Algorithm 29.1
MR signal intensity of the vaginal wall layers
Blood supply to the vagina is supplied by branches of the internal iliac artery. The vaginal adventitia is surrounded by a rich venous plexus which drains into the internal iliac vein. Lymphatic drainage is also often divided into thirds. The upper one-third of the vagina drains into the internal and external iliac nodes, the middle third to the internal iliac nodes, and the lower third to the superficial inguinal nodes [3].
Estrogen fluctuations may affect the MR appearance of the vagina. Hormonal variations affect the size of the vagina, appearance of the inner mucosal layer, and the signal characteristics of the vaginal wall. An increase in the central mucosal layer has been noted during the mid-secretory phase of the menstrual cycle as well as with pregnancy [4].
Embryology
The upper two-thirds of the vagina, along with the uterus and cervix, are mesodermal in origin and are formed by midline fusion and canalization of the paramesonephric ducts. Alternatively, the lower one-third of the vagina is derived from endoderm through canalization of the sinovaginal node on the posterior aspect of the urogenital sinus. Eventually, the fused paramesonephric ducts and sinovaginal node combine to form a complete vaginal vestibule [5]. Failure of canalization or proper fusion of the paramesonephric ducts may result in distinct congenital anomalies of the uterus and vagina.
Current Techniques and Protocols
Computed Tomography (CT)
Although larger lesions may be identified, evaluation of vaginal anatomy is somewhat limited by CT due to close proximity of surrounding structures of similar soft tissue characteristics. The vagina may be differentiated from the cervix by inserting a tampon before imaging, which distends the vaginal canal with air density (Fig. 29.1).
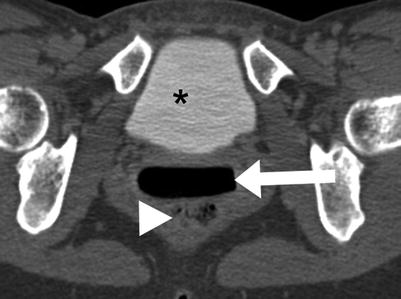

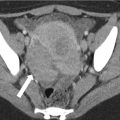
Fig. 29.1
Vaginal tampon. Axial oblique CT of the pelvis: Air density (white arrow) related to an intra-vaginal tampon expanding the vaginal canal, which is located between the urinary bladder (asterisk) and rectum (arrowhead). Image courtesy of Dr. Timothy McClure
The vaginal canal may also be expanded and differentiated from surrounding soft tissue structures by the insertion of other objects, such as a vaginal pessary (Fig. 29.2).
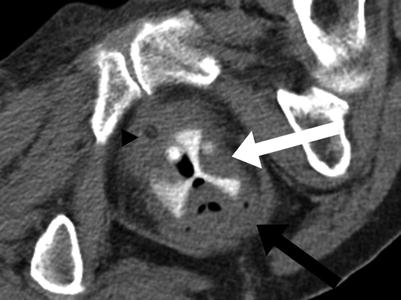

Fig. 29.2
Vaginal pessary. Axial CT through the vagina with a pessary (white arrow) expanding the vaginal canal, located between the urethra (black arrowhead) and rectum (black arrow). Image courtesy of Dr. Timothy McClure
Magnetic Resonance Imaging (MRI)
MRI is the ideal imaging modality for the vagina due to its superb soft tissue resolution. A standard pelvic MRI protocol, utilizing a pelvic or torso coil, includes axial T1-weighted (T1W), T2-weighted (T2W), and sagittal T2W images from the aortic bifurcation to the vulva (Fig. 29.3). Oblique high-resolution axial images may also be obtained to provide an axial view along the longitudinal axis of the vagina.
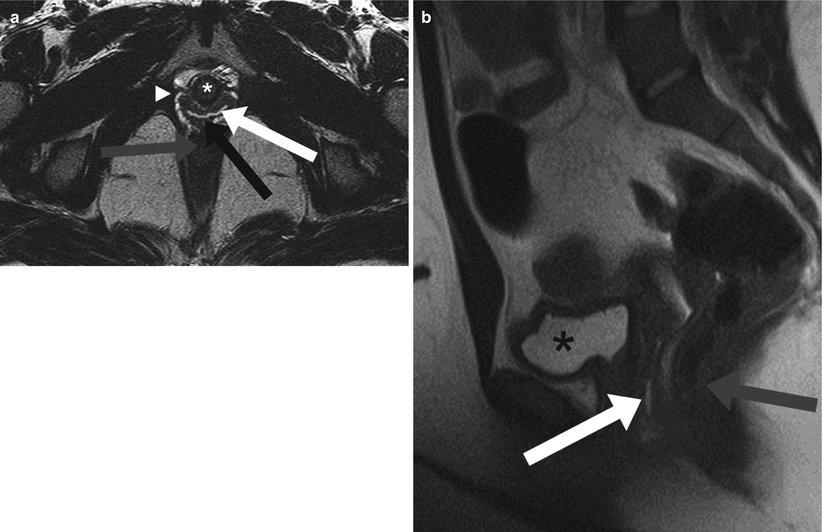

Fig. 29.3
Normal MRI of the vagina. Axial (a) and Sagittal (b) T2-weighted HASTE images. On axial images the vagina resides between the urethra (white asterisk) and rectum (gray arrow). This best demonstrates normal vaginal layers. The vaginal mucosa and luminal secretions located centrally appears T2 hyperintense (white arrow). The middle muscularis layer (black arrow) has low signal intensity while the adventitia (white arrowhead) has high signal intensity on T2-weighted images. On the sagittal image (b), the vagina is located between the urinary bladder (black asterisk) and rectum
Additional sequences and techniques may be used to evaluate suspected vaginal pathology (Table 29.1). For example, a large field of view coronal T2W sequence may be acquired when Mullerian and other congenital anomalies are suspected, to evaluate for associated renal pathology. The addition of axial T1W fat-suppressed images may aid detection of hemorrhagic or proteinaceous lesions, as well as in differentiating these lesions from fat.
Table 29.1
Evaluating suspected vaginal pathology
Axial T1W | Assess hemorrhage or mucin in vaginal mass |
Axial T2W | Detect mass and relationship to other pelvic structures |
Sagittal T2W | Relationship of vaginal lesions to other pelvic structures |
Assess involvement of cervix, rectum, or bladder | |
Coronal T2W | Assess associated genitourinary lesions |
Evaluate for hydronephrosis or lymphadenopathy | |
Axial FS postcontrast T1W | Evaluate vascularity of lesions |
Differentiate hemorrhagic or proteinaceous lesions from fat | |
Assist evaluation of vaginal fistulas |
Axial images provide the best information about the vaginal wall and its different layers. In the setting of vaginal malignancy, sagittal images will aid in the evaluation of urinary bladder or rectal involvement. Vaginal distention by intravaginal aqueous gel is thought to improve visualization of the vaginal wall (Fig. 29.4) [6].
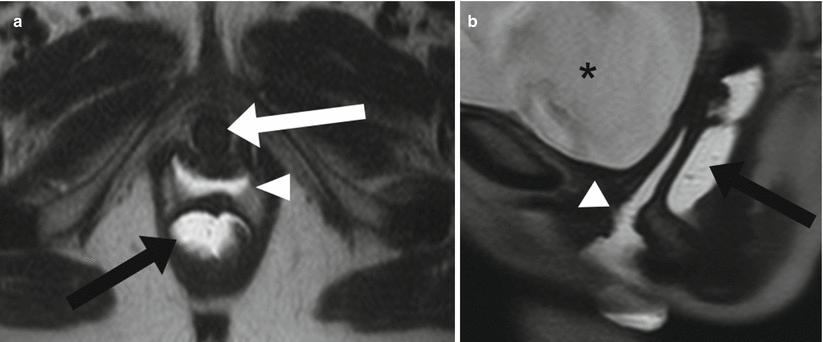

Fig. 29.4
Vaginal gel. Axial (a) and sagittal (b) T2-weighted HASTE images with aqueous gel inserted into the vagina (white arrowhead) and rectum (black arrow) to differentiate these structures from each other. The urethra (white arrow) and urinary bladder (black asterisk) are also well visualized
An endovaginal coil provides a small field of view (FOV) and higher resolution for improved evaluation of suspected vaginal pathology without altering most imaging parameters (Fig. 29.14). However, when using an endovaginal coil, larger pelvic masses may be partially excluded from the field of view and result in incomplete characterization. These large vaginal masses may even preclude the proper insertion of the endovaginal coil. Given the invasive nature of the coil, patient discomfort and embarrassment may also limit its use.
Preimaging intravenous or intramuscular glucagon administration is suggested to limit bowel peristalsis and their potential artifacts, which limit evaluation of the pelvis and other Mullerian structures [6].
Review of the Literature; Pattern Recognition Approach
Classification of Vaginal Pathologies
Vaginal pathologies may be classified based on their origin (Table 29.2). Congenital vaginal defects may be sporadic or a result of Mullerian anomalies. Changes in the radiologic appearance of the vagina may also be inflammatory in nature, such as with vaginitis or fistulous tracts. Mass-like benign and malignant vaginal neoplasms as well as postsurgical and postirradiation appearances of the vagina may be delineated on MR imaging.
Table 29.2
Pathological classification
Congenital | Inflammatory/infectious | Benign neoplasm | Malignant neoplasm |
|---|---|---|---|
Vaginal agenesis | Vulvovaginitis | Gartner’s duct cyst | Vaginal carcinoma |
Congenital vaginal outlet obstruction | Postsurgical/radiation changes | Bartholin gland cyst | Rhabdomyosarcoma |
Vaginal duplication | Fistula formation | Urethral diverticulum | Melanoma |
Complex Mullerian anomalies | Leiomyoma | Metastatic disease |
Congenital Anomalies
Vaginal Agenesis
Vaginal agenesis and hypoplasia are rare Mullerian anomalies resulting in dysgenesis of the uterus and upper two-thirds of the vagina [7]. Partial vaginal agenesis or vaginal hypoplasia is usually related to defects in vertical fusion of the urogenital sinus with the paramesonephric ducts. Vaginal hypoplasia therefore usually maintains a normal midline functioning uterus and cervix [5]. Complete vaginal agenesis is also known as Mayer-Rokitansky-Kuster-Hauser syndrome (MRKH) and is estimated to occur only once in every 5,000 women. Anomalies of cervical and uterine development are associated in 90 % of MRKH patients. Additionally, 10 % of these patients will have rudimentary Mullerian ducts with functioning endometrium [8].
Seeing that ovarian development and formation of the vaginal vault are preserved, development of secondary sexual characteristics and the external genitalia are normal [9]. Patients with vaginal agenesis present clinically around menarche with symptoms depending mainly on the presence or absence of rudimentary functioning endometrium. Lack of functioning endometrium most commonly presents with primary amenorrhea. The presence of functioning endometrium results in primary amenorrhea with cyclic pelvic pain and acute hematometra [8].
MR imaging aids in differentiating complete vaginal agenesis from hypoplasia as well as in identifying accompanying abnormalities such as rudimentary endometrial tissue, secondary endometriosis, or associated urologic anomalies. The sagittal plane is particularly useful in evaluating the extent and length of vaginal atresia (Fig. 29.5) [10].
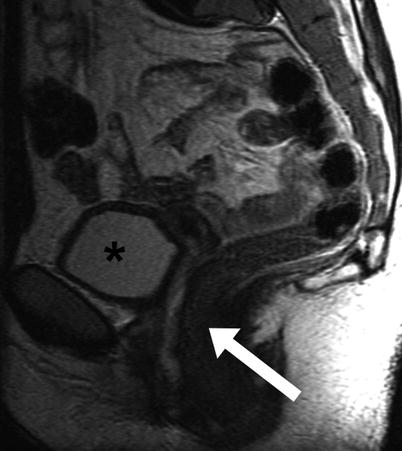

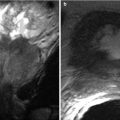
Fig. 29.5
Vaginal Agenesis. Sagittal T2-weighted FSE image of a 26 year old woman with known vaginal agenesis. The vagina is not visualized in its normal location between the urinary bladder (asterisk) and rectum (arrow)
Congenital Vaginal Outlet Obstruction
Anomalous obstruction of the vaginal canal may cause an accumulation of secretions in the vagina and uterus and result in hydrometrocolpos in embryos and neonates or hematometrocolpos at puberty (Fig. 29.6). Failure in embryologic vertical fusion of the Mullerian ducts results in variable anatomic anomalies which present as hydrometrocolpos/hematometrocolpos. These congenital anomalies include segmental vaginal atresia and transverse vaginal septum.
A transverse vaginal septum most commonly occurs in the proximal third of the vagina and results in the division of the vagina into two segments. Failure of the vaginal plate and Mullerian ducts to fuse may result in a partial or complete septum. The cervix and uterus are generally normal. More severe vertical fusion defects may result in partial vaginal atresia as already described above.
Hydrocolpos or hematocolpos may also occur with obstruction from an imperforate hymen. Unlike most congenital vaginal anomalies, this condition is unrelated to the Mullerian ducts and is a result of failed sporadic canalization of the hymen membrane. MR and CT imaging may demonstrate blood products filling the vagina and uterus with bulging at the introitus [8].
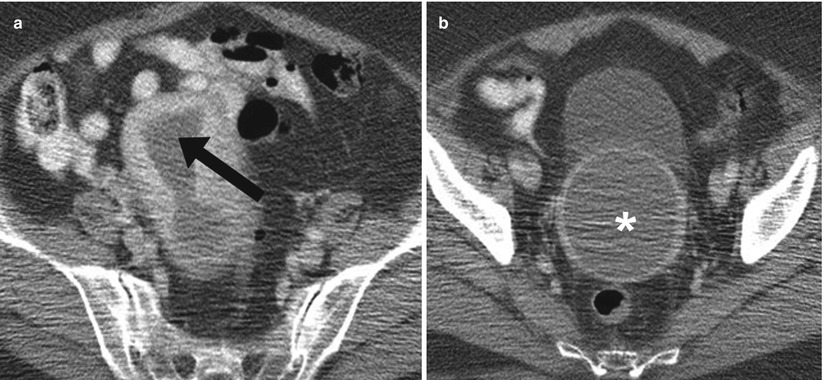

Fig. 29.6
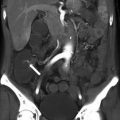
Hematometrocolpos. Axial contrast enhanced CT images of the pelvis at the level of the uterus (a) and lower uterine segment/cervix (b) demonstrates fluid density significantly distending the endometrial cavity (arrow) and vagina (asterisk)
Similar vaginal hypoplasia, patients with transverse vaginal septum or imperforate hymen may present during adolescence with primary amenorrhea and cyclical pelvic pain [8]. Evaluation with MR imaging assists in locating the region of pathology and thickness of the vaginal septum, as well as assisting in surgical planning. Given similar clinical presentations and imaging findings, differentiation between a low transverse vaginal septum and imperforate hymen may be difficult [11].
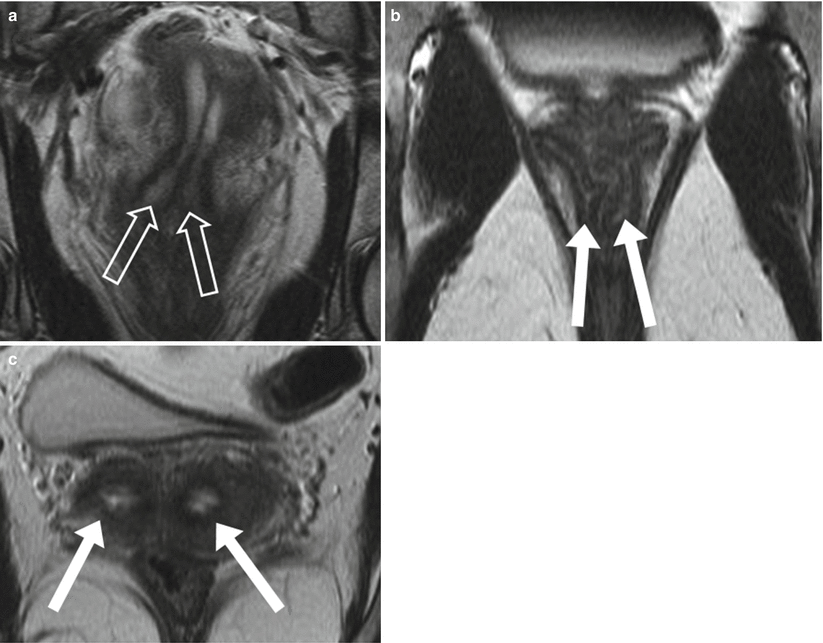
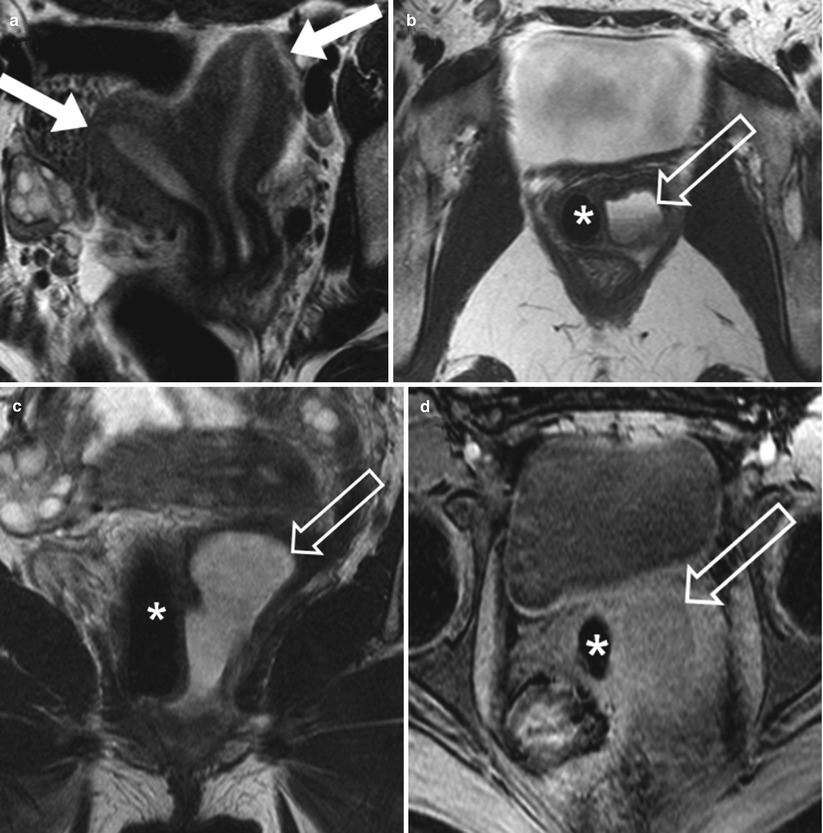
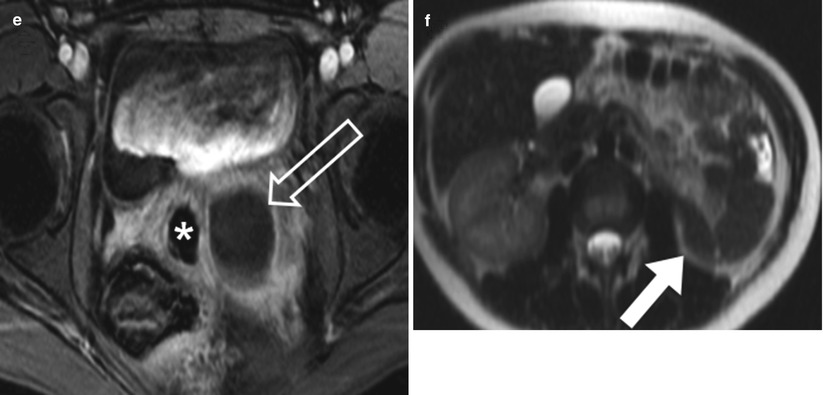

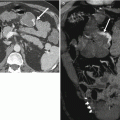
Fig. 29.7
Duplex cervix and vagina. Coronal (a & b) and axial (c) T2-weighted FSE images show the Mullerian anomalies of duplex cervix (transparent arrows) and vagina (white arrow) with T2 hyperintense mucosa and contained secretions


Fig. 29.8
Young woman with complex Mullerian duct fusion anomaly and pyocolpos. Axial (a & b) and coronal (c) T2W FSE images show widely splayed uterine horns (white arrows in a) as well as duplicated upper vaginas. Hypointense T2W signal without enhancement of the right vagina is due to an inserted tampon (white asterisk). Proteinaceous debris is noted in the left vagina (transparent arrow). Axial pre and postcontrast fat saturated T1-weighted images (d and e) demonstrate mucosal enhancement (transparent arrow), compatible with pyocolpos. Axial T2-weidgted-SSFSE image at the level of the kidneys (f) shows empty left renal fossa (white arrow) indicating renal agenesis
Vaginal Duplication
Mullerian duct anomalies may also lead to vaginal duplication. Failed midline fusion of the paramesonephric ducts or incomplete canalization of the vertical septum creates a longitudinal vaginal septum, dividing the length of the vagina in half. When due to anomalous Mullerian duct fusion, uterine pathology such as uterus didelphys and bicornuate or septate uterus may also be seen (Fig. 29.7). A focal transverse fusion anomaly or obstruction of one of the vaginal canals may result in ipsilateral hematometrocolpos (Fig. 29.8). A longitudinal vaginal septum is commonly asymptomatic. However, it may present with dyspareunia, particularly with sexual activity or child birth [8].
MR imaging is often helpful in identifying the vaginal septum, as well as accompanying uterine or renal pathology. The septum is best depicted on T2-weighted images as a hypointense thin longitudinal structure in the vaginal canal surrounded by hyperintense vaginal secretions and mucosa [12].
Diagnostic Approach of Vaginal Lesions
Vaginal lesions may initially be classified as either well or ill defined in appearance (Algorithm 29.2). Well-defined vaginal masses may be further categorized as cystic or solid lesions. Cystic masses are best described by their location, with urethral diverticulum, Gartner’s duct cyst, and Bartholin duct cyst originating from different characteristic sites.
Ill-defined soft tissue lesions may appear mass-like, as seen with malignant lesions of primary vaginal carcinoma or metastatic disease. Other infectious or inflammatory processes typically present with ill-defined non mass-like soft tissue involvement.
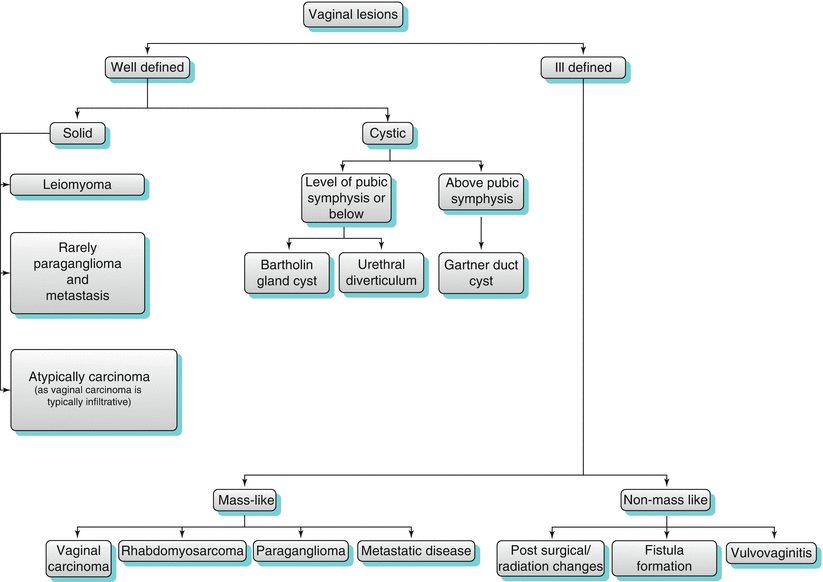

Algorithm 29.2
Vaginal lesion algorithm
Well Defined: Cystic Lesions
Urethral Diverticulum
Inflammation or traumatic injury to the periurethral glands and ducts may lead to fistulization to the urethra and create a urethral diverticulum. Visualization of a direct communication with the urethra is diagnostic; however, it is rarely visualized. Urethral diverticula are characteristically located along the posterolateral wall of the midurethra near the level of the pubic symphysis [13]. On MRI, these lesions may appear as a rounded cyst or demonstrate circumferential involvement around the urethra, creating the characteristic horseshoe appearance (Fig. 29.9). In patients, urethral diverticulum may be asymptomatic or result in pain, urinary incontinence, recurrent urinary tract infections, and dyspareunia [14].
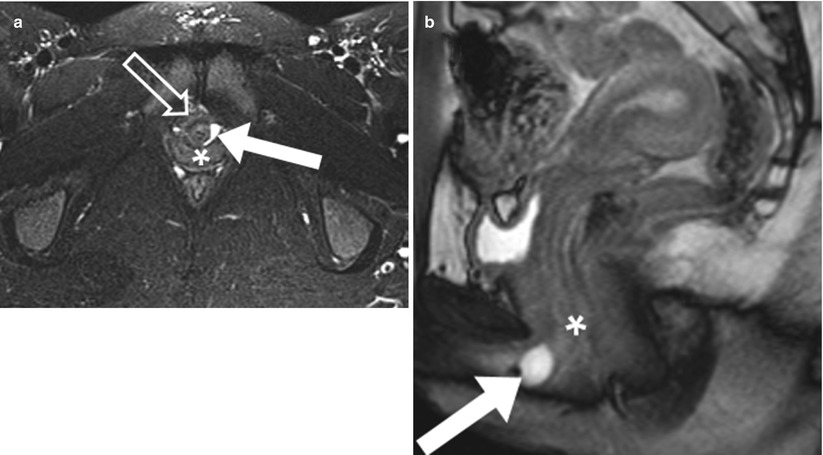

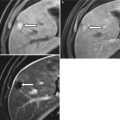
Fig. 29.9




Urethral diverticulum. Axial fat-suppressed T2-weighted (a) and sagittal T2-weighted (b) images of a urethral diverticulum. The axial T2W image demonstrates a curvilinear cystic diverticulum (white arrow) along the posterolateral urethral wall (transparent arrow) at the level of the pubic symphysis. The vagina (white asterisk) is noted posterior to the urethral diverticulum. The urethral diverticulum (white arrow) appears as a round cystic lesion on sagittal imaging
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree