogenic stripe encircling the testis. The epididymis can be readily differentiated from the rest of the testicle in normal as well as pathologic instances. It has similar echogenicity to the testicle but can appear slightly brighter. While the head of the epididymis is readily seen, the body (2–4 mm in diameter) and tail (2-5 mm in diameter) may be harder to differentiate when no inflammation is present. The mediastinum testis is seen as a linear echogenic band extending across the testes in a craniocaudal direction (Figure 13-14). The thickness and length of mediastinum testis are variable. Adjacent to the mediastinum testis, the normal rete testis is seen as a hypoechoic area with a striated configuration in approximately 18% of patients.26 The appendix testis (5 mm in length) is a small oval structure and is normally hidden by the epididymal head, thus making it nearly impossible to differentiate in normal examinations. On B-mode, the appendix testis appears isoechoic to the testis. If a hydrocele is present, the appendix testis often becomes outlined by the fluid and is seen as a defined structure (Figure 13-15). When torsed, it may cause not only local inflammation but also an epididymitis-like appearance due to diffuse inflammation of the epididymal head. The appendix testes may be located in different areas on the testicle. Additionally, the epididymis can have appendages. The appendix epididymis is approximately the same size as the appendix testis but is usually pedunculated and located at the head of the epididymis (Figure 13-16) Sonographically, the spermatic cord is seen as multiple hypoechoic linear structures in the long axis and circular hypoechoic structures in the transverse axis (Figure 13-17). The normal thickness of the spermatic cord in the inguinal canal is approximately 4 mm. The normal spectral waveform of the intratesticular arteries and arteries supplying the epididymis (deferential and cremasteric arteries) have a low-resistance, high-flow pattern. However, the arteries supplying the scrotal wall have a low-flow, high-resistance pattern. The RI of normal testicular artery ranges from 0.5 to 0.8.15
Figure 13-14. Longitudinal view of the normal testis with an echogenic mediastinum testis extending craniocaudally.
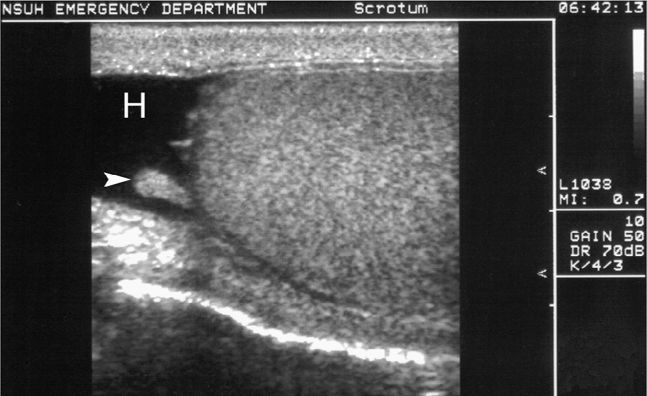
Figure 13-15. A small hydrocele outlines the appendix testis (arrowhead) in this patient presenting for chronic testicular pain. H: hydrocele.
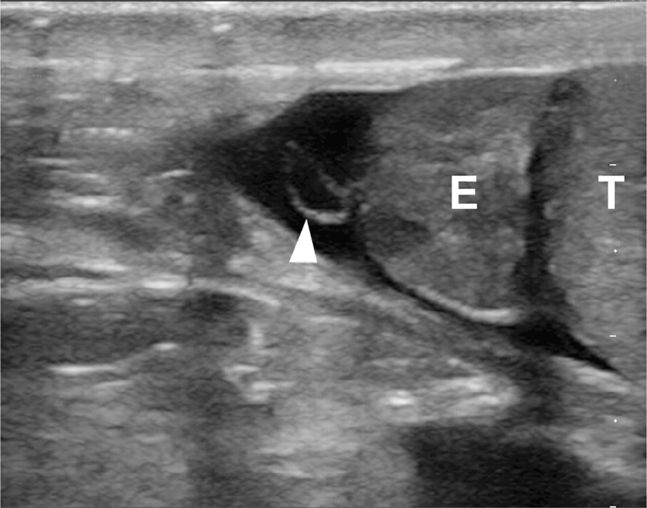
Figure 13-16. An appendix epididymis (arrowhead) outlined by hydrocele is seen adjacent to the head of the epididymis. E = head of the epididymis, T = testicle.
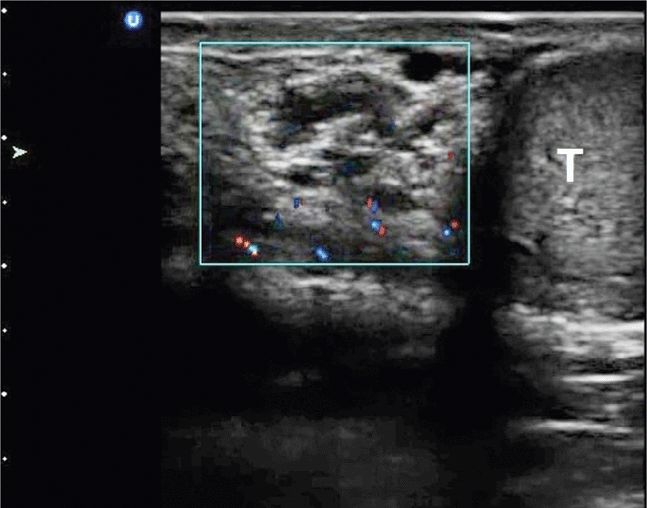
Figure 13-17. Spermatic cord is shown as multiple hypoechoic structures (within the power Doppler box) superior to the testis (T) in the long-axis view.
 COMMON AND EMERGENT ABNORMALITIES
COMMON AND EMERGENT ABNORMALITIES
EPIDIDYMITIS
Epididymitis is the most common cause of scrotal pain in adolescents and adults.28 The disease is insidious in nature; however, a significant proportion of patients present with sudden onset of pain mimicking testicular torsion. Infection of the epididymis or testicle usually results from retrograde spread of bacteria from the bladder or prostate via the vas deferens.28 Another disease process that can present with signs and symptoms of epididymitis in prepubertal males is torsion of the testicular appendage. In fact, it is believed that many cases of appendiceal torsion are misdiagnosed as epididymitis in prepubertal males.
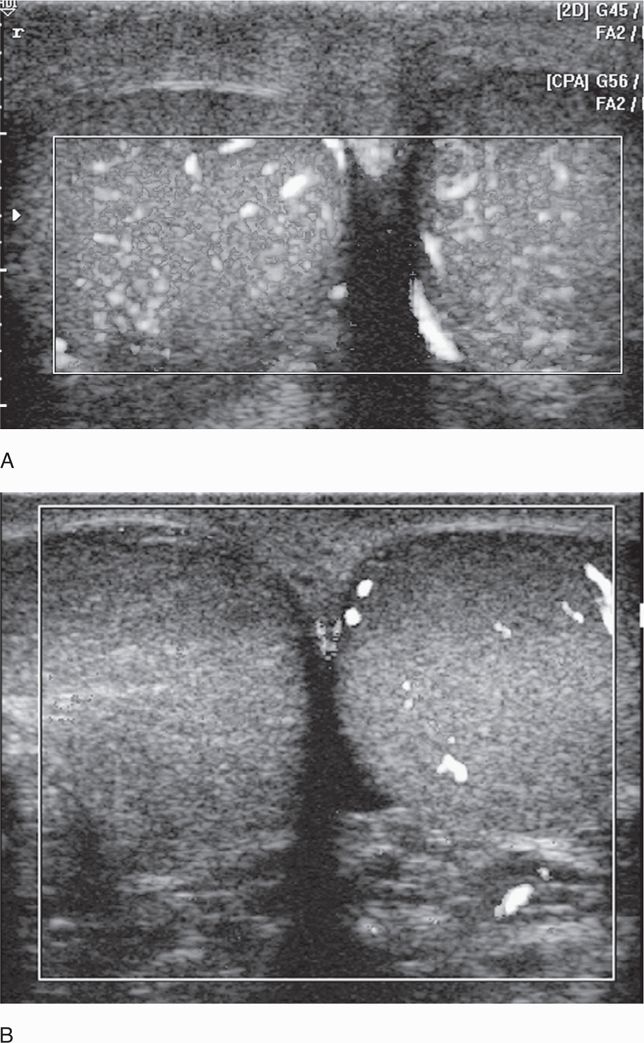
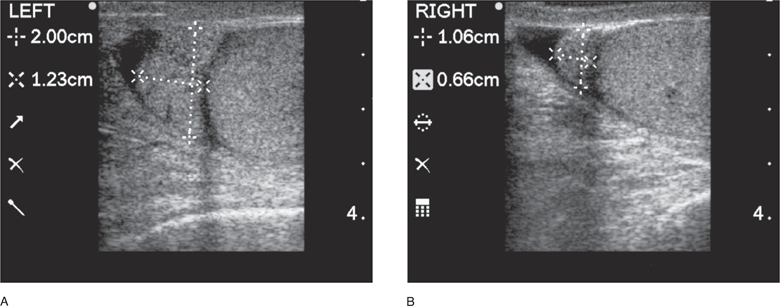
The epididymal head is the most commonly affected region, but the entire epididymis may be involved in acute epididymitis. Infection usually starts in the head or tail of the epididymis, and then spreads to the entire epididymis and the testis. Comparison with the contralateral testis is crucial to make an accurate diagnosis. Sonographically, the affected epididymis appears enlarged, which is usually confirmed by comparing both the affected and unaffected sides (Figure 13-18). It will often have decreased echogenicity on standard B-mode examination due to associated edema. Although the epididymis is diffusely involved, focal inflammation of a region of the epididymis is seen in one-third of the patients leading to an area of well-defined swelling or enlargement. This is usually either the head or the body of the epididymis. Scrotal wall thickening is frequently seen, which is an indirect sign of inflammation. Epididymitis can cause accumulation of a reactive hydrocele similar to testicular torsion or torsion of the appendix testis. Thus, the presence of a small or moderate amount of fluid in the hemiscrotum is not a reliable sign for differentiating between the disease processes. In cases with isolated epididymitis, the testis looks normal on ultrasound.26
Figure 13-18. Epididymitis. The left epididymal head is enlarged (A). Measurements for normal comparison on the right are included (B). (Courtesy of James Mateer, MD)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


 CLINICAL CONSIDERATIONS
CLINICAL CONSIDERATIONS CLINICAL INDICATIONS
CLINICAL INDICATIONS ANATOMICAL CONSIDERATIONS
ANATOMICAL CONSIDERATIONS GETTING STARTED
GETTING STARTED TECHNIQUE AND NORMAL ULTRASOUND FINDINGS
TECHNIQUE AND NORMAL ULTRASOUND FINDINGS COMMON AND EMERGENT ABNORMALITIES
COMMON AND EMERGENT ABNORMALITIES COMMON VARIANTS AND SELECTED ABNORMALITIES
COMMON VARIANTS AND SELECTED ABNORMALITIES PITFALLS
PITFALLS CASE STUDIES
CASE STUDIES