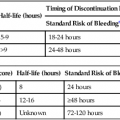
Renal histopathologic examination remains the diagnostic gold standard for most renal parenchymal diseases1,2 and is therefore essential for clinical management of renal diseases such as proteinuria, hematuria, and renal failure.3,4 The conventional technique for obtaining renal tissue involves percutaneous puncture of the kidney. This is a safe technique when performed in low-risk patients by experienced operators.5 The first description of renal tissue sampling by percutaneous needle biopsy was published in 1951.6 Improved technology and ultrasound guidance have considerably reduced the risk of complications with renal biopsy techniques and improved safety and efficacy. Nevertheless, significant risk still attends renal biopsy, and serious complications have been reported. Even under ideal circumstances, overt complications occur in up to 3.5% of cases,7 and the incidence of perirenal hematoma has been reported to be 57% to 85%.8,9 Percutaneous renal biopsy is therefore considered high risk in patients with abnormal clotting or low platelets. Furthermore, a number of other clinical conditions such as solitary, small, or obstructed kidneys, uncontrolled hypertension, horseshoe kidney, mechanical ventilation, and uncooperative patients present relative contraindications to percutaneous approach. An aging population, diabetes, and more widespread use of anticoagulant and antiplatelet drugs have led to more frequent clinical encounters with high-risk biopsy candidates. Some novel techniques have been developed in the last decade as alternatives to percutaneous biopsy in patients with contraindications.10–12 They include performing renal biopsies through an open surgical approach, transvenous approaches using transjugular and transfemoral access, and laparoscopic techniques. Development of a cutting core biopsy needle has made transjugular renal biopsy the most important of these. Open biopsy has the added risk of general anesthesia and its associated morbidity and mortality. Transjugular biopsy is theoretically safer because the needle is advanced as distally as possible into the medullary interlobar veins; the needle then passes through the vein wall into the surrounding parenchyma, directed away from the larger blood vessels. When bleeding does occur, it will do so back into the venous system, limiting extravascular blood loss.13 Another theoretical advantage of the transjugular approach is a lower likelihood of capsular perforation with the inside-out approach, in comparison to the 100% capsular perforation rate with percutaneous biopsy. Furthermore, if capsular perforation occurs and there is significant extravasation, elective coil embolization of the biopsy track can be performed during the same procedure. Transjugular renal biopsy technique was developed as a modification of the classical transjugular liver biopsy14 and described in 1990 by Mal et al.15 Authors first reported using a modified 9F liver core biopsy needle in 50 patients for transjugular renal cortical biopsies,15 and then in 200 patients with contraindications to percutaneous biopsy.10 In a recent comparison of 400 transjugular transvenous renal biopsies to an equal number of percutaneous biopsies, similar results were reported for both.7 This is a significant finding because 75.8% of patients in the transjugular renal group had bleeding disorders. In clinical practice, the proportion of patients with contraindications to percutaneous biopsy is small (≈7%).16 In high-risk patients, transjugular renal biopsy provides clinicians with expanding opportunities for obtaining renal histologic samples. Clinical utility of this procedure is also emphasized by the fact that diabetic patients undergoing renal biopsy frequently have nondiabetic renal disease.17,18 It has been established that transjugular renal biopsies, especially in patients with acute renal failure, affects patient management.19 Initial studies used a modified Colapinto aspiration needle for the biopsy. More recently, transjugular 19G Quick-Core side-cut biopsy needle systems (Cook Medical, Bloomington, Ind.) have been popular, with good cortical sampling.7,20 However, there is a higher incidence of capsular perforation.20 The blunt-tipped Quick-Core biopsy needle is a modification of the device that has been shown to not only provide sufficient cortical tissue for histopathologic diagnosis but also possibly reduce capsular penetration and hence significant bleeding in an animal study21 and a subsequent study of seven patients.22
Transvenous Renal Biopsy
Clinical Relevance
Equipment
Radiology Key
Fastest Radiology Insight Engine