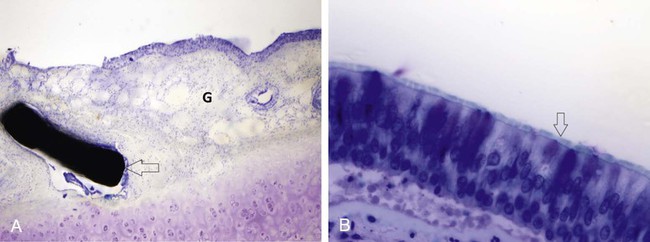
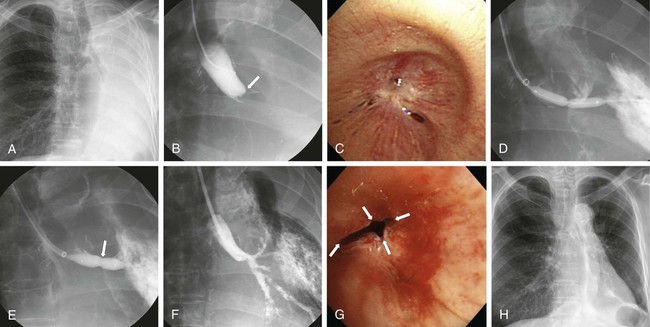
Ji Hoon Shin, Auh Whan Park, John F. Angle and Alan H. Matsumoto Central airway obstruction represents a great challenge to physicians from all subspecialties. Narrowing of the main tracheobronchial airways and proximal branches can cause significant and distressing symptoms for the patient and can be life threatening. Signs and symptoms of large-airway compromise include breathlessness, wheezing, stridor, and recurrent infections.1 Treatment for airway obstruction varies depending upon the length of the lesion, etiology of the obstruction, and age and overall prognosis for the patient. Malignancy is the most frequent cause of tracheobronchial obstruction in the adult population. Of the approximately 200,000 new cases of lung cancer diagnosed yearly in the United States, 20% to 30% will develop associated complications related to airway obstruction that can cause significant clinical problems such as resting and exertional dyspnea, atelectasis, postobstructive pneumonia, and hemoptysis.2–4 Resection of the pathology and reconstructive airway surgery provide the most definitive therapy, but many of these patients are not amenable to curative or corrective surgery. External beam irradiation and endobronchial brachytherapy, laser therapy, argon plasma coagulation, cryotherapy, photodynamic therapy, and electrocautery have been used in this setting. Most of these treatment options only provide temporary relief of symptoms because of the rapid regrowth of residual tumor.5–7 Surgical options, although rarely feasible, should always be explored first for both malignant and benign lesions, but a large number of patients with symptomatic and life-threatening airway pathology are not candidates for definitive surgical correction because of the extent of the disease or comorbidities. A multidisciplinary approach to obstructing malignancies, with bronchoscopic debulking or removal of the obstructing lesion, balloon dilation of the airway, stenting of the airway, or a combination of techniques, has proven to be highly effective in reestablishing functional airway patency and palliating patient symptoms.1 Benign lesions have been treated with a variety of endoscopic and fluoroscopic techniques. These therapeutic modalities usually provide immediate improvement and occasional long-term successes, but recurrences are the general rule.1,5,6 The etiology for the obstruction will determine the need for bronchoscopic tumor debulking, airway stenting (covered or uncovered), balloon dilation, or a combination of these techniques. Central airway obstruction can be divided into four main categories: intraluminal, extraluminal or extrinsic, dynamic (due to loss of airway integrity and dynamic collapse causing obstruction), or mixed. Although intraluminal obstruction typically requires tumor debulking prior to stent placement, pure extraluminal or extrinsic compression and dynamic airway collapse will mainly benefit from airway stenting, with or without prior bronchoscopic balloon dilation.1 On occasion, intraluminal benign scar tissue (e.g., lung transplant anastomosis or postinflammatory synechiae) can be treated with balloon dilation alone, but often stenting will be necessary.8–11 There are three major indications for airway stenting (Table 156-1). Two of these are for (1) enhancing airway patency by stenting an obstructed bronchus or trachea and creating a barrier for further tumor ingrowth (by using a covered stent) and (2) preventing extrinsic compression or dynamic obstruction by serving as a scaffold or supporting weakened airway walls (due to chondromalacia) and preventing airway collapse related to the static and dynamic behaviors of the stent.12,13 A third indication for using stents in the airway is for sealing airway dehiscences and fistulas. TABLE 156-1 Specific Indications for Tracheobronchial Stent Placement There have been no recent major advances in airway stent technology. The ideal stent should have limited migration, be easily removed if necessary, demonstrate long-term luminal patency without causing ischemia, be nonallergic, not erode into adjacent structures, induce minimal granulation tissue, be easy to accurately position, allow patency of bronchial branches, be durable, and allow for continued functioning of the mucociliary system. However, such an ideal stent is not yet available. A variety of stents are available from many different manufacturers (Table 156-2). Tracheobronchial stents can be divided into two main categories according to materials used in their construction: silicone and metallic-based stents. Hybrid stents result from the combination of these two major categories.13 TABLE 156-2 ePTFE, Expanded polytetrafluoroethylene; FDA, U.S. Food and Drug Administration. In 1965, Montgomery described the use of a T-shaped silicone tube designed to be used both as a tracheal stent and as a tracheostomy tube. The tube is positioned in the trachea, with the sidearm of the T projecting through the tracheostomy. The sidearm prevents tube migration and provides access for clearance of secretions.14 In 1990, Dumon developed the first silicone stent that could be inserted with a bronchoscope and did not require a tracheostomy. It was designed with no external components and could be inserted using a rigid bronchoscope. The outside surface of this stent had rounded studs protruding from its surface. The studs are designed to prevent the stent from sliding or turning. The Dumon stent is available in several sizes for both tracheal and bronchial applications. Different types or designs of silicone stents are also available.15 Silicone stents are fairly well tolerated and have been shown to be effective in relieving respiratory symptoms; they have the advantage of being easily removed and exchanged when necessary. The most frequent problems associated with silicone stents are migration and mucous plugging of the stents.15 Migration is most likely to occur in short conical stenoses with intact smooth mucosa, or in the presence of tracheobronchomalacia. In both situations, the underlying anatomy does not permit firm anchorage of the stent. The mucosa under most silicone stents undergoes a metaplastic alteration, reducing the effectiveness of the mucociliary clearance mechanism, leading to recurrent stent plugging. In most cases, migration or mucous plugging of the stent can be managed by frequent endoscopic repositioning or replacement of the silicone stent.16 Metallic tracheobronchial devices have been in use since the early 1950s, but were refined when metallic stents were developed for use in the vascular system. Metallic stents offer several potential advantages over silicone stents for treatment of complex airway obstruction. Metallic stents have a small profile and are fairly easy to insert; the open lattice design allows treatment of more peripheral bronchi with less fear of causing obstructive pneumonia and/or atelectasis. Following placement of a metallic stent, the normal respiratory epithelium can protrude through the open lattice, and metaplastic squamous epithelium overgrows and incorporates the stent into the wall of the airway. The neoepithelium overlying the stent also appears to maintain some rudimentary ciliary function (Fig. 156-1).16,17 However, granulation tissue and tumor can grow through the open lattice of these stents and lead to recurrence of airway obstruction. In addition, once metallic stents become incorporated into the wall of the airway, they are very difficult to remove without surgery. Covered stents are used to prevent intraluminal tumor ingrowth or when a fistula has to be excluded or sealed. Covered stents also have the advantage of relatively easy removal or exchange compared with uncovered stents (Table 156-3). However, covered stents may prevent neoepithelialization and compromise the mucociliary clearance mechanism of the airway. TABLE 156-3 Comparison Between Silicone and Metallic-Based Stents Balloon dilation has been proposed for management of benign stenoses of the airways. The noncompliant balloon dilates the stenotic trachea or bronchus by stretching, tearing, and expanding scar tissue and the airway wall. Balloon dilation is associated with little morbidity and mortality,18 but this technique is of little value in the treatment of malacic segments or airway narrowing secondary to tumor ingrowth or extrinsic compression. In addition, balloon dilation has failed to be of durable benefit for lung-transplant anastomotic strictures.11 Increased availability and ease of delivery of metallic balloon-expandable or self-expanding stents has resulted in a lower threshold for considering placement of airway stents. However, other therapeutic modalities and possible long-term complications inherent with the use of a metallic stent should be considered, because these patients will likely require lifelong management and revision. Indeed, an increasing number of adverse events reported in association with use of metallic stents for treatment of patients with benign airway disease led the U.S. Food and Drug Administration (FDA) to publish an advisory in 2005 on their use.19 These were the recommendations: • Appropriate patient selection is crucial. • Use metallic tracheal stents in patients with benign airway disorders only after thoroughly exploring all other treatment options (e.g., tracheal surgical procedures or placement of silicone stents). • Using metallic tracheal stents as a bridging therapy is not recommended, because removal of the metallic stent can result in serious complications. • If a metallic tracheal stent is the only option for a patient, insertion should be done by a physician trained or experienced in metallic tracheal stent procedures. • Should removal be necessary, the procedure should be performed by a physician trained or experienced in removing metallic tracheal stents. • Always review the indications for use, warnings, and precautions. • Be aware of the guidelines from professional organizations regarding recommended provider skills and competency for these procedures (i.e., training requirements and clinical experience). The primary indication for using balloon dilation is for treatment of benign tracheobronchial strictures. Postintubation tracheal stenosis, postoperative anastomotic stenosis, chemical aspiration–induced scarring, granulation tissue due to granulomatous disease (tuberculosis, sarcoidosis, histoplasmosis), postinflammatory stricture, and stenosis induced by radiation therapy are the principal etiologies for benign airway stenosis.20–24 Balloon dilation can also be used either before or after stent placement to optimize airway patency.20,21 Presence of an active inflammatory process or infection of the tracheobronchial tree is a relative contraindication to balloon dilation, owing to the potential for aggravating the underlying process. Small case series and limited reports in which balloon dilation was the only treatment used for benign tracheobronchial stenoses have shown a high initial technical success rate, but recurrence of symptoms necessitated further treatment with repeat dilation, stenting, or laser therapy in 71% to 80% of patients.25,26 Ipsilateral bronchial stenosis is found in 7% to 15% of lung transplantation recipients,27 and ischemic damage (which often affects the bronchial anastomosis), rejection, and infection have been considered as individual and/or concomitant predisposing causes.28 In one retrospective study of lung-transplant bronchial stenoses, bronchoscopic balloon dilation showed effective results in only 50% (5 of 10 bronchial stenosis) after an average of four balloon dilation procedures, suggesting that single or multiple sessions of balloon dilation could be used as a possible approach to management of bronchial stenoses after lung transplantation.22,27 In cases of bronchial stenosis refractory to repeated sessions of balloon dilation, stent placement was often employed.22,29 For treatment of benign bronchial strictures resistant to conventional balloon dilation, use of a cutting balloon has shown some preliminary promise.30 Kim et al. reported successful use of the cutting balloon, no major complications, and a demonstrated clinical benefit of approximately 60% (11 patients) at 2 years (Fig. 156-2).
Tracheobronchial Interventions
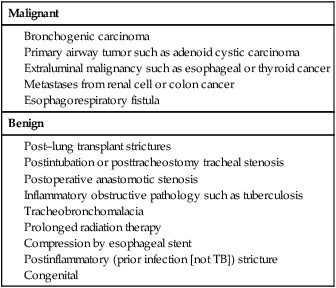
Malignant
Benign
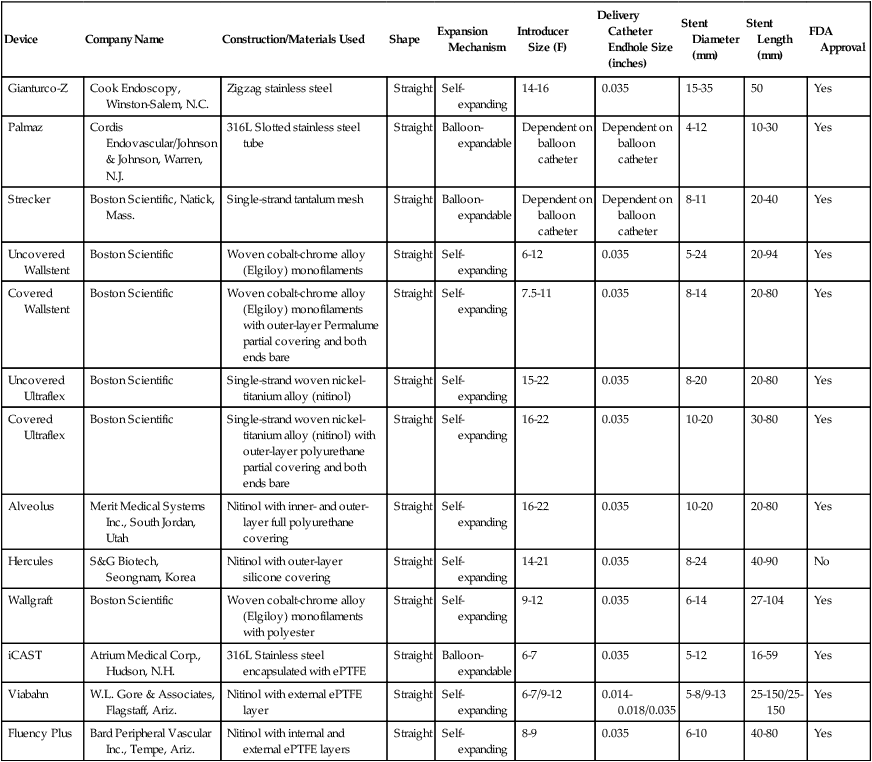
Device
Company Name
Construction/Materials Used
Shape
Expansion Mechanism
Introducer Size (F)
Delivery Catheter Endhole Size (inches)
Stent Diameter (mm)
Stent Length (mm)
FDA Approval
Gianturco-Z
Cook Endoscopy, Winston-Salem, N.C.
Zigzag stainless steel
Straight
Self-expanding
14-16
0.035
15-35
50
Yes
Palmaz
Cordis Endovascular/Johnson & Johnson, Warren, N.J.
316L Slotted stainless steel tube
Straight
Balloon-expandable
Dependent on balloon catheter
Dependent on balloon catheter
4-12
10-30
Yes
Strecker
Boston Scientific, Natick, Mass.
Single-strand tantalum mesh
Straight
Balloon-expandable
Dependent on balloon catheter
Dependent on balloon catheter
8-11
20-40
Yes
Uncovered Wallstent
Boston Scientific
Woven cobalt-chrome alloy (Elgiloy) monofilaments
Straight
Self-expanding
6-12
0.035
5-24
20-94
Yes
Covered Wallstent
Boston Scientific
Woven cobalt-chrome alloy (Elgiloy) monofilaments with outer-layer Permalume partial covering and both ends bare
Straight
Self-expanding
7.5-11
0.035
8-14
20-80
Yes
Uncovered Ultraflex
Boston Scientific
Single-strand woven nickel-titanium alloy (nitinol)
Straight
Self-expanding
15-22
0.035
8-20
20-80
Yes
Covered Ultraflex
Boston Scientific
Single-strand woven nickel-titanium alloy (nitinol) with outer-layer polyurethane partial covering and both ends bare
Straight
Self-expanding
16-22
0.035
10-20
30-80
Yes
Alveolus
Merit Medical Systems Inc., South Jordan, Utah
Nitinol with inner- and outer-layer full polyurethane covering
Straight
Self-expanding
16-22
0.035
10-20
20-80
Yes
Hercules
S&G Biotech, Seongnam, Korea
Nitinol with outer-layer silicone covering
Straight
Self-expanding
14-21
0.035
8-24
40-90
No
Wallgraft
Boston Scientific
Woven cobalt-chrome alloy (Elgiloy) monofilaments with polyester
Straight
Self-expanding
9-12
0.035
6-14
27-104
Yes
iCAST
Atrium Medical Corp., Hudson, N.H.
316L Stainless steel encapsulated with ePTFE
Straight
Balloon-expandable
6-7
0.035
5-12
16-59
Yes
Viabahn
W.L. Gore & Associates, Flagstaff, Ariz.
Nitinol with external ePTFE layer
Straight
Self-expanding
6-7/9-12
0.014-0.018/0.035
5-8/9-13
25-150/25-150
Yes
Fluency Plus
Bard Peripheral Vascular Inc., Tempe, Ariz.
Nitinol with internal and external ePTFE layers
Straight
Self-expanding
8-9
0.035
6-10
40-80
Yes
Silicone
Metallic
Bronchoscopy
Rigid
Rigid or flexible
Flexibility
No
Easily conform to the anatomy
Secretions
Mucous plugging
Less epithelial/cilia ingrowth
Migration
More frequent
Less frequent
Tumor ingrowth
No
Possible
Granulation tissue formation
Little
Profound
Mucociliary function
Impaired
Preserved
Removal/adjustment
Easy
Difficult
Tracheobronchial Balloon Dilation
Indications and Contraindications
Outcomes of Balloon Dilation
Tracheobronchial Interventions