12 Ultrasound Assessment of the Intracranial Arteries
In 1965, Miyazaki and Kato1 first reported the use of continuous-wave Doppler ultrasound for the assessment of extracranial cerebral vessels. Despite its rapid development in other medical fields, this technique was not applied to the intracranial vessels until 1982. At that time, Aaslid and colleagues2 developed a transcranial Doppler (TCD) device with a pulsed-wave sound emission of 2 MHz that could successfully penetrate the skull and accurately measure blood flow velocities in the basal arteries and the circle of Willis. With the introduction of TCD, it became possible to record intracranial blood flow velocity directly, and TCD became an important noninvasive method for assessing cerebral hemodynamics and for evaluating intracranial cerebrovascular disease. The continuous development and refinement of ultrasonography during the past two decades lead to a broad spectrum of clinical TCD applications. The introduction of transcranial color-coded duplex sonography (TCCS) into clinical use was an important technical refinement. TCCS combines B-mode imaging with frequency-based color flow imaging and Doppler sonography.3 By means of TCCS, direct on-line visualization of the basal cerebral arteries and their flow directions became possible, allowing for angle-corrected measurements of blood flow velocities at defined depths. Subsequently, power-based4 and three-dimensional TCCS5 were added, and ultrasound contrast agents were introduced,6 further enhancing the diagnostic capability of this innovative technique7 (see Chapter 4).
Ultrasound contrast agents have likewise provided the opportunity to detect right-to-left cardiac shunts8 and to perform perfusion studies of the brain parenchyma based on indicator dilution principles.9
The detection of microembolic signals (MESs) by means of TCD constitutes another developmental landmark by allowing the noninvasive estimation of microemboli reaching the intracranial arteries.10
A recent development of transcranial ultrasonography, the therapeutic use of ultrasound-assisted thrombolysis (sonothrombolysis),11 has opened a new era of neurovascular ultrasound.
Examination Techniques
General Prerequisites
Two prerequisites should be fulfilled before performing a TCD examination: (1) the status of the extracranial arteries has to be known, and (2) the patient needs to rest comfortably to avoid major fluctuations in blood carbon dioxide levels and movement artifacts. In addition, two main anatomic considerations must be dealt with by the examiner: (1) the ultrasonic “windows” through which the ultrasound beam can penetrate the skull are often limited or difficult to identify; and (2) the arteries at the base of the skull vary greatly in respect to size, course, development, and site of access.12–15 The transmission of ultrasound signals through the cranium has been extensively studied.16,17 It depends on the skull structure, which consists of three layers, each of which influences ultrasound transmission in different ways. Grolimund17 has performed a number of in vitro experiments showing that a wide range of energy loss occurs in different skull samples, and that the energy loss varies greatly from one location to another and among individuals. In no case was the power measured behind the skull greater than 35% of the transmitted power. It was further shown that the skull can provide the effect of an acoustic lens, and that refraction or distortion of the beam depends more on the variation of bone thickness than on the angle of insonation.
Ultrasonic Windows
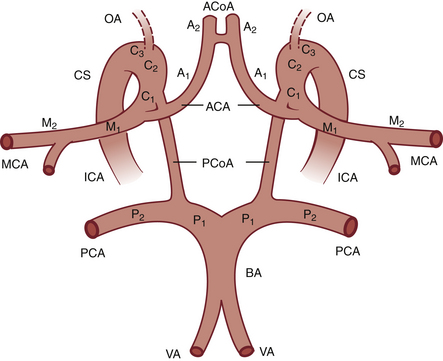
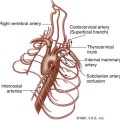
Four main ultrasound approaches have been described to insonate the intracranial arteries: the transtemporal, transorbital, suboccipital (i.e., transforaminal), and submandibular approaches,18,25 as illustrated in Figure 12-1. An extensive nomenclature has been developed for describing the segments of the intracranial cerebral arteries, and this terminology is used in this chapter. If you are unfamiliar with cerebral artery nomenclature, please refer to Figure 12-2.
Transtemporal Approach
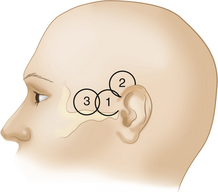
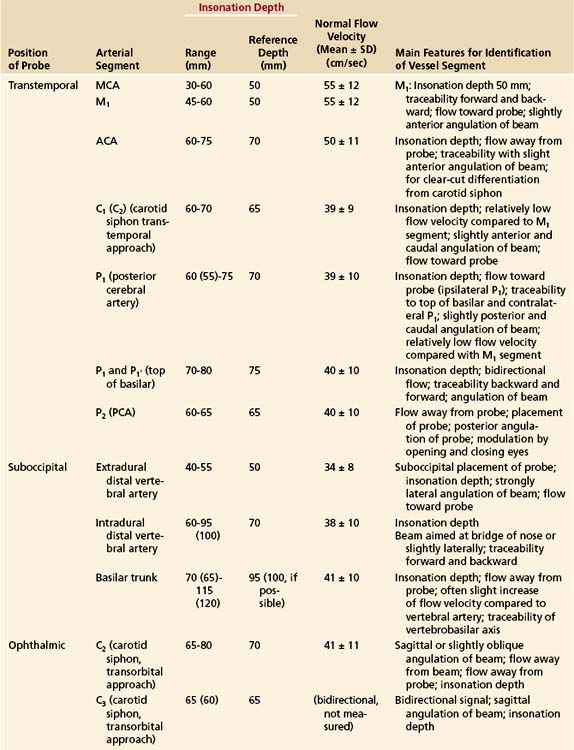
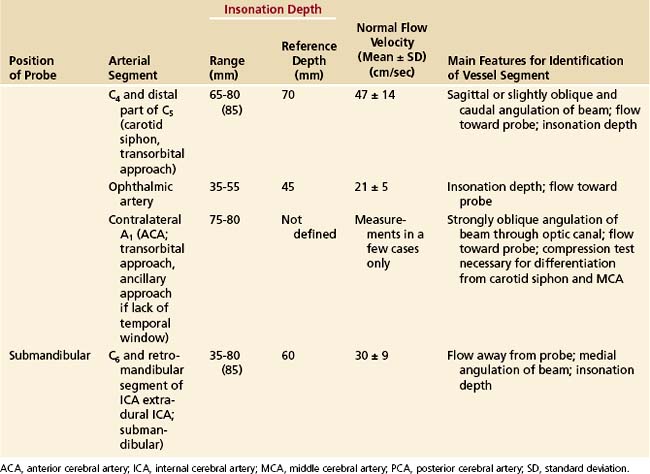
The probe is placed on the temporal aspect of the head, cephalad to the zygomatic arch and immediately anterior and slightly superior to the tragus of the ear conch (Figure 12-3, position 1). This is usually the most promising examination site. A more posterior window immediately cephalad and slightly dorsal to the first one (Figure 12-3, position 2) may be more appropriate in a minority of cases, especially for insonation of the P2 segment of the posterior cerebral arteries (PCAs). In some patients, a more frontally located temporal ultrasonic window may be present (Figure 12-3, position 3). By using these transtemporal approaches, the beam can be angulated anteriorly or posteriorly relative to the corresponding probe positions on the opposite side of the head. The anterior orientation of the beam allows for the insonation of the M1 and M2 segments of the middle cerebral arteries (MCAs), the C1 segment of the carotid siphon (CS), the A1 segment of the anterior cerebral artery (ACA), and often the anterior communicating artery (Figure 12-4, A). The posteriorly angulated beam insonates the P1 and P2 segments of the PCA, the top of the basilar artery (BA), and the posterior communicating arteries (Figure 12-4, B).
Transorbital Approach
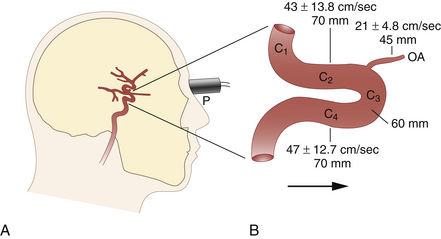
Components of the anterior cerebral circulation may be evaluated by placing the transducer against the closed eyelid.13 To avoid damage to the lenses of the eyes, the power of the ultrasound transmission has to be reduced. The ophthalmic artery can usually be insonated at depths of 45 to 50 mm, whereas the C3 segment (anterior knee of the CS) is normally met at insonation depths of 60 to 65 mm (Figure 12-5, A). At slightly greater insonation depths of 70 to 75 mm, the C2 segment shows flow away from the probe (upward deflection), and the C4 segment shows flow toward the probe (downward deflection). These flow directions apply only when the beam is nearly sagittal (slight medial obliquity) and enters the skull through the supraorbital or infraorbital fissures. Typical insonation depths and velocities are shown in Figure 12-5, B. The transorbital approach is much less established and validated than the transtemporal or suboccipital approach.
Suboccipital (Transforaminal) Approach
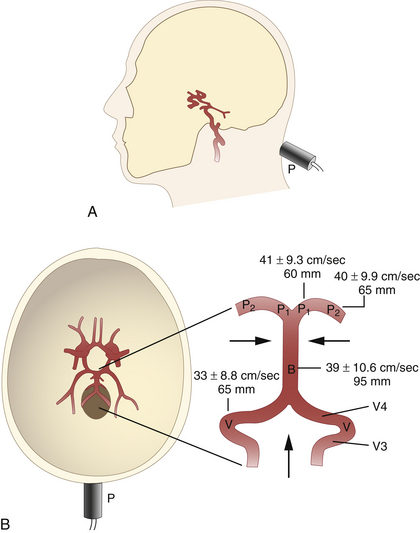
The suboccipital (transforaminal) approach is essential for screening the distal vertebral artery (VA; V4 segment) and the BA throughout its entire length. The probe is placed exactly between the posterior margin of the foramen magnum and the palpable spinous process of the first cervical vertebra, with the beam aimed at the bridge of the nose (Figure 12-6, A).2 The insonation depth is set at 65 mm, and the right and left VAs are tracked individually from this (deepest) point back toward the foramen magnum, using progressively smaller insonation depths (from 65 down to 35 mm). As the depth decreases, the sound beam is angled more and more sharply toward the side of the head. The extradural part of the VA, on the posterior arch of the atlas (V3 segment), can also be screened. Flow is toward the transducer in this segment. The BA can be tracked cephalad from the point at which the VAs unite. The superior end of the BA is reached at a depth of approximately 95 to 125 mm. Flow in the intradural VAs and the BA is normally directed away from the probe. Typical insonation depths and flow velocities are shown in Figure 12-6, B.
Submandibular Approach
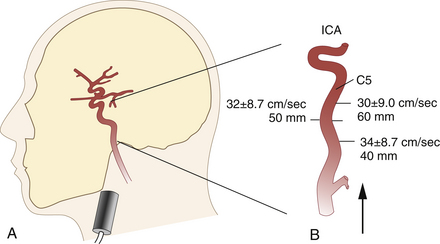
The submandibular approach completes the examination in that the retromandibular and more distal extradural parts (C5-C6 segments) of the internal carotid artery (ICA) can be evaluated. This particular examination is a useful complement to extracranial studies, because it facilitates the detection of ICA dissection and chronic ICA occlusion with abundant collateralization through the external carotid artery. With the transducer positioned as shown in Figure 12-7, A, the beam is directed slightly medially and posteriorly. The ICA can regularly be tracked to a depth of 80 to 85 mm, at which point it bends medioanteriorly to form the CS. Typical insonation depths and flow velocities are shown in Figure 12-7, B.
Diagnostic Approach
Basic TCD Examination
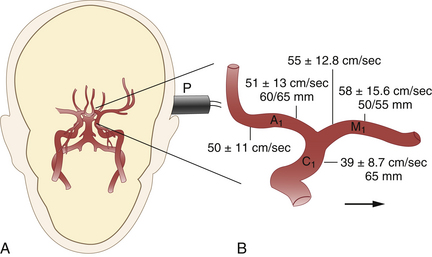
In general, it is most convenient to start with transtemporal insonation, to identify the MCA on either side at an insonation depth of 50 to 55 mm, and then to track the ipsilateral arterial network, step by step, in various directions. Proof of traceability of the MCA is necessary for its unequivocal identification. This is also true for other arteries at the base of the brain. Traceability refers to the fact that the MCA (and usually other arteries) can be tracked in incremental steps from a more shallow insonation depth (35 mm) to deeper sites (55 mm) without changes in the character of the flow profile and flow direction. When tracking the MCA medially (65-70 mm), an abrupt change in flow direction (away from, rather than toward the probe) indicates insonation of the A1 segment of the ACA. Flow signals toward the probe at this depth usually emanate from the CS at its junction with the MCA. Typical depths and flow velocities are shown in Figure 12-8.
By angling the beam more posteriorly from a transtemporal approach, the P1 segment of the PCA can be picked up most readily at an insonation depth of 65 to 70 mm. The PCA can then be tracked to the top of the BA (75 mm) and from there to the contralateral PCA (80-85 mm) (see Figure 12-4, B). The two criteria of traceability (i.e., the display of bilateral blood flow at the junction with the BA and the change of flow direction within the contralateral PCA) are very important features for identifying the PCAs without compression tests.
After the completion of the examination from both temporal windows, additional information may be obtained through the orbital, suboccipital, or submandibular pathways. The vessels that are accessible from these sites, as well as the techniques for identifying these vessels, were described previously. A protocol for TCD examination is presented in Table 12-1.
TCCS Examination
TCCS is now a well-established diagnostic method, allowing direct noninvasive imaging of intracranial vascular structures.3,19 This visual approach provides for more rapid and reliable vessel identification, permitting exact localization of the Doppler sample volume, and shortening the examination time.19,20 This technique has evolved rapidly over the past years and includes not only vascular (i.e., arterial and venous) imaging but also imaging of the brain parenchyma. Usually the transtemporal and suboccipital approaches are used for TCCS examinations. No systematic data exist for the submandibular approach. The transorbital TCCS investigation has been performed chiefly by ophthalmologists until now, and thus systematic data are mainly available in ophthalmologic journals.21
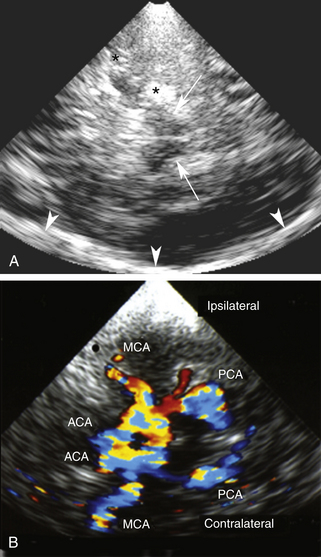
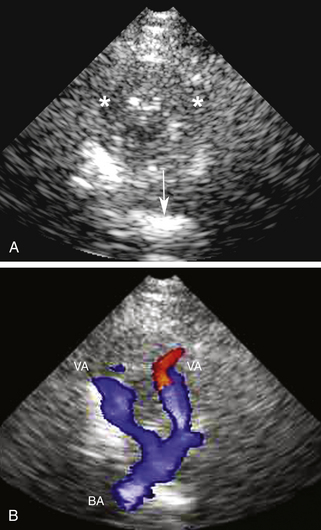
For transtemporal insonation, the probe is positioned axially along the orbitomeatal line and the hypoechoic, butterfly-shaped midbrain is visualized as an anatomic landmark at a depth of 6 to 8 cm. From this perspective, the circle of Willis can then easily be depicted (Figure 12-9). For the suboccipital approach, the hypoechoic foramen magnum and the hyperechoic clivus serve as the anatomic landmarks, with both VAs located at their lateral edges (Figure 12-10). The origin of the BA can also be visually identified in most cases at a depth of 75 to 95 mm. Generally, the reference depths of the target vessels are similar to the values given previously for TCD examination. TCCS also allows for the examination of cerebral venous sinuses and large basal cerebral veins, although this has not become part of the clinical routine.
Vessel Identification
The primary TCD parameters for identifying the cerebral arteries are the following:
2. Direction of blood flow at insonation depth
3. Flow velocity (mean flow velocity and systolic or diastolic peak flow velocity)
4. Probe position (e.g., temporal, orbital, suboccipital, submandibular)
5. Direction of the ultrasonic beam (e.g., posterior, anterior, caudad, cephalad)
Compression of the extracranial carotid arteries, as a means for intracranial vessel identification, has gradually been excluded from the clinical routine because of the low, but definite, risk for cerebral embolism.22,23 This is especially the case since the advent of TCCS used in conjunction with ultrasound contrast agents, because the identification of the major cerebral arteries and their collateral pathways is possible, for the most part, without compression maneuvers. Carotid compression should be definitely avoided in patients with extracranial atheromatous disease.
Blood Flow Velocity Measurements
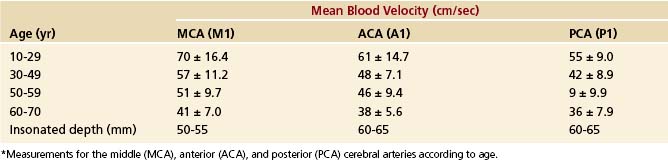
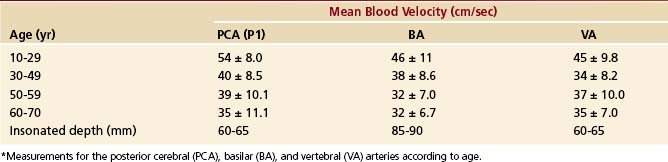
The mean blood flow velocities of various arterial segments, and their age dependency, are shown in Tables 12-2 and 12-3.24 Normal flow velocity values in adults show little variation among different investigators.14,25–27 The highest velocities are almost always found in the MCA or the ACA. The PCAs and BAs have lower Doppler shifts than the MCA in normal subjects. The same pattern has not been noted, however, in cerebral blood flow studies, in which flow is measured in cubic centimeters per second. Two explanations have been offered for this discrepancy between velocity and volume flow: (1) The measurement sites may be different28 or (2) more probably, different velocities occur as a compensatory mechanism to keep volume flow constant in arteries of different size.25 Thus, velocities are slower in large vessels and faster in small ones. Normal angle-corrected blood flow velocity values using TCCS have likewise been established and are only slightly higher than those obtained with TCD.20,29 The TCD documentation of decreasing flow velocities with increasing age20,27 correlates well with age-related changes in cerebral blood flow28 and underlines the validity and sensitivity of TCD and TCCS data as a semiquantitative estimate of cerebral blood flow.
Functional Reserve Testing
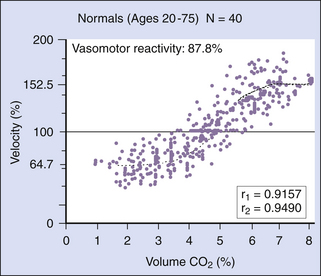
Transcranial Doppler is an ideal functional test for detecting rapid changes in cerebral perfusion, because the technique provides excellent resolution of flow velocity changes occurring over time. Functional tests are predominantly aimed at the evaluation of the reserve mechanism of the cerebral vasculature, using various stimuli such as hypocapnia or hypercapnia, shift in the pH by acetazolamide, increased or reduced systemic arterial pressure, and hypoxia. The CO2 (carbon dioxide) dilatory effect is mainly restricted to the peripheral arterial vascular bed, particularly the small cortical vessels.28 With changing CO2 concentrations, the relationship between flow velocity and volume flow within a large cerebral artery is linear,30 provided that the CO2 level does not directly affect the diameter of the large proximal arterial segment.31 Velocities measured from the MCA with changing CO2 concentrations show a biasymptotic, S-shaped curve (Figure 12-11).
A “preserved” vasomotor reserve implies that a drop in perfusion pressure can be counterbalanced by vasodilatation of cortical arterioles to maintain sufficient cortical blood supply. The vasomotor reserve may become exhausted if the resistance vessels in brain areas with low perfusion pressure are already maximally dilated.30–33 In this state, the resistance vessels are refractory to any further vasodilatory stimuli, and hypercapnia cannot increase blood flow. This condition may be critical, because ischemic brain injury can occur if the perfusion pressure is further reduced for any reason. Measurements of the vasomotor reserve capacity are useful in evaluating the hemodynamic impact of extracranial occlusive carotid disease.
The pulsatility index, as defined by Gosling (see Chapter 3), reflects the resistance in the peripheral vascular bed and has been suggested as a sensitive index of diastolic runoff—that is, with increased peripheral vasodilatation, diastolic runoff is expected to increase and the pulsatility index to decrease.34 However, in a large series of patients with carotid artery occlusion, the pulsatility index appeared to be much poorer for predicting the intracranial hemodynamic situation than the vasomotor reserve capacity.35
Diagnostic Parameters for Specific Clinical Applications
Intracranial Stenosis and Occlusion
The detection of carotid siphon (CS) stenosis using TCD was first reported in 1986 by Spencer and Whisler,13 who used similar criteria to those used for carotid bifurcation disease. Since then, a number of authors have reported similar findings for the CS and have extended TCD applications to other brain arteries.36–39 The most obvious clinical advantage of ultrasound is the rapid screening of the acute stroke patient for intracranial vessel obstruction. Normal TCD findings in stroke patients have considerable clinical impact.
Definition of Stenosis With TCD
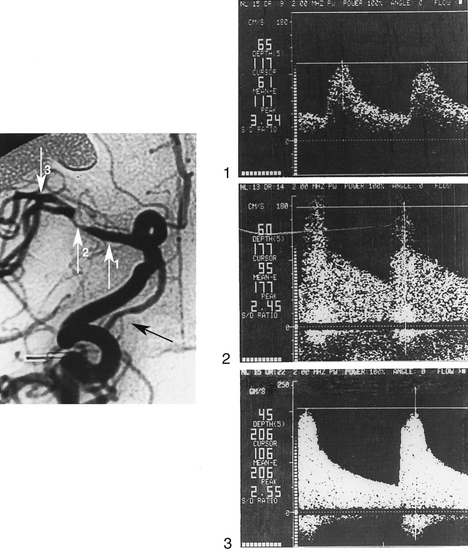
The following are typical TCD features of circumscribed stenosis of a large basal cerebral artery (Figure 12-12): (1) increased flow velocity; (2) disturbed flow (spectral broadening and enhanced systolic and low-frequency echo components); and (3) covibration phenomena (vibration of vessel wall and surrounding soft tissue).25,35 It is unclear whether the PSV (>120-160 cm/sec) or the mean systolic velocity (>80-120 cm/sec) should be used as a threshold value.39 With a mean velocity value of 100 cm/sec, a sensitivity of 100%, and a specificity of 97.9%, as well as positive and negative predictive values of 88.8% and 94.9%, were reported in detecting intracranial stenoses with a diameter of 50% or more.38 For the vertebrobasilar system, a threshold of more than 2 kHz peak systolic Doppler shift showed a sensitivity of 80% and a specificity of 97% in detecting stenoses of 50% or more.37 Most authors agree that, in comparison with the contralateral vessel segment, a relative increase in PSV of more than 30% is suspicious for hemodynamically significant stenosis, and a relative increase of more than 50% indicates a definite intracranial artery stenosis.
Definition of Occlusion With TCD
Basal cerebral artery occlusion can be detected by three observations: (1) the absence of arterial signals at an expected depth; (2) the presence of signals in vessels that communicate with the occluded artery; and (3) altered flow in communicating vessels, indicating collateralization. For example, occlusion of the MCA is diagnosed from the lack of an MCA signal in the presence of flow signals from other vessels (i.e., the PCA, the ACA, or the distal CS). This combination of findings also confirms that the temporal window is satisfactory. In a recent study, TCD showed a sensitivity of 83% and a specificity of 94.4%, with an overall accuracy of 91.6%, in the detection of intracranial vessel occlusion.40 In accordance with the well-established, angiography-based Thrombolysis in Myocardial Infarction (TIMI) criteria in cardiology, Demchuk and colleagues41 have proposed the so-called Thrombolysis in Brain Ischemia (TIBI) criteria for the TCD-based classification of the MCA status during and after thrombolysis. The TIBI scale, ranging from 0 (MCA occlusion) to 5 (normal MCA), is given in Table 12-4
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree