24 Ultrasound Diagnosis of Venous Insufficiency
Pathophysiology of Venous Insufficiency
Normal venous anatomy and physiology were described in Chapters 20 and 21. It is necessary to appreciate that venous valves are present throughout the lower extremity venous system in the deep, superficial, and perforating veins. The concentration of valves is higher in the calf veins than in the deep veins of the thigh.
Chronic venous insufficiency may affect only the superficial veins, or it may be related to deep venous thrombosis. The valves below the knee are most often implicated in the clinical sequelae of venous thrombosis. Patients who develop ulceration following an episode of deep venous thrombosis quite often exhibit both deep venous incompetence and incompetence of the great and small saphenous veins. It is of interest to note that patients without significant incompetence of the deep veins below the knee may not suffer from ulceration if there is normal valvular function in the superficial veins.
Clinical Signs and Symptoms
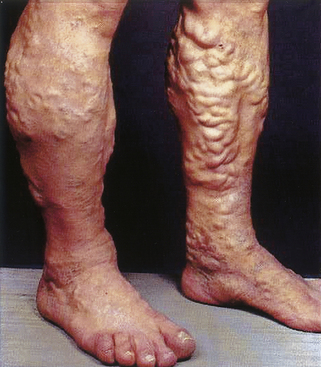
The increased venous pressure that results from incompetence of the superficial vein valves causes dilatation of the superficial veins in the distal extremity (Figure 24-1). This is generally noted first on the medial aspect of the lower calf and around the ankle. With progressively worsening dysfunction, the veins become enlarged and tortuous.
Patients with valvular dysfunction frequently complain of a feeling of heaviness and aching in the legs after prolonged standing or sitting with the legs dependent. In patients with valvular incompetence in the absence of venous obstruction, the feeling may subside with walking or with elevation of the limb, actions that relieve venous congestion. In contrast, if the deep veins are obstructed, exercise results in venous claudication, consisting of severe cramping, burning pain that persists as long as the veins remain congested. Several investigators have shown that venous claudication is caused by a rapid increase in pressure in both the superficial and deep venous systems.1,2 This is usually the result of obstruction of the iliofemoral venous segment with inadequate collateral compensatory flow.
Vascular Laboratory Test Procedures
Historically, investigators relied on the invasive procedures (namely, ascending and descending venography and ambulatory venous pressure measurements) to evaluate chronic venous insufficiency. Venography was considered to be the gold standard for visualization of anatomy, confirmation of the presence of venous obstruction and collateralization, and definition of the location and extent of valvular reflux. Ambulatory venous pressure measurements were used as a hemodynamic complement to the anatomic information obtained from venography.3 Pressures could be measured with the patient at rest in a supine position, while standing, and during exercise. This procedure had value as a means for recording venous pressure recovery time, which has been used as the basis for more recent plethysmographic studies.
The modern vascular laboratory evaluation of venous insufficiency has evolved steadily from continuous-wave Doppler velocimetry to indirect, plethysmographic procedures and finally, to quantification of venous reflux using duplex ultrasound imaging. Although duplex sonography is currently the most accurate method for assessing venous incompetence, continuous-wave Doppler remains in use as a convenient, “low-tech” method for diagnosing venous reflux. This technique is therefore included in this chapter. Plethysmographic methods also remain in clinical use as a means of assessing venous hemodynamics, and for that reason, they too are discussed.
Bidirectional Continuous-Wave Doppler
Equipment
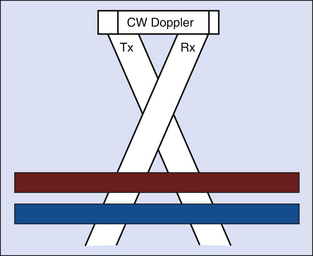
Bidirectional, continuous-wave Doppler uses separate transmitting and receiving crystals that operate continuously to detect flow at all depths along the emitted sound beam (Figure 24-2). Because of this, the signal that is received may contain echoes from more than one vessel lying within the beam path. The depth of penetration of a sound beam is inversely proportional to the carrier Doppler frequency. For this reason, lower-frequency transducers (3-5 MHz) are required when studying the deeper veins of the thigh, whereas higher frequencies (8-10 MHz) may be used for evaluating the superficial and calf veins of patients with a normal body habitus.
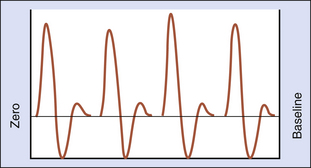
Quadrature phase separation is used to detect the direction of flow. The analog waveform of the Doppler signal is displayed using a frequency-to-voltage converter and a zero-crossing detector. The voltage output is proportional to the number of zero crossings (Figure 24-3). This display method is highly dependent on the signal-to-noise ratio and on the amplitude of the return signal.4,5
Examination Technique and Diagnostic Criteria
Normal venous blood flow is spontaneous and phasic during respiration, yielding a wind-like audible Doppler signal. Manual compression of the limb below the probe should augment forward flow, with resultant increased amplitude of the audible Doppler signal. When the limb is compressed above the probe, the Doppler signal will normally cease, because competent valves restrict retrograde venous flow. When compression above the probe is released, an augmented, forward flow signal should be noted. Blood flow signals will also decrease when the patient coughs or performs a Valsalva maneuver. Both actions cause an increase in intra-abdominal pressure, which restricts escape of blood from the lower limb. The same compression and/or respiration procedure is repeated over the femoral, popliteal, and posterior tibial veins and in the regions of the saphenofemoral and saphenopopliteal junctions. Spontaneous Doppler signals are most often present in the larger-diameter thigh and popliteal veins. If the patient is examined in a cool room, however, vasoconstriction may reduce extremity blood flow, and augmentation of flow signals may be required to confirm patency of the small-diameter tibial veins.
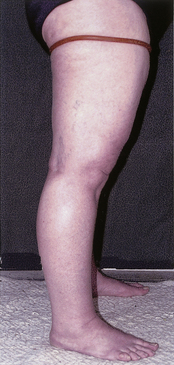
The anatomic location of valvular incompetence can be inferred by simple compression maneuvers that exclude the superficial venous system during the examination. The continuous-wave Doppler probe is placed over the region of the saphenofemoral junction, and the presence of retrograde flow is confirmed with release of compression of the limb below the level of the probe. A tourniquet is placed around the limb approximately 10 cm distal to the expected location of the saphenofemoral junction and tightened sufficiently to compress the great saphenous vein (Figure 24-4). Compression of the limb below the level of the probe is repeated. The continued presence of reversed blood flow signals suggests incompetence of the common femoral and/or proximal femoral vein(s). If retrograde blood flow is abolished by tourniquet application, incompetence of the great saphenous vein is suggested. The saphenopopliteal junction should be examined in a similar manner to distinguish popliteal/gastrocnemius reflux from incompetency of the small saphenous vein.
Advantages
In the hands of experienced examiners, bidirectional, continuous-wave Doppler velocimetry has been shown to have excellent sensitivity (92%) with acceptable specificity (73%) for the assessment of venous incompetence.6 While some applaud this method as a valuable portable tool for detection of valvular incompetence or obstruction of the deep and superficial veins,7–9 others note that the continuous-wave Doppler test is extremely operator dependent and subjective.10
Limitations
It is important to be aware of the considerable limitations associated with continuous-wave Doppler examination of the extremity veins. Because this is a nonimaging modality, there is no way to be certain which veins are being insonated. Duplication of the deep and superficial veins is common, and a Doppler signal may be elicited from a patent vein that lies adjacent to a thrombosed venous segment or from a large collateral vein. It is often quite difficult to differentiate reflux in the deep venous system from reflux in a superficial vein or major tributary at the saphenofemoral and saphenopopliteal junctions. Similarly, incompetence of large perforating veins may be confused with reflux in the saphenous or deep veins. Finally, standardization of the testing protocol is not possible because of the variability associated with tourniquet application. There is no assurance that the superficial veins are adequately compressed or that the compression does not obliterate flow in the deep venous system or perforating veins.
Photoplethysmography
Equipment
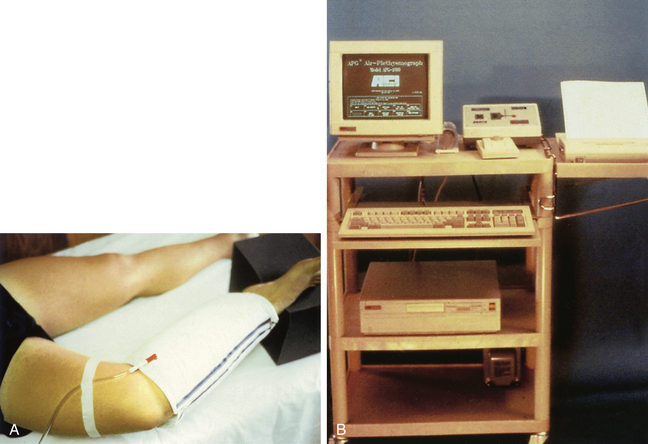
Photoplethysmography is a relatively simple tool used to screen for valvular incompetence. This technique employs an infrared light-emitting diode, with a second diode used to sense light reflected from subdermal venous flow. The photoplethysmographic probe is most commonly affixed to the skin in the supramalleolar region, using double-stick tape (Figure 24-5). The plethysmograph is coupled to a direct current recorder (DC mode) to track the average changes in reflected light that occur over time in association with alterations in blood flow volume. In the normal limb, the volume of blood in the skin decreases in response to manual compression of the calf or dorsiflexion of the foot and ankle. In the absence of obstruction to arterial inflow, the venous microcirculation refills slowly. If venous valves are incompetent, however, reflux occurs and the microcirculation refills rapidly. The quality of venous emptying with calf muscle compression can be assessed subjectively, and the length of time required for venous refill can be calculated from the calibrations on the strip chart recording.
Examination Technique and Diagnostic Criteria
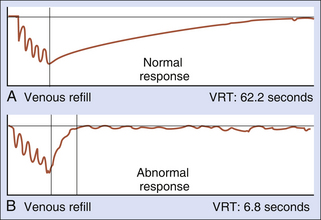
The patient is then requested to dorsiflex the foot four or five times. This causes the calf muscles to contract, simulating ambulation, and empties the calf veins in normal individuals. Manual calf compression can be used for patients who are unable to achieve adequate emptying of the venous pool with dorsiflexion. When the leg is relaxed and immobile, the calf veins refill. The venous refilling time is defined as the number of seconds required for the photoplethysmographic tracing to reach a stable endpoint for at least 5 seconds. The refill time is measured from the time exercise ceases to the stable endpoint (Figure 24-6, A). As noted previously, normally there is a rapid reduction of venous volume (and venous pressure) with limb exercise. Capillary refilling is primarily a function of arterial inflow when vein valves are competent and venous refilling is relatively slow. In patients with competent deep and superficial veins, the venous refill time is lengthened and usually exceeds 20 seconds.
A venous refill time less than 20 seconds suggests venous insufficiency (Figure 24-6, B). Superficial venous reflux can be differentiated from deep venous reflux by application of tourniquets to compress the great and small saphenous veins. A tourniquet (latex tubing or blood pressure cuff inflated to 45 mm Hg) is initially placed above the knee. The test is repeated as described previously. If the venous refill time normalizes to longer than 20 seconds, the superficial venous system is implicated as the source of incompetence. If the refill time improves but does not normalize, the data imply that both the deep and superficial systems are incompetent. The tourniquet is then moved below the knee. If the refill time normalizes, this is diagnostic of superficial venous incompetence alone. If the refill time remains less than 20 seconds with tourniquet compression of the superficial veins, this suggests deep venous insufficiency.
Advantages
Photoplethysmographic determination of venous refill time correlates with ambulatory venous pressure measurements.11 The application is technically simple, and the equipment is inexpensive and portable. This modality serves as a useful screening tool for evaluation of patients in whom venous insufficiency is suspected on the basis of history or physical findings.
Air Plethysmography
Air plethysmography (APG) was first introduced in the 1960s to study lower extremity volume changes that occur in response to alterations in posture and muscular exercise. Once it became possible to calibrate the system, interest was renewed in this noninvasive modality that could replace the older diagnostic devices such as strain gauge, segmental volume, and water plethysmographs. Christopoulos and colleagues12 introduced APG as a diagnostic tool in 1987 to detect global limb volume changes that occur with exercise and gravity.
Equipment
APG uses an air-filled, polyvinyl cuff, which surrounds the calf and functions as a sensing device to detect calf volume changes. The cuff is connected to an air-calibrated pressure transducer, amplifier, and recorder (Figure 24-7).
Patient Positioning
The patient initially reclines in the supine position with the heel slightly elevated on a support and the limb externally rotated and flexed to allow application of the cuff. Volume changes in the limb are recorded during limb elevation (which empties the veins), venous refilling, and a series of maneuvers with the patient upright, as shown in Figure 24-8 and described in the following section.
Examination Technique and Diagnostic Criteria
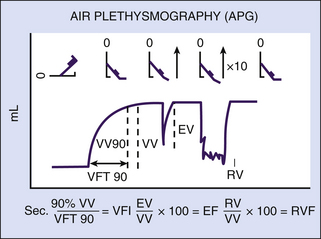
With the patient’s heel supported and the limb properly positioned, the air-filled cuff is adjusted over the calf so that it encloses the calf from the knee to the ankle. The patient’s limb is elevated 45 degrees to empty the calf veins (see Figure 24-8). Maximal venous emptying is indicated when the baseline recording stabilizes. The patient is then quickly brought to a standing position with the weight supported on the opposite limb. Filling of the calf veins is recorded continuously until a steady baseline is again obtained. This indicates that functional venous volume (VV) has been reached. Venous filling should result in an increase in leg VV of 100 to 150 mL in limbs with competent vein valves and 100 to 350 mL in limbs with venous insufficiency.
The venous filling index (VFI) is the ratio of 90% of the VV divided by the time required to achieve 90% venous filling (venous filling time, or VFT90%). The VFI, which evaluates overall valvular competence, is calculated from the equation VFI = 90% VV/VFT90%. This measurement of average filling rate is expressed in milliliters per second. A VFI of 2 mL/sec or less indicates normal valvular function, while a VFI greater than 7 mL/sec is consistent with deep and/or superficial incompetence and is associated clinically with symptoms of chronic venous insufficiency. Application of a narrow below-knee tourniquet to occlude the small and great saphenous veins may reduce the VFI to less than 5 mL/sec in limbs with incompetent common femoral vein valves but competent popliteal valves.13 Christopoulos and associates12 found that a VFI between 2 and 30 mL/sec was associated with superficial venous incompetence, while patients with a VFI between 7 and 28 mL/sec had evidence of deep venous insufficiency.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree