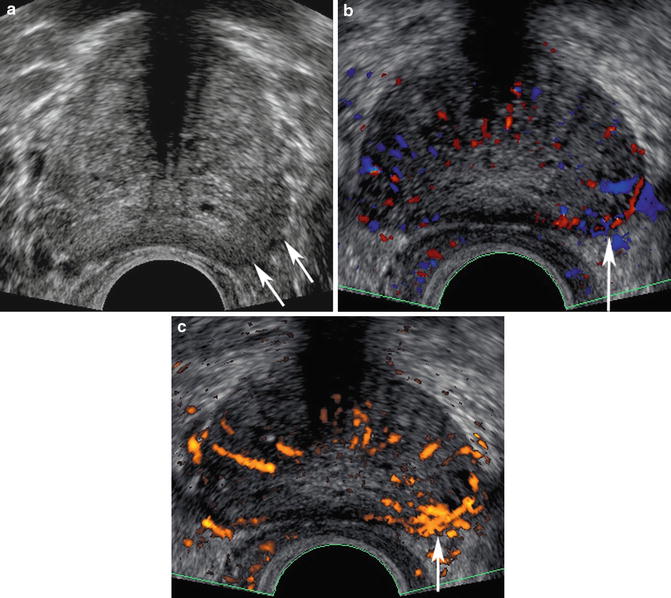
Fig. 10.1
TRUS findings suspicious for prostate cancer. (a) Lobar asymmetry, capsular bulging, increased blood flow, and deflection of the junction between the TZ and PZ can all be indicators for prostate cancer on TRUS. (b) Transverse image of the prostate demonstrating upward deflection of the junction between the TZ and PZ. The corpora amylacea (arrows) indicate this junction and also demarcate the border between the inner and outer gland
TRUS is used to define the anatomy and calculate the volume of the prostate in symptomatic BPH prior to surgical therapy or minimally invasive therapy (MIT) for BPH [18, 19]. The size or anatomy of the prostate can recommend or exclude certain MIT procedures for patients. Specific volume limits are used to stratify patients for transurethral resection of the prostate (TURP), MITs such as transurethral microwave therapy (TUMT) or transurethral radiofrequency needle ablation (TUNA) or open simple prostatectomy. In addition to prostate volume measurements, the presence or absence of a large intravesical median lobe of the prostate visualized on TRUS may also help guide the optimal technique for surgical treatment of BPH.
Minimally invasive prostate cancer treatment options are dependent on TRUS technology to accurately monitor and guide treatment planning and delivery [20]. TRUS provides the clinician the anatomy and specifically an accurate volume assessment of the prostate prior to any procedure. Certain patients may be excluded from some treatment options for prostate cancer or may require hormonal downsizing prior to treatment based on TRUS findings. For cryotherapy, real-time TRUS is vital to accurately place probes and monitor the freezing area during the procedure [21]. Similarly, in brachytherapy, real-time TRUS is used to volume the prostate and construct the plan for seed implantation. Continuous real-time TRUS is also used during high intensity focused ultrasound (HIFU ) treatment of the prostate [22, 23]. As part of image-guided radiotherapy for prostate cancer, TRUS is used to accurately place prostate markers such as fiducial gold seeds within the prostate for daily monitoring of prostate position and accurate imaged-guided treatment planning, as shown in Fig. 10.2 [24, 25].
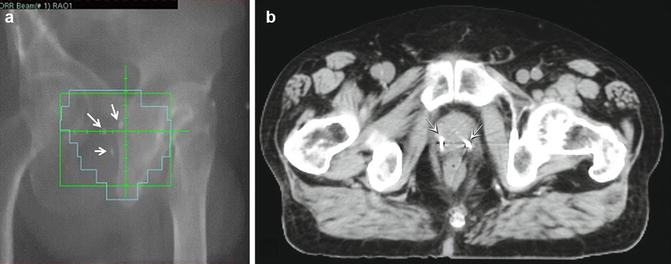

Fig. 10.2
Port film after fiducial marker placement for prostate cancer. Three markers are visible on plain film within the prostate and are used for daily position adjustments during radiotherapy treatment
In patients with azoospermia or ejaculatory dysfunction , TRUS may also help diagnose cysts of the SV or ejaculatory ducts as well as obstruction of the vas deferentia. Accurate diagnosis of these rare conditions can change the management strategy of infertility patients [6–8]. Another rare but useful application of TRUS is for the treatment of prostatic abscesses which can be associated with prostatitis or epididymitis [26, 27]. These abscesses can then be surgically unroofed during transurethral or transrectal procedures.
Techniques
A complete TRUS evaluation of the prostate includes scanning in both the sagittal and transverse planes to obtain a volume calculation. The CZ and the PZs are evaluated for hypoechoic lesions and contour abnormalities. The SVs and the vas deferentia are also visualized during the ultrasound procedure. Any abnormalities or ectasia are measured and documented. Additionally, the rectal wall is examined for abnormal thickening or focal lesions as occasional rectal tumors can be encountered on TRUS.
Prior to the transrectal ultrasound procedure, patients are typically asked to do a cleansing enema at home before the procedure. The enema helps decrease the amount of stool in the rectum, thereby producing a superior acoustic window for prostate imaging. Although an enema is not absolutely mandatory, it is helpful in obtaining optimal images during TRUS. In patients undergoing real-time imaging for cryotherapy, brachytherapy or HIFU , preoperative or intra-operative enema is integral for adequate real-time imaging and image guidance during the procedure. If any invasive procedure is planned, antiplatelet and anti-coagulation medications such as ASA, NSAIDs, clopidogrel, warfarin, vitamin E, and fish oil should be stopped at least 1 week prior to the procedure reducing bleeding risk associated with the procedure. Oral antibiotics should also be given to reduce the risk of bacterial septicemia. This should be given prior to the procedure as well as continued post-procedurally.
Patients are positioned either in the left lateral decubitus or the lithotomy position. Most clinicians would prefer the left lateral decubitus position, especially for a straightforward TRUS. In this position, an arm board is usually placed parallel to the table and a pillow is placed between the knees to help maintain the position. The buttocks should be flushed with the end of the table to allow manipulation of the probe. Next, a digital rectal exam should be performed prior to insertion of the TRUS probe. Any palpable contour abnormalities should be documented including descriptive identifiers and their location on the gland. The lithotomy position is used with patients who are undergoing brachytherapy, cryotherapy for external beam radiation. In addition, the lithotomy position is used for patients in whom color flow and power Doppler imaging of the prostate is planned [28]. Because the vascularity pattern of the prostate is dependent on patient positioning and gravity, the lithotomy position is preferred to help accurately identify areas of hyperemia for targeted biopsies of the prostate.
Gray-scale TRUS has been the most common imaging modality of the prostate. Although Doppler imaging with color flow, power Doppler, and harmonic imaging have been advocated in the diagnosis of prostate cancer, their exact role for other treatment modalities such as cryotherapy or brachytherapy is still under investigation. These techniques will be further discussed in a separate section of this chapter.
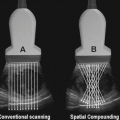
There are two different types of probes available for TRUS, with either side or end firing models, based on surgeon preference and experience. Either type of probe transmits mid-level frequencies between 6 and 10 megahertz (MHz). Models with new biplane probe technology provide a simultaneous image of both sagittal and transverse images during the transmission period. By increasing the frequency used, the spatial resolution is improved. As the frequency of the probe is increased, however, the depth of tissue penetration declines, and the portion of the image that has adequate returning echo amplitude is closer to the transducer [29]. Increased resolution may be helpful in patients who have peripheral zone cancers which may be identified as a hypoechoi c nodule with TRUS, but the anterior portions of the prostate furthest from the probe may suffer from poorer resolution, especially for marked BPH . Lower frequency transducers, such as the 4 MHz transducers, have a greater depth of penetration (between 2 and 8 cm) but with much lower resolution. Lower frequency transducers improve the anterior delineation of larger glands with deeper tissue penetration, thus increasing the accuracy of volume measurements but provide poorer resolution.
A coupling medium, which is usually sonographic jelly or a lubricant, is usually placed between the probe and the rectal surface. This is essential since ultrasound waves propagate inefficiently through air and are completely reflected when they strike tissues of a significant impedance difference. Most probes are also covered with a protective condom. A coupling medium is placed between the probe and the condom as well as the condom and rectal mucosa.
Any TRUS scanning protocol must involve the prostate and should be performed in both the transverse and the sagittal planes. By advancing the probe in a cephalad direction in the rectum, images of the prostate base, seminal vesicles and the bladder neck are obtained. By pulling the probe caudally towards the anal sphincter, the prostatic apex and proximal urethra are visualized. Transverse imaging with end fire, side fire, and some biplane probes is accomplished by angling the handle of the probe, right or left using the anal sphincter as a fulcrum. Angling the probe towards the scrotum produces more cephalad images while angling the probe towards the sacrum produces more caudal images. There are also two approaches to probe manipulation for sagittal imaging. One method is rotation of the probe either clockwise or counterclockwise. Clockwise rotation visualizes the left side of the prostate and counterclockwise rotation yields images of the right side. Alternatively, sagittal images can also be viewed by angling the probe up or down the anal sphincter which functions as a fulcrum. If the patient is placed in the left lateral decubitus position, angling the probe up or towards the ceiling images the left side of the prostate while angling the handle of the probe down or towards the floor images the right side. Prostate volume is usually calculated after complete scanning of the prostate is accomplished. Volume calculation requires measurement in three dimensions. The transverse and anterior posterior (A-P) dimensions are measured at the point of the widest diameter in the axial plane. The longitudinal dimension is measured in the sagittal plane just off the midline as sometimes the bladder neck may obscure the cephalad extent of the gland. Most formulas to assess prostate volumes assume an ideal geometric shape of the gland. These shapes can include an ellipse (π/6 × transverse diameter × A-P diameter × longitudinal diameter), a sphere (π/6 × the transverse diameter3), or an egg-shaped spheroid (π/6 × transverse diameter2 × the A-P diameter). Despite the inherent inaccuracies that arise from these geometric hypotheses, these formulas reliably estimate the gland volume and weight with correlation coefficients greater than 0.90 when compared to prostatectomy specimen weights (based on the assumption that 1 cm3 equals 1 g of prostate tissue) [30]. By calculating the prostate volume, one can then derive the PSA density (PSAD) which is the ratio of serum PSA to prostate volume. The PSAD can help improve prostate cancer detection specificity and has been shown to be higher in patients diagnosed with cancer on initial and repeat prostate biopsies [31].
In patients undergoing brachytherapy, more accurate gland volume may be required. This is accomplished by a technique called planimetry . With the patient in the lithotomy position, the probe is mounted to a stepping device and transverse images are obtained at set intervals. This is done throughout the length of the gland. The surface area of each serial image is determined and the sum of these measurements is multiplied by the total gland length to obtain the prostate volume.
TRUS imaging of the prostate alone is a relatively benign procedure. Other than patient discomfort due to the physical presence of an ultrasound probe in the rectum, there is little patient risk in receiving a stand-alone TRUS. However, complications can result after TRUS coupled with penetration of the rectal wall (e.g., fiducial marker placement). These include hematuria, hematospermia, rectal bleeding, and urinary retention. Hematuria after needle biopsy occurs in roughly 47 % of patients and typically resolves after 5–7 days [32]. Hematospermia can be observed in up to 36 % of biopsies and small traces of blood may even persist up to several months [33]. Rectal bleeding is usually mild and controllable with digital or TRUS probe pressure. Bleeding requiring transfusion, bacteremia requiring IV antibiotics, and other more major complications are uncommon, but antibiotic resistant organisms are becoming more common and troublesome, raising questions about the appropriate antibiotic prophylactic coverage for these procedures [34]. Fortunately, most complications are generally minor and usually self-limiting.
Documentation
A standard protocol or a standard template should be used in documenting a transrectal ultrasound procedure. The template should state the indication, PSA, if appropriate, and the findings on digital rectal exam. The technique should be described and the position of the patient should be documented. The machine and the probes used in the procedure should be described. The anatomy and the calculated volume of the prostate should be annotated. Any abnormal findings should be noted. A sample TRUS documentation sheet is shown in Fig. 10.3a, a sample prostate biopsy documentation template is shown in Fig. 10.3b.
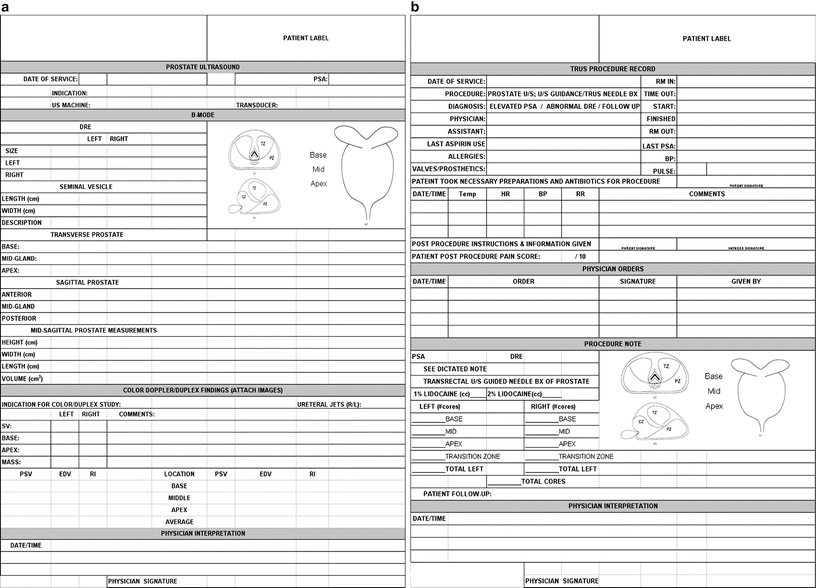

Fig. 10.3
Sample TRUS documentation sheets are shown. (a) Patient unique identifiers need to be appropriately labeled. Date of exam, indication for procedure, pre-evaluation PSA, and specific TRUS machine/probe models should also be documented. A DRE should be performed and any findings acknowledged. All features of the TRUS findings should be completely documented. These include (1) all lengths necessary for volume measurements, (2) the volume of the gland, (3) any areas suspicious for neoplasm, and (4) any other interesting or concerning findings. The presence or absence of urethral jets can also be commented on if visualization of the bladder is desired. Color or power Doppler findings can also be noted. PZ peripheral zone , CZ central zone , TZ transition zone, SV seminal vesicle, PSV peak systolic velocity, EDV end diastolic velocity, RI resistive index. (b) Documentation for prostate biopsy with TRUS. Akin to standard operative documents, all necessary components such as physician/assistant names, time in/out, allergies, and pre-procedural antibiotics should be written. The number of biopsies at each prostate site should be documented and correlated to eventual pathology reports
Multiparametric Ultrasound and Emerging Technologies
Despite the most meticulous and systematic approach to TRUS, the sensitivity of prostate biopsy remains disappointingly low, with false negative rates of 25 % or higher in multiple studies [35, 36]. In an attempt to make the biopsy procedure less random and more targeted, enhanced ultrasonographic techniques have been utilized to better identify areas of the prostate more suspicious for cancer to allow targeted biopsies. Similar to multiparametric MRI, multiparametric ultrasound has emerged. Various ultrasound modalities include: color Doppler ultrasound , dynamic contrast-enhanced ultrasound , power Doppler ultrasound , computerized transrectal ultrasound , and elastography [37]. Neoplastic prostate tissue is histologically distinct from normal prostate tissue, differentiated by a loss of glandular architecture, increased cellular density and increased microvascularity [38]. The loss of glandular architecture reduces the content of reflective interfaces causing a hypoechoic appearance on TRUS [38]. Increased cellular density may change the normal elasticity of prostate tissue which may be measurable by real-time elastography [39]. These characteristics can be used as potential targets in advanced ultrasound imaging.
Color and power Doppler have both been used to enhance TRUS biopsy detection of prostate cancer [40, 41]. Color Doppler measures the frequency shift in ultrasound waves as a function of the velocity and direction of vascular flow. Increased blood flow as a result of increased microvascularity to cancerous tissue forms the theory behind use of color and power Doppler sonography [38]. Amplified flow patterns to areas of tumor, which harbor increased number and size of blood vessels, is the hallmark of prostate cancer detection using color Doppler imaging (Fig. 10.4a–c). Targeted biopsies of the prostate can then be performed where there is enhanced flow. Power Doppler reflects the integrated amplitude or power of the Doppler signal. This technique is more sensitive to low velocity flow than color Doppler (Fig. 10.4c) can detect vessels as small as 1 mm allowing for visualization of prostate tumor feeding vessels. Several studies have shown an increased sensitivity of power Doppler to detect cancer [42, 43]. Unfortunately, when looking at the overall picture, the use of color and power Doppler has yielded mixed results. Sensitivity for tumor detection lies anywhere from 13 to 49 % in recent studies and ultrasound abnormalities has been shown in many patients with subsequently demonstrated benign pathology [40, 41, 44, 45]. These findings may be due to the presence of benign conditions, such as prostatitis, that result in increased blood flow to the prostate thereby clouding the picture for targeted cancer biopsies using this technique. In a study by Eisenberg et al., pathology from radical prostatectomy specimens from men between 2002 and 2007 were compared to preoperative TRUS and power Doppler ultrasound findings. When a hypoechoic lesion visualized on TRUS was combined with the finding of a hypervascular lesion on Power Doppler imaging, the specificity improved from 47 to 74 % [46]. However, the sensitivity decreased from 59 to 47 % [46]. Several studies have attempted to use power Doppler ultrasound in predicting tumor stage and grading, but, so far, results have been inconsistent [46].